Use these links to rapidly review the document
TABLE OF CONTENTS
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 20-F
o |
REGISTRATION STATEMENT PURSUANT TO SECTION 12(b) OR (g) OF THE SECURITIES EXCHANGE ACT OF 1934 |
|
OR |
||
ý |
ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 For the fiscal year ended December 31, 2019 |
|
OR |
||
o |
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 For the transition period from to |
|
OR |
||
o |
SHELL COMPANY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
|
Date of event requiring this shell company report
Commission File Number 001-38178
| ZEALAND PHARMA A/S (Exact name of registrant as specified in its charter and translation of registrant's name into English) |
| The Kingdom of Denmark (Jurisdiction of incorporation or organization) |
Sydmarken 11 2860 Søborg (Copenhagen) Denmark (Address of principal executive offices) |
Emmanuel Dulac President and Chief Executive Officer Zealand Pharma A/S Sydmarken 11 2860 Søborg (Copenhagen) Denmark Tel: +45 88 77 36 00 Fax: +45 88 77 38 98 (Name, Telephone, E-mail and/or Facsimile number and Address of Company Contact Person) |
Securities registered or to be registered pursuant to Section 12(b) of the Act.
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||
|---|---|---|---|---|
| American Depositary Shares, each representing one ordinary share, DKK 1 nominal value per share | ZEAL | The Nasdaq Global Select Market | ||
Ordinary shares, DKK 1 nominal value per share* |
The Nasdaq Global Select Market* |
- *
- Not for trading, but only in connection with the registration of the American Depositary Shares. Securities registered or to be registered pursuant to Section 12(g) of the Act. None
Securities for which there is a reporting obligation pursuant to Section 15(d) of the Act. None
Indicate the number of outstanding shares of each of the issuer's classes of capital or common stock as of the close of the period covered by the annual report.
| 36,054,661 Ordinary Shares (Including Shares Underlying American Depositary Shares) |
||
2,726,647 American Depositary Shares |
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act.
Yes o No ý
If this report is an annual or transition report, indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934.
Yes o No ý
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days.
Yes ý No o
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files).
Yes ý No o
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, or a non-accelerated filer. See definition of accelerated filer and large accelerated filer in Rule 12b of the Exchange Act. (Check one):
| Large accelerated filer o | Accelerated filer ý | Non-accelerated filer o | Emerging growth company ý |
If an emerging growth company that prepares its financial statements in accordance with U.S. GAAP, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards†† provided pursuant to Section 13(a) of the Exchange Act. o
Indicate by check mark which basis of accounting the registrant has used to prepare the financial statements included in this filing:
| o US GAAP | ý International Financial Reporting Standards as issued by the International Accounting Standards Board |
o Other |
If "Other" has been checked in response to the previous question, indicate by check mark which financial statement item the registrant has elected to follow
o Item 17 o Item 18
If this is an annual report, indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act).
Yes o No ý
- †
- The term "new or revised financial accounting standard" refers to any update issued by the Financial Accounting Standards Board to its Accounting Standards Codification after April 5, 2012.
i
In this Annual Report on Form 20-F the terms the "Company", "Zealand Pharma", "Zealand" and the "Group" refer to the parent company Zealand Pharma A/S together with its consolidated subsidiaries. The term "Zealand Pharma A/S" is used when addressing issues specifically related to this legal entity.
Pursuant to Rule 12b-23 of the Securities Exchange Act of 1934, as amended (the "Exchange Act"), we incorporate information for certain items of this Annual Report on Form 20-F by reference to certain pages of the Zealand Pharma A/S statutory Annual Report 2019 (the "Annual Report 2019") included as Exhibit 99.1(a) to Form 6-K furnished to the SEC on March 13, 2020. Therefore, the information in this Annual Report on Form 20-F should be read in conjunction with the Annual Report 2019, as furnished to the SEC on March 13, 2020. Items not contained or not specifically referenced to within the Annual Report 2019 should not be deemed to be part of this Annual Report on Form 20-F.
FORWARD-LOOKING STATEMENTS
This Annual Report on Form 20-F contains forward-looking statements concerning our business, operations and financial performance and condition, as well as our plans, objectives and expectations for our business operations and financial performance and condition. Any statements contained herein that are not statements of historical facts may be deemed to be forward-looking statements. In some cases, you can identify forward-looking statements by terminology such as "aim," "anticipate," "assume," "believe," "contemplate," "continue," "could," "due," "estimate," "expect," "goal," "intend," "may," "objective," "plan," "predict," "potential," "positioned," "seek," "should," "target," "will," "would," and other similar expressions that are predictions of or indicate future events and future trends, or the negative of these terms or other comparable terminology.
These forward-looking statements include, but are not limited to, statements about:
- •
- our expectations regarding the potential market size and the size of the patient populations for our product candidates, if approved for
commercial use;
- •
- our expectations regarding the potential advantages of our product candidates over existing therapies;
- •
- our development plans with respect to our product candidates;
- •
- our ability to develop, acquire and advance product candidates into, and successfully complete, clinical trials;
- •
- the initiation, timing, progress and results of our preclinical studies and clinical trials, and our research and development programs;
- •
- the timing or likelihood of regulatory filings and approvals for our product candidates;
- •
- the implementation of our business model and strategic plans for our business, product candidates and technology;
- •
- the success of our license collaborations;
- •
- the uncertainties associated with our potential acquisition of substantially all assets of Valeritas, including, but not limited to, whether we are successful in our "stalking horse" bid, our ability to close and integrate the business and operations of Valeritas successfully and on a timely basis in order to obtain any anticipated benefits and costs savings from the transaction, the significant increase to the number of our employees as a result of the acquisition and the impact such
1
- •
- the uncertainties associated with the United Kingdom's exit from the European Union, or the EU, on January 31, 2020, following which EU
law will continue to apply in and to the United Kingdom for a transition period expiring on December 31, 2020 (subject to any extension agreed by the United Kingdom and the EU); and
- •
- estimates of our expenses, future revenue, capital requirements, our needs for additional financing and our ability to obtain additional capital.
increase may have on our operating results going forward, and our ability to identify and eliminate redundant and underperforming functions and assets;
With reference to our Annual Report 2019, examples of forward-looking statements can be found under the headings '2020 Outlook and objectives' in our Annual Report 2019 and elsewhere.
The forward-looking statements contained or incorporated by reference herein involve a number of known and unknown risks and uncertainties that could cause our future results, performance or achievements to differ significantly from the results, performance or achievements expressed or implied by such forward-looking statements.
You should understand that many important factors, in addition to those discussed or incorporated by reference herein, could cause our results to differ materially from those expressed in the forward-looking statements. Potential factors that could affect our results include, in addition to others not described in this report, those described under "Item 3-Key Information—D. Risk Factors." These are factors that we think could cause our actual results to differ materially from expected results.
Forward looking statements speak only as of the date on which they are made, and we undertake no obligation to update any forward-looking statements or other information contained in this report, whether as a result of new information, future events or otherwise. You are advised, however, to consult any additional disclosures we make in our reports on Form 6-K filed with the U.S. Securities and Exchange Commission (the "SEC"). Please also see the cautionary discussion of risks and uncertainties under "Item 3—Key Information—D. Risk Factors". This discussion is provided as permitted by the Private Securities Litigation Reform Act of 1995.
PRESENTATION OF FINANCIAL AND OTHER INFORMATION
The consolidated financial statements contained in this Annual Report on Form 20-F have been prepared in accordance with International Financial Reporting Standards, or IFRS, as issued by the International Accounting Standards Board, or IASB.
We maintain our books and records in Danish kroner (DKK) and report under IFRS as issued by the IASB. Our consolidated financial statements are not prepared in accordance with accounting principles generally accepted in the United States. We use the symbol "$" to refer to U.S. Dollars and the symbol "€" to refer to Euros herein.
This Annual Report on Form 20-F includes trademarks, tradenames and service marks, certain of which belong to us and others that are the property of other organizations or third parties. Solely for convenience, trademarks and tradenames referred to in this Annual Report on Form 20-F appear without the ® and ™ symbols, but the absence of those references is not intended to indicate, in any way, that we will not assert our rights or that the applicable owner will not assert its rights to these trademarks and tradenames to the fullest extent under applicable law.
Unless otherwise indicated, information contained in this Annual Report on Form 20-F concerning our industry and the markets in which we operate, including our general expectations, market position, market opportunity and market share, is based on information from our own management estimates and research, as well as from industry and general publications, research, surveys and studies conducted by third parties. Management estimates are derived from publicly available information, our knowledge
2
of our industry and assumptions based on such information and knowledge, which we believe to be reasonable. In addition, assumptions and estimates of our and our industry's future performance are necessarily subject to a high degree of uncertainty and risk due to a variety of factors, including those described in "Item 3—Key Information—D. Risk Factors". These and other factors could cause our future performance to differ materially from our assumptions and estimates. See "Forward-Looking Statements" above.
Enforceability of civil liabilities
The Company is a Danish corporation and a majority of its directors and officers, as well as certain experts named herein, are non-residents of the United States. A substantial portion of the assets of Zealand Pharma A/S, its subsidiaries and such persons are located outside the United States. As a result, it may be difficult for shareholders of the Company to effect service within the United States upon directors, officers and experts who are not residents of the United States or to enforce judgments in the United States. In addition, there can be no assurance as to the enforceability in Denmark against the Company or its respective directors, officers and experts who are not residents of the United States, or in actions for enforcement of judgments of United States courts, of liabilities predicated solely upon the federal securities laws of the United States.
ITEM 1 IDENTITY OF DIRECTORS, SENIOR MANAGEMENT AND ADVISORS
Not applicable.
ITEM 2 OFFER STATISTICS AND EXPECTED TIMETABLE
Not applicable.
A. SELECTED FINANCIAL DATA
The following tables present selected consolidated financial data for our business for the periods indicated. We derived the selected consolidated income statement and statement of financial position data as of December 31, 2019 and 2018 and for the years ended December 31, 2019, 2018, and 2017 from our audited consolidated financial statements, incorporated by reference from our Annual Report 2019 on pages 50-52. We derived the selected consolidated income statement data for the years ended December 31, 2016 and 2015 and statement of financial position data as of December 31, 2016 and 2015 from our audited consolidated financial statements not included or incorporated by reference herein with the exception for amounts that have been restated. You should read this data in conjunction with our consolidated financial statements and related notes appearing on pages 48-86 in our Annual Report 2019. Our historical results are not necessarily indicative of our future results.
The consolidated financial statements represents the consolidated financial statements of Zealand Pharma A/S and its subsidiaries.
3
Selected financial data
IFRS figure in DKK thousand, except per share numbers and number of shares |
2019 | Restated(1) 2018 |
Restated(1) 2017 |
Restated 2016 |
Restated 2015 |
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Income statement data |
||||||||||||||||
Revenue |
41,333 | 37,977 | 136,322 | 230,864 | 182,573 | |||||||||||
Operating result |
–587,942 | 652,385 | –248,526 | –110,271 | –79,681 | |||||||||||
Result before tax |
–576,677 | 625,051 | –279,913 | –154,035 | –118,186 | |||||||||||
Net result for the year |
–571,541 | 581,278 | –274,413 | –148,535 | –112,311 | |||||||||||
Earnings/loss per share data |
||||||||||||||||
Earnings/loss per share—basic (DKK) |
–16.91 | 18.94 | –9.85 | –6.11 | –4.87 | |||||||||||
Earnings/loss per share—diluted (DKK) |
–16.91 | 18.94 | –9.85 | –6.11 | –4.87 | |||||||||||
Statement of financial position data |
||||||||||||||||
Total assets |
1,599,514 | 1,229,797 | 721,285 | 683,116 | 627,621 | |||||||||||
Equity (net assets) |
1,242,673 | 1,116,281 | 514,669 | 267,381 | 244,803 | |||||||||||
Share capital (000 shares) |
36,055 | 30,787 | 30,751 | 26,142 | 24,353 | |||||||||||
Treasury shares (000 shares) |
64 | 64 | 64 | 564 | 564 | |||||||||||
Dividends per share |
0.00 | 0.00 | 0.00 | 0.00 | 0.00 | |||||||||||
Number of shares |
36,054,661 | 30,786,827 | 30,751,327 | 26,142,365 | 24,352,769 | |||||||||||
- (1)
- Reference is made to Note 1 'Significant accounting policies, and significant accounting estimates and assessments' on pages 54-60 and the 'Consolidated financial statements' on pages 50-52 in our Annual Report 2019 for further information.
B. CAPITALIZATION AND INDEBTEDNESS
Not applicable.
C. REASONS FOR THE OFFER AND USE OF PROCEEDS
Not applicable.
D. RISK FACTORS
Risks Related to Our Business
We have incurred net losses in recent periods and may continue to do so.
We recognized net losses of DKK 571.5 million in 2019, DKK 274.4 million in 2017 and DKK 148.5 million in 2016. Although we made a net profit of DKK 581.3 million in 2018, this profit was primarily as a result of the sale of future royalties and milestones from our Sanofi license agreement and may not necessarily be indicative of future periods, particularly because we have no ongoing source of revenue except certain development milestone payments under our other existing collaborations. Our losses have primarily been the result of our internal and external research expenditures and development costs for conducting preclinical studies and clinical trials in respect of our internal product portfolio. Our ability to generate revenue from our internal product portfolio depends on our ability to successfully develop and commercialize our product candidates and to obtain the regulatory and marketing approvals necessary to commercialize one or more of our product candidates.
Our ability and our collaboration partners' ability to generate future revenue from product sales or pursuant to milestone payments depend heavily on many factors, including, but not limited to:
- •
- completing research activities and preclinical and clinical development of our out-licensed and internal product candidates;
4
- •
- on our own, or together with our strategic collaboration partners, obtaining regulatory approvals for our product candidates;
- •
- negotiating favorable terms of and entering into further collaboration, licensing or other arrangements;
- •
- the ability of our collaboration partners to successfully commercialize or our ability to commercialize or co-promote our product candidates;
- •
- obtaining market acceptance of our product candidates, if approved;
- •
- addressing any competing technological or market developments;
- •
- identifying, assessing, acquiring, in-licensing or developing new product candidates;
- •
- maintaining, protecting and expanding our portfolio of intellectual property rights, including patents, trade secrets and know-how, and our
ability to develop, manufacture and commercialize our product candidates and products without infringing the intellectual property rights of others; and
- •
- attracting, hiring, and retaining qualified personnel.
In cases where we, or our collaboration partners, are successful in obtaining regulatory approvals to market one or more of our product candidates, our revenue will be dependent, in part, upon the size of the markets in the territories for which regulatory approval is granted, the price or prices at which we or our collaboration partners are able to sell such products and our ability to get paid or reimbursed for such products. If the number of individuals suitable for our product candidates is not as significant as we estimate, the indications approved by regulatory authorities are narrower than we expect, or the reasonably accepted population for treatment is narrowed by competition, physician choice or applicable guidelines, we may not generate significant revenue from the sale of such products, even if approved. Our failure to generate revenue from sales of one or more of our product candidates or pursuant to license or milestone payments or if the level of revenue generated therefrom is lower than our or the market's expectations, could have a material adverse effect on our business, financial position, results of operations and future growth prospects.
We expect our expenses to continue to increase and that we will continue to incur losses as we further develop our internal product portfolio. In particular, we anticipate that our expenses and losses will increase substantially if and as we:
- •
- continue the preclinical and clinical development of our internal product candidates;
- •
- expand the scope of or otherwise materially modify our current clinical trials for our internal product candidates;
- •
- begin new clinical trials for our internal product candidates;
- •
- develop our commercial manufacturing capabilities for our internal product candidates;
- •
- seek regulatory and marketing approvals for any internal product candidates that successfully complete clinical trials;
- •
- establish a sales, marketing and distribution infrastructure to commercialize any drugs for which we may obtain marketing approval and for
which we have not entered into a commercialization collaboration with a third party;
- •
- seek to identify and validate additional product candidates;
- •
- acquire or in-license product candidates and technologies;
- •
- maintain, protect and expand our intellectual property portfolio;
5
- •
- attract new and retain existing skilled personnel; and
- •
- create additional infrastructure to support our operations.
Any net losses we incur may fluctuate significantly from year to year, such that a year-to-year comparison of our results of operations may not be a good indication of our future performance. In any particular period or periods, our operating results could be below the expectations of securities analysts or investors, which could cause the price of our American Depository Shares, or ADSs, or the price of our ordinary shares to decline.
The regulatory approval processes of the FDA, the EMA and other comparable regulatory authorities are lengthy, time consuming and inherently unpredictable, and if we or our collaboration partners are ultimately unable to obtain regulatory approval for our internal or out-licensed product candidates, our business could be substantially harmed.
The time required to obtain approval by the U.S. Food and Drug Administration, or the FDA, the European Medicines Agency, or the EMA and other comparable regulatory authorities is unpredictable, but typically takes many years following the commencement of clinical trials and depends upon numerous factors, including the substantial discretion of the regulatory authorities. In addition, approval policies, regulations or the type and amount of clinical data necessary to gain approval may change during the course of a product candidate's clinical development and varies among jurisdictions. We have not obtained regulatory approval in the United States or in any other country for any product candidate for which we retain full development, commercialization and marketing control, and it is possible that none of our existing product candidates or any product candidates that we may seek to develop in the future will ever obtain regulatory approval. Although we intend to submit a new drug application, or NDA, for our dasiglucagon HypoPal Rescue Pen in early 2020, there is no assurance that FDA will accept the NDA for filing or that it will obtain approval.
Our product candidates could fail to receive regulatory approval for many reasons, including, but not limited to, the following:
- •
- the FDA, the EMA or other comparable regulatory authorities may disagree with the design or implementation of our clinical trials;
- •
- we or our collaboration partners may be unable to demonstrate to the satisfaction of the FDA, the EMA or other comparable regulatory
authorities that a product candidate is safe and effective for its proposed indications;
- •
- we or our collaboration partners may be unable to demonstrate that a product candidate's clinical and other benefits outweigh its safety risks;
- •
- the FDA, the EMA or other comparable regulatory authorities may disagree with our interpretation of data from preclinical studies or clinical
trials;
- •
- the data collected from clinical trials of our product candidates may not be sufficient to support the submission of a new drug application, or
NDA, the submission of a supplemental NDA or other submission or to obtain regulatory approval in the United States, Europe or elsewhere;
- •
- the FDA, the EMA or any other comparable regulatory authority may fail to approve the labeled conditions for use that we or our collaboration
partners propose for a product candidate;
- •
- the FDA, the EMA or other comparable regulatory authorities may fail to approve the manufacturing processes or facilities of any third party manufacturers with which we may contract for clinical and commercial supplies or such processes or facilities may not pass a preapproval inspection; and
6
- •
- the approval policies or regulations of the FDA, the EMA or other comparable regulatory authorities may change or differ significantly from one another in a manner rendering our clinical data insufficient for approval.
This lengthy approval process, as well as the unpredictability of ongoing clinical trial results, may result in our or our collaboration partners' failure to obtain regulatory approval to market our product candidates, which would harm our business, financial position, results of operations and future growth prospects significantly. In addition, even if we or our collaboration partners were to obtain approval, regulatory authorities may approve any of our product candidates for fewer or more limited indications than requested, may grant approval contingent on the performance of costly post-marketing clinical trials or may approve a product candidate with a label that does not include the labeling claims necessary or desirable for the successful commercialization of that product candidate. In certain jurisdictions, regulatory authorities may not approve the price we or our collaboration partners intend to charge for our products, which would reduce the ability to sell in that territory and therefore any payments or royalties we may receive. Any of the foregoing scenarios could materially harm the commercial prospects of our product candidates.
For certain marketed products, product candidates and clinical development programs, we depend on collaboration partners to develop and conduct clinical trials with, obtain regulatory approvals for, and market and sell our product candidates. If such collaboration partners fail to perform as expected, the potential for us to generate future revenue from such product candidates would be significantly reduced and our business would be significantly harmed.
For certain marketed products, product candidates and clinical development programs, we do, and may in the future continue to, rely on our collaboration partners to develop, conduct clinical trials of, and commercialize our product candidates and approved products. We have existing collaborations with Alexion Pharmaceuticals, Inc., or Alexion, Beta Bionics Inc., or Beta Bionics, and Boehringer Ingelheim GmbH, or Boehringer Ingelheim. We may also enter into collaboration agreements with other parties in the future relating to product candidates. Ultimately, if such out-licensed product candidates are advanced through clinical trials and receive marketing approval from the EMA, the FDA or similar regulatory authorities, certain of our collaboration partners will be responsible for commercialization of these out-licensed products. The potential for us to obtain future development milestone payments and, ultimately, generate revenue from royalties on sales of such out-licensed products depends on the successful development, regulatory approval, marketing and commercialization by our collaboration partners. If our collaboration partners do not perform in the manner we expect or fail to fulfill their responsibilities in a timely manner or at all, if our agreements with them terminate or if the quality or accuracy of the clinical data they obtain is compromised, the clinical development, regulatory approval and commercialization efforts related to our out-licensed product candidates could be delayed or terminated, and it could become necessary for us to assume the responsibility at our own expense for the clinical development of such product candidates. In that event, we would likely be required to limit the size and scope of efforts for the development and commercialization of such product candidate; we would likely be required to seek additional financing to fund further development or identify alternative strategic collaboration partners; our potential to generate future revenue from royalties and milestone payments from such product candidates would be significantly reduced or delayed; and it could have a material adverse effect on our business, financial position, results of operations and future growth prospects.
Collaborations involving our out-licensed product candidates pose a number of risks, including the following:
- •
- collaboration partners have significant discretion in determining the efforts and resources that they will apply to these partnerships;
7
- •
- collaboration partners may not perform their obligations as expected;
- •
- collaboration partners may not pursue development and commercialization of our out-licensed product candidates or may elect not to continue or
renew development or commercialization programs, based on clinical trial results, changes in the collaboration partners' strategic focus or available funding or external factors, such as an
acquisition, that divert resources or create competing priorities;
- •
- collaboration partners may delay clinical trials, provide insufficient funding for a clinical trial program, stop a clinical trial or abandon a
product candidate, repeat or conduct new clinical trials or require a new formulation of a product candidate for clinical testing;
- •
- collaboration partners may have or could independently develop, or develop with third parties, products that compete directly or indirectly
with our out-licensed product candidates;
- •
- disagreements with collaboration partners, including disagreements over proprietary rights, contract interpretation or the conduct of product
research, development or commercialization programs, may cause delays or lead to termination of such programs, or require us to assume unplanned expenditures, responsibilities or liabilities with
respect to product candidates we have out licensed, or may result in costly and time consuming litigation or arbitration;
- •
- collaboration partners may infringe the intellectual property rights of third parties, which may result in costly and time consuming litigation
or arbitration in which we may be involved, as a party or in support of our collaboration partners, and which may also have the effect of reducing the base on which royalties are calculated;
- •
- collaboration partners with marketing and distribution rights to one or more products may not commit sufficient resources to the marketing and
distribution of such product or products;
- •
- collaboration partners with marketing and distribution rights may incur costs that have the effect of reducing the base on which royalties are
calculated;
- •
- collaboration partners may infringe the intellectual property rights of third parties, which may expose us to litigation and potential
liability; and
- •
- collaboration agreements may be terminated and, if terminated, may result in a need for additional capital to pursue further development or commercialization of the applicable product candidates.
In addition, certain collaboration agreements provide our collaboration partners with rights to terminate such agreements and licenses granted under such agreements under various conditions, which, if exercised, would adversely affect our product development efforts, could make it difficult for us to attract new collaboration partners and may adversely affect our reputation. Our collaboration partners may have the right to terminate their respective collaboration agreements with us. Any such termination of any agreement or any future agreement that we may enter into with our collaboration partners could have a material adverse effect on our business, financial position and results of operations.
The timing and amount of any milestone and royalty payments we may receive under our agreements with our collaboration partners will depend on, among other things, the efforts, allocation of resources, and successful development and commercialization of our product candidates. We cannot be certain that any of the development and regulatory milestones will be achieved or that we will receive any future milestone payments under these agreements. In addition, in certain circumstances we may believe that we have achieved a particular milestone and the applicable collaboration partner may disagree with our belief. In that case, receipt of that milestone payment may be delayed or may never be received, which may require us to adjust our operating plans.
8
Pursuant to our Sale and Purchase Agreement with Royalty Pharma to sell and transfer the royalty streams from the Sanofi License Agreement, we have assigned our right to receive royalty revenue from the sales of Adlyxin/Lyxumia and/or Soliqua 100/33/ Suliqua, and with the exception of certain development milestone payments under our other existing collaborations, we have no ongoing source of revenue.
In 2003 we entered into our global license agreement, or the Sanofi License Agreement, with Sanofi-Aventis Deutschland GmbH, or Sanofi GmbH, a wholly-owned subsidiary of Sanofi S.A., or Sanofi. The Sanofi License Agreement granted Sanofi the exclusive worldwide rights to develop, manufacture, commercialize and market lixisenatide, both as a stand-alone product and combination therapy. Historically, the majority of our revenue has been derived from milestone payments made by Sanofi, as well royalty payments received from Sanofi on sales of these products.
Lixisenatide is out-licensed to and marketed by Sanofi both as a stand-alone therapy under the brand names Adlyxin in the United States and Lyxumia in the EU and in various other jurisdictions, and as a combination therapy with Lantus, the brand name of insulin glargine developed by Sanofi, under the brand name Soliqua 100/33 in the United States, and in some European countries under the brand name Suliqua.
In September 2018 we, together with two of our wholly-owned subsidiaries, entered into a purchase and sale agreement, or the Royalty Pharma Agreement, with Royalty Pharma Investments ICAV, or Royalty Pharma, to sell and transfer our and our subsidiaries' respective rights to receive royalties and $85 million of potential commercial milestones in respect of global net sales of Soliqua® 100/33/ Suliqua® and Lyxumia®/Adlyxin® from and after July 1, 2018, payable under the Sanofi License Agreement in consideration for an upfront one-time payment of $205 million. We and our subsidiaries also remain eligible to receive payments from Sanofi of up to $15 million upon the fulfillment of certain conditions, of which up to $5 million is expected in 2020 and up to $10 million is expected in 2022. However, we cannot be certain with regards to the timing and final amount of this payment, as both are dependent on factors that are outside of our control.
With the transfer to Royalty Pharma of all the royalties that we were due to earn under the Sanofi License Agreement, with the exception of the aforementioned payments of up to $15 million together with certain development milestone payments we may become entitled to under agreements with our other collaboration partners and potential future royalties should any products concerning those collaborations be approved, we currently have no source of revenue and are reliant on our cash on hand and potential capital raising efforts to fund the development of our internal pipeline of product candidates. Additionally, we have agreed to pay some of our revenue in deferred payments or royalties to third parties, including but not limited to a portion of any future payments we receive in respect of lixisenatide under the Sanofi License Agreement.
Further, while we are no longer directly exposed to the level of any royalty or milestone payments from Sanofi during the term of the Royalty Pharma Agreement, we have certain administrative obligations toward Royalty Pharma which require that we must maintain the intellectual property on lixisenatide and pay the relevant renewals thereon in a timely manner. In addition to this, certain of our obligations to Sanofi under the Sanofi License Agreement will also continue.
A failure by us to comply with the terms of the agreements with Sanofi and/or Royalty Pharma may place us in breach of our contractual obligations with either party and expose us to liability for indemnification of either party and/or may result in arbitration and/or litigation against us. An adverse ruling in such litigation may lead, to an award of significant damages, loss of profits and/or award of attorney fees against us.
9
We may need to raise additional funding, which may not be available on acceptable terms, or at all, and failure to obtain this capital when needed may force us to delay, limit or terminate our product development efforts or other operations.
We are currently advancing our internal product candidates through clinical development and are conducting preclinical studies with respect to other programs. Developing product candidates is expensive, lengthy and risky, and we expect our Research and Development, or R&D, expenses to increase in connection with our ongoing activities, particularly as we seek to advance our internal product candidates toward commercialization.
As of December 31, 2019, our cash and cash equivalents were DKK 1,081.1 million, and we had marketable securities of DKK 299.5 million. We expect that our existing cash and cash equivalents will be sufficient to fund our current operations for at least the next 12 months. However, our operating plans may change as a result of a variety of factors, and we may need to seek additional funds sooner than planned through public or private equity or debt financings, government or other third party funding, marketing and distribution arrangements and other collaborations, strategic alliances and licensing arrangements or a combination of these approaches. Any additional fundraising efforts may divert our management from their day-to-day activities, which may adversely affect our ability to develop and commercialize our product candidates. In addition, we cannot guarantee that future financing will be available in sufficient amounts or on terms acceptable to us, if at all. Moreover, the terms of any financing may adversely affect the holdings or the rights of our shareholders and the issuance of additional securities, whether equity or debt, by us, or the possibility of such issuance, may cause the market price of the ADSs to decline. The sale of additional equity or convertible securities could be dilutive to our shareholders. The incurrence of indebtedness would result in increased fixed payment obligations and we may be required to agree to certain restrictive covenants, such as limitations on our ability to incur additional debt, limitations on our ability to acquire, sell or license intellectual property rights and other operating restrictions that could adversely impact our ability to conduct our business. If we are unable to obtain adequate financing, we may be required to delay, reduce or eliminate the number or scope of our projects and internal product candidates (including our preclinical studies and clinical trial programs). We could also be required to seek funds through arrangements with collaboration partners or at an earlier stage than otherwise would be desirable and we may be required to relinquish rights to some of our technologies or internal product candidates or otherwise agree to terms unfavorable to us. If we are unable to obtain funding on a timely basis, we may be required to significantly curtail, delay or discontinue one or more of our research or development programs or the commercialization of any internal product candidate or be unable to expand our operations or otherwise capitalize on our business opportunities, as desired, which could impair our prospects.
We may not be successful in our efforts to use cash flows from our approved out-licensed products to expand our novel, internal target discovery platform to build a pipeline of product candidates.
A key element of our strategy is to use cash flows from our portfolio of approved, out-licensed drug products to build a pipeline of novel internal product candidates and progress these product candidates through clinical development for the treatment of a variety of diseases. Although our R&D efforts to date have resulted in the development of out-licensed product candidates directed at various diseases, we may not be able to develop additional product candidates in a sufficient timeframe, if at all, to provide for the further development of our pipeline of internal product candidates. Additionally, with our entering into the Royalty Pharma Agreement in September 2018, we and two of our wholly-owned subsidiaries transferred all the royalties and $85 million of potential commercial milestones that we were due to earn from the Sanofi License Agreement in exchange for a one-time upfront payment of $205 million. As a result we no longer have the rights to receive ongoing royalties in respect of global net sales of Soliqua® 100/33/ Suliqua® and Lyxumia®/Adlyxin® from and after July 1, 2018. Our
10
other ongoing collaborations with Alexion, Beta Bionics and Boehringer Ingelheim do not have any approved products, and other than milestone payments that may become due under our ongoing collaborations with Alexion, Beta Bionics and Boehringer Ingelheim upon the achievement of certain clinical milestones, we are not currently entitled to any royalty or other payments thereunder which could be used to help progress our internal pipeline of product candidates through clinical development. Our current internal product candidates are in late stages of clinical development and will require further clinical development and testing, and eventually regulatory approval, prior to commercialization. Even if we are successful in continuing to develop our out-licensed pipeline, the potential product candidates that we identify may not be suitable for clinical development, including as a result of being shown to have harmful side effects or other characteristics that indicate that they are unlikely to be products that will receive marketing approval and achieve market acceptance. If we do not continue to successfully develop our out-licensed product candidates and if these out-licensed product candidates are not successfully commercialized by our collaboration partners, we will face difficulty in funding our internal pipeline of product candidates and in generally obtaining product revenue in future periods, which could result in significant harm to our financial position and adversely affect the price of the ADSs or our ordinary shares.
The proposed acquisition of Valeritas may not be completed on the anticipated terms or at all.
On February 10, 2020, we announced a bid to acquire substantially all assets from Valeritas for a total cash consideration of $23 million and the assumption of certain liabilities related to the ongoing business (including up to approximately $13.3 million related to open purchase orders, license payments and cure costs relating to prepetition contracts that will be assumed by Valeritas under the U.S. Bankruptcy Code upon exiting Chapter 11 proceedings), pursuant to the terms of the "stalking horse" asset purchase agreement entered into with Valeritas. The proposed sale is to be conducted through a court-supervised sale process under Section 363 of the U.S. Bankruptcy Code and will be subject to court-approved bidding procedures and receipt of competing offers at auction. A hearing was held before the Bankruptcy Court on March 6, 2020, during which the Bankruptcy Court determined the procedures for the bidding and potential auction of Valeritas, which is expected to occur in March 2020. If our bid is selected, the sale will be subject to approval by the U.S. Bankruptcy Court and certain other closing conditions, including a condition relating to manufacturing performance to address supply disruptions experienced by Valeritas in December 2019. There can be no certainty that the transaction will be concluded on any particular time frame or at all. We may be required to increase our bid if other bidders arise to contest the bid. The court overseeing the bidding process may also require changes in the terms of the asset purchase agreement. If we are required to increase our bid or otherwise change the scope of the acquisition, it could be detrimental to the Company following any consummation of the asset purchase, including with respect to the synergies that we may expect to achieve through the acquisition of Valeritas, if any.
We may be unable to successfully integrate Valeritas into our organization.
If our proposed acquisition of Valeritas is completed, the successful integration of its operations into ours and our ability to realize synergies and benefits therefrom are subject to a number of risks and uncertainties, many of which are outside of our control, including:
- •
- the challenge of integrating complex organizations, systems, operating procedures, compliance programs, technology, networks and other assets
of Valeritas;
- •
- the challenge of integrating Valeritas' sales force into our organization;
- •
- the difficulties harmonizing differences in the business cultures of our company and Valeritas;
11
- •
- the inability to combine successfully our respective businesses in a manner that permits us to achieve the synergies and other benefits from
the acquisition of Valeritas;
- •
- the inability to minimize the diversion of management attention from ongoing business concerns during the process of integrating Valeritas into
our businesses; and
- •
- the challenge of managing the expanded operations of a significantly larger and more complex company and coordinating geographically separate organizations.
We will be required to devote significant management attention and resources to integrating business practices, cultures and operations of the two businesses. We will incur substantial expenses to consummate the proposed acquisition of Valeritas and may not realize the synergies or other benefits on the timeline expected or at all. Even if we are able to integrate Valeritas successfully into our business, any anticipated benefits of the acquisition of Valeritas may not be realized fully, or at all, and may take longer than anticipated to realize than expected.
We expect our operating loss to increase in the near-term if the acquisition of Valeritas is consummated. Valeritas may also have liabilities that are not known to us.
Valeritas reported revenue of $22.4 million and loss before income taxes of $41.9 million for the nine months ended September 30, 2019. Valeritas reported in December 2019 that it experienced certain manufacturing performance issues that resulted in supply disruptions and are expected to have a material negative impact on its revenue and loss before income taxes. Although we expect that, if the Valeritas transaction is consummated, we may be able to reduce costs in the Valeritas business associated with some aspects of how the business is run (for instance, rationalizing some of the sites and removing duplicative costs like insurance policies) relative to historical amounts, there can be no such assurance that any such cost reductions will be realized on a timely basis or at all. Even if such cost reductions are realized, we expect that the Valeritas acquisition will materially increase our operating loss and cash used in operations in the near term.
Valeritas may also have liabilities that we failed, or were unable, to discover in the course of performing our due diligence investigations of Valeritas or that are larger than we currently expect based on those diligence investigations. We may learn additional information about Valeritas that materially adversely affects us, such as unknown or contingent liabilities and liabilities related to compliance with applicable laws. Any such liabilities, individually or in the aggregate, could have a material adverse effect on our business, financial condition and results of operations.
Risks Related to Our Products and Product Candidates
We are dependent on the clinical success of our internal product candidates, including glepaglutide and dasiglucagon.
We are dependent on our ability to develop successfully, obtain regulatory approval for, and then successfully commercialize our other internal product candidates, including glepaglutide and dasiglucagon. Our internal product candidates will require additional R&D clinical development, management of clinical and manufacturing activities, regulatory approval in multiple jurisdictions (if regulatory approval can be obtained at all), securing sources of commercial manufacturing supply, building of, or partnering with, a commercial organization, substantial investment and significant marketing efforts before any revenue can be generated from product sales. We are not permitted to market or promote any of our product candidates in any jurisdiction before we receive regulatory approval from the FDA, the EMA or any other comparable regulatory authority in that jurisdiction, and we may never receive such regulatory approval for any of our product candidates in any particular jurisdiction or at all. We cannot assure you that our clinical trials for glepaglutide or dasiglucagon will be completed in a timely manner, or at all, or that we will be able to obtain approval from the FDA,
12
EMA or any other comparable regulatory authority for any of our product candidates. We cannot be certain that we will advance any other product candidates that are part of our early non-clinical pipeline into clinical trials. If any of our advanced product candidates such as glepaglutide and dasiglucagon or any future product candidate is not approved and commercialized in any particular jurisdiction, we may not be able to generate any royalties or product revenue, as the case may be, for that product candidate at all or in such jurisdiction. Moreover, any delay or setback in the development of any product candidate could materially adversely affect our business and cause the price of the ADSs or our ordinary shares to fall.
Our product candidates will need to undergo clinical trials that are time consuming and expensive, the outcomes of which are unpredictable, and for which there is a high risk of failure. If clinical trials of our product candidates fail to satisfactorily demonstrate safety and efficacy to the FDA, the EMA and any other comparable regulatory authority, we may incur additional costs or experience delays in completing, or ultimately be unable to complete, the development of these product candidates.
The FDA in the United States, the EMA in Europe, and any other comparable regulatory authorities in other jurisdictions must approve new product candidates before they can be marketed, promoted or sold in those territories. We must provide these regulatory authorities with data from preclinical studies and clinical trials that demonstrate that our product candidates are safe and effective for a specific indication before they can be approved for commercial distribution. We cannot be certain that our clinical trials for our product candidates will be successful or that any of our other internal or out-licensed product candidates will receive approval from the FDA, the EMA or any other comparable regulatory authority.
Preclinical studies and clinical trials are long, expensive and unpredictable processes that can be subject to extensive delays. We cannot guarantee that any clinical trials will be conducted as planned or completed on schedule, if at all. It may take several years and require significant expenditures to complete the preclinical studies and clinical trials necessary to commercialize a product candidate, and delays or failure are inherently unpredictable and can occur at any stage. Interim results of clinical trials do not necessarily predict final results, and success in preclinical studies and early clinical trials does not ensure that later clinical trials will be successful. A number of companies in the pharmaceutical, biopharmaceutical and biotechnology industries have suffered significant setbacks in advanced clinical trials even after promising results in earlier trials, and we cannot be certain that we will not face similar setbacks. The design of a clinical trial can determine whether its results will support approval of a product, and flaws in the design of a clinical trial may not become apparent until the clinical trial is well advanced or completed. Changing the design of a clinical trial can be expensive and time consuming. An unfavorable outcome in one or more trials would be a major setback for our product candidates and for us. An unfavorable outcome in one or more trials may require us to delay, reduce the scope of or eliminate one or more product development programs, which could have a material adverse effect on our business, financial position, results of operations and future growth prospects.
In connection with clinical trials of our product candidates, we face a number of risks, including risks that:
- •
- a product candidate is ineffective, inferior to existing approved products for the same indications, unacceptably toxic or has unacceptable
side effects;
- •
- patients may die or suffer other adverse effects for reasons that may or may not be related to the product candidate being tested;
- •
- extension studies on long-term tolerance could invalidate the use of our product;
- •
- the results may not confirm the positive results of earlier trials;
13
- •
- the results may not meet the level of statistical significance required by the FDA, the EMA or other relevant regulatory agencies to establish
the safety and efficacy of our product candidates for continued trial or marketing approval; and
- •
- our collaboration partners or contract research organizations, or CROs, are unable or unwilling to perform under their contracts.
The results of preclinical studies do not necessarily predict clinical success, and larger and later-stage clinical trials may not produce the same results as earlier-stage clinical trials. Our and our collaboration partners' clinical trials of our product candidates conducted to date have generated favorable safety and efficacy data. However, we may have different enrollment criteria in our future clinical trials. As a result, we may not observe a similarly favorable safety or efficacy profile as in our prior clinical trials. In addition, we cannot assure you that during the course of potential widespread use of any of our product candidates in future, we will not suffer setbacks in maintaining production quality or stability. In addition, clinical trials of potential products often reveal that it is not possible or practical to continue development efforts for these product candidates. If we do not successfully complete preclinical and clinical development, we will be unable to market and sell our product candidates and generate additional revenue. Even if we successfully complete clinical trials, those results are not necessarily predictive of results of additional trials that may be needed before marketing applications may be submitted to the FDA, the EMA or other regulatory authority, as applicable.
Two of our late stage clinical programs have been designated as orphan products. In the case of glepaglutide, its principle indication is intended for the treatment of short bowel syndrome and for dasiglucagon for the treatment of chronic hyperinsulinism, or CHI. Products that developed are for the treatment of orphan diseases receive additional assistance, faster processing and reduced application fees from regulatory authorities like the FDA, EMA or comparable authorities in other countries. In addition to these benefits, orphan indications also receive additional exclusivity protections that are not awarded to non-orphan drugs and enable orphan drugs to be awarded additional market exclusivity. If we are unable to obtain orphan exclusivity protection for glepaglutide or for dasiglucagon or our other products, we may be unable to prevent other companies from producing very similar products in the same therapeutic area or even competition from generic producers.
Furthermore, we sometimes estimate for planning purposes the timing of the accomplishment of various scientific, clinical, regulatory and other product development objectives. These milestones may include our expectations regarding the commencement or completion of scientific studies, clinical trials, the submission of regulatory filings or commercialization objectives. From time to time, we may publicly announce the expected timing of some of these milestones, such as the completion of an ongoing clinical trial, the initiation of other clinical programs, receipt of marketing approval or a commercial launch of a product. The achievement of many of these milestones may be outside of our control. All of these milestones are based on a variety of assumptions, which may cause the timing of achievement of the milestones to vary considerably from our estimates. If we fail to achieve announced milestones in the timeframes we expect, the commercialization of our product candidates may be delayed, we may not be entitled to receive certain contractual payments, which could have a material adverse effect on our business, financial position, results of operations and future growth prospects.
The COVID-19 coronavirus could materially adversely impact our business, including our clinical trials.
In December 2019, a novel strain of coronavirus, COVID-19, was reported to have surfaced in Wuhan, China. Since then, the COVID-19 coronavirus has spread to multiple countries, including the United States and several European countries, including countries in which we have planned or ongoing clinical trials. If the COVID-19 coronavirus continues to spread in the United States, we may experience disruptions that could severely impact our business and clinical trials, including:
- •
- delays or difficulties in enrolling patients in our clinical trials;
14
- •
- delays or difficulties in clinical site initiation, including difficulties in recruiting clinical site investigators and clinical site staff;
- •
- diversion of healthcare resources away from the conduct of clinical trials, including the diversion of hospitals serving as our clinical trial
sites and hospital staff supporting the conduct of our clinical trials;
- •
- interruption of key clinical trial activities, such as clinical trial site monitoring, due to limitations on travel imposed or recommended by
federal or state governments, employers and others; and
- •
- limitations in employee resources that would otherwise be focused on the conduct of our clinical trials, including because of sickness of employees or their families or the desire of employees to avoid contact with large groups of people.
For our clinical trials that are being conducted at sites outside the United States, particularly in countries which are experiencing heightened impact from the COVID-19 coronavirus, in addition to the risks listed above, we may also experience the following adverse impacts:
- •
- delays in receiving approval from local regulatory authorities to initiate our planned clinical trials;
- •
- delays in clinical sites receiving the supplies and materials needed to conduct our clinical trials;
- •
- interruption in global shipping that may affect the transport of clinical trial materials, such as comparator drugs used in certain of our
clinical trials;
- •
- changes in local regulations as part of a response to the COVID-19 coronavirus outbreak which may require us to change the ways in which our
clinical trials are conducted, which may result in unexpected costs, or to discontinue the clinical trials altogether;
- •
- delays in necessary interactions with local regulators, ethics committees and other important agencies and contractors due to limitations in
employee resources or forced furlough of government employees; and
- •
- refusal of the FDA to accept data from clinical trials in these affected geographies.
The global outbreak of the COVID-19 coronavirus continues to rapidly evolve. The extent to which the COVID-19 coronavirus may impact our business and clinical trials will depend on future developments, which are highly uncertain and cannot be predicted with confidence, such as the ultimate geographic spread of the disease, the duration of the outbreak, travel restrictions and social distancing in the United States and other countries, business closures or business disruptions and the effectiveness of actions taken in the United States and other countries to contain and treat the disease.
We selectively rely on third parties to conduct our clinical trials and perform data collection and analysis, which may result in costs and delays that prevent us from successfully commercializing our product candidates.
We currently, and expect to continue to, selectively rely on public and private research institutions, medical institutions, clinical investigators, CROs, contract laboratories and collaboration partners to conduct some of our early-stage product development activities, perform data collection and analysis and carry out our clinical trials. Our development activities or clinical trials conducted in reliance on third parties may be delayed, suspended or terminated if:
- •
- the third parties do not devote a sufficient amount of time or effort to our activities or otherwise fail to successfully carry out their
contractual duties or to meet regulatory obligations or expected deadlines;
- •
- we replace a third party; or
15
- •
- the quality or accuracy of the data obtained by third parties is compromised due to their failure to adhere to clinical protocols, regulatory requirements or for other reasons.
We do not have the ability to control the performance of third parties in their conduct of development activities. Third party performance failures may increase our development costs, delay our ability to obtain regulatory approval and delay or prevent the commercialization of our product candidates. While we believe that there are alternative sources to provide these services, in the event that we seek such alternative sources, we may not be able to enter into replacement arrangements without incurring delays or additional costs.
We rely on third parties to manufacture our preclinical and clinical drug supplies and we intend to rely on third parties to produce commercial supplies of any approved product candidate.
If, for any reason, we were to experience an unexpected loss of supply of our product candidates or placebo or comparator drug used in certain of our clinical trials, whether as a result of manufacturing, supply or storage issues or otherwise, we could experience delays, disruptions, suspensions or terminations of, or be required to restart or repeat, any pending or ongoing clinical trials. We do not currently have, nor do we plan to acquire, the infrastructure or capability internally to manufacture our preclinical and clinical drug supplies and we lack the resources and the capability to manufacture any of our product candidates on a clinical or commercial scale. The facilities used by our contract manufacturers or other third party manufacturers to manufacture our product candidates are subject to the FDA's, the EMA's and other comparable regulatory authorities' preapproval inspections that will be conducted after we submit our NDA to the FDA or the required approval documents to any other relevant regulatory authority. We do not control the implementation of the manufacturing process of, and are completely dependent on, our contract manufacturers or other third party manufacturers for compliance with the regulatory requirements, known as current good manufacturing practices, or cGMPs, for manufacture of both active drug substances and finished drug products. If our contract manufacturers or other third party manufacturers cannot successfully manufacture material that conforms to applicable specifications and the strict regulatory requirements of the FDA, the EMA or other comparable regulatory authority, we will not be able to secure and/or maintain regulatory approvals for our products manufactured at these facilities. In addition, we have no control over the ability of our contract manufacturers or other third party manufacturers to maintain adequate quality control and quality assurance procedures and qualified personnel. If the FDA, the EMA or another comparable regulatory authority finds deficiencies at these facilities for the manufacture of our product candidates or if it withdraws any approval because of deficiencies at these facilities in the future, we may need to find alternative manufacturing facilities, which would significantly impact our ability to develop, obtain regulatory approval for or market our product candidates, if approved.
We rely on our manufacturers to purchase from third party suppliers the materials necessary to produce our product candidates for our clinical trials. There are a limited number of suppliers for raw materials that we use to manufacture our drugs and there may be a need to assess alternate suppliers to prevent a possible disruption of the manufacture of the materials necessary to produce our product candidates for our clinical trials, and if approved, for commercial sale. We do not have any control over the process or timing of the acquisition of these raw materials by our manufacturers. Moreover, we currently do not have any agreements in place for the commercial production of these raw materials. Although we generally do not begin a clinical trial unless we believe we have access to a sufficient supply of a product candidate to complete the clinical trial, any significant delay in the supply of a product candidate, or the raw material components thereof, for an ongoing clinical trial due to the need to replace a contract manufacturer or other third party manufacturer could considerably delay completion of our clinical trials, product testing and potential regulatory approval of our product candidates. If our manufacturers or we are unable to purchase these raw materials after regulatory approval has been obtained for our product candidates, the commercial launch of our product
16
candidates would be delayed or there would be a shortage in supply, which would impair our ability to generate revenue from the sale of our product candidates. Additionally, if we receive regulatory approval for our product candidates, we may experience unforeseen difficulties or challenges in the manufacture of our product candidates on a commercial scale compared to the manufacture for clinical purposes.
We face substantial competition from companies with considerably more resources and experience than we have, which may result in others discovering, developing, receiving approval for or commercializing products before or more successfully than us.
The pharmaceutical and biotechnology industries are characterized by intense competition and significant and rapid technological change as researchers learn more about diseases and develop new technologies and treatments. Any product candidates that we successfully develop and commercialize will compete with existing drugs and new drugs that may become available in the future. We have competitors in each of the disease fields in which we compete, many of which have substantially greater name recognition, commercial infrastructure and financial, technical and personnel resources than we have. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with larger and established companies. Significant competitive factors in our industry include product efficacy and safety, quality and breadth of an organization's technology, skill of an organization's employees and its ability to recruit and retain key employees, timing and scope of regulatory approvals, government reimbursement rates for, and the average selling price of, products, the availability of raw materials and qualified manufacturing capacity, manufacturing costs, intellectual property and patent rights and their protection and sales and marketing capabilities. While we believe that our product and product candidate platform, development expertise and scientific knowledge provide us with competitive advantages, we face potential competition from many different sources, including major pharmaceutical, specialty pharmaceutical and biotechnology companies, academic institutions and governmental agencies and public and private research institutions.. There can be no assurance that our competitors will not deploy their superior resources to damage our and our drug candidates' prospects. Given the intense competition in our industry, we cannot assure you that any of the products that we successfully develop will be clinically superior or scientifically preferable to products developed or introduced by our competitors.
In addition, significant delays in the development of our product candidates could allow our competitors to succeed in obtaining the FDA, the EMA or other regulatory approvals for their product candidates more rapidly than us, which could place us at a significant competitive disadvantage or deny us marketing exclusivity rights.
Competitors may develop novel products or other technologies that could make our product candidates obsolete or uneconomical. Any of our product candidates that competes with an approved product may need to demonstrate compelling advantages, such as increased efficacy, convenience, pricing, tolerability and/or safety in order to be commercially successful. As a result, the pricing of certain of our products and product candidates, if and when approved for marketing, will depend, in part, on the pricing strategies adopted by our competitors. Any of our product candidates that are approved could also face other competitive factors in the future, including biosimilar competition, which could force us to lower prices or could result in reduced sales. Any failure to compete effectively against our current and future competitors could have a material adverse effect on our business, financial position, results of operations and future growth prospects.
In addition, many of our competitors have significantly greater financial resources and expertise in R&D, manufacturing, conducting preclinical studies and clinical trials, obtaining regulatory approvals and marketing drugs. Mergers and acquisitions in the pharmaceutical and biotechnology industries may result in even more resources being concentrated among a smaller number of competitors, particularly through partnership arrangements with large established companies. These companies also compete
17
with us in recruiting and retaining qualified scientific and management personnel and establishing clinical trial sites and patient registration for clinical trials, as well as in acquiring technologies complementary to, or necessary for, our programs.
Certain of our peptide product candidates are expected to be delivered parenterally by medical devices that may be regulated as combination products that are required to obtain separate FDA clearance or pre-market approval and/or approval by other regulatory authorities.
Certain of our peptide product candidates are intended to be used in combination with a delivery device, such as an injector or other delivery system. Medical products containing a combination of new drugs, biological products or medical devices may be regulated as "combination products" in the United States and Europe. A combination product generally is defined as a product comprised of components from two or more regulatory categories (such as drug/device, device/biologic, drug/biologic). Each component of a combination product is subject to the requirements established by the FDA for that type of component, whether a new drug, biologic or device. In order to facilitate pre-market review of combination products, the FDA designates one of its centers to have primary jurisdiction for the pre-market review and regulation of the overall product based upon a determination by the FDA of the primary mode of action of the combination product. The determination whether a product is a combination product or two separate products is made by the FDA on a case-by-case basis. Our product candidates intended for use with such devices, or expanded indications that we may seek for our products used with such devices, may not be approved or may be substantially delayed in receiving approval if the devices do not gain and/or maintain their own regulatory approvals or clearances.
Where approval of the drug or biologic product and device is sought under a single application, the increased complexity of the review process may delay approval. The FDA review process and criteria is not a well-established area, which could also lead to delays in the approval process. The EMA has a parallel review process in place for combination products, the potential effects of which in terms of approval and timing could independently affect our ability to market our combination products in Europe. In addition, because these delivery devices are provided by unaffiliated third party companies, we are dependent on the sustained cooperation and effort of those third party companies both to obtain regulatory approval and to maintain their own regulatory compliance. Failure of third party companies to assist in the approval process or to maintain their own regulatory compliance could delay or prevent approval of our product candidates, or limit our ability to sell a product once approved.
We currently have no sales function. If we are unable to establish a sales function or enter into sales, marketing and distribution arrangements with third parties, we may not be successful in commercializing our internal product candidates if and when they are approved.
We do not have a sales or marketing infrastructure and have no experience in the sale, marketing or distribution of pharmaceutical products. To achieve commercial success for any internal product candidate for which we obtain marketing approval, we will need to establish a sales and marketing function or make arrangements with third parties to perform sales and marketing functions on our behalf, and we may not be successful in doing so.
If we enter into arrangements with third parties to perform sales, marketing and distribution services on our behalf, our product revenue or the profitability of our drug revenue may be lower, perhaps substantially lower, than if we were to directly market and sell our drugs. Furthermore, we may be unsuccessful in entering into the necessary arrangements with third parties or may be unable to do so on terms that are favorable to us.
18
Even if we are able to enter into acceptable partnerships, we may have little or no control over such third parties, and our future collaboration partners may fail to devote the necessary resources and attention to sell and market our drugs effectively. Budgeting restrictions or strategy changes of our future collaboration partners could delay or prevent successful clinical development or marketing efforts. Similarly, our future collaboration partners could decide to give priority to the clinical development or marketing of product candidates or develop or seek to develop product candidates in competition with our product candidates.
Our failure to establish and maintain successful partnerships could have a material adverse effect on our business, financial position, results of operations and future growth prospects.
Risks Related to Our Operations
There is a risk that our products may have major side effects that may give rise to substantial liability claims.
As a biopharmaceutical company, we operate in a market that is subject to risk of liability. To our knowledge, we are not currently subject to any product liability suits. However, we may be subject to future liability claims alleging adverse effects from the use of our products in clinical trials or medical practice. Any liability claims could have a material adverse effect on our business, financial position, results of operations and future growth prospects.
There is a risk that we may not be able to maintain insurance coverage, and that existing or any future insurance policies or our own resources will not sufficiently cover claims for damages that we may receive in the future.
Our business exposes us to potential product liability and other liability risks that are inherent in clinical development, manufacturing, marketing and use of human therapeutic products. It is generally necessary for us to secure certain levels of insurance as a condition for the conduct of clinical trials and any sale or use of our products. We have taken out product liability insurance with respect to all clinical trials and ongoing trials performed to date for which we were responsible (i.e., in respect of our internal product pipeline).
We may seek to expand our insurance coverage if we obtain marketing approval for any of our internal product candidates or if other risks related to our business increase. We may not be able to obtain or maintain adequate protection against potential liabilities at a cost that is acceptable to us. If we are unable to obtain insurance or other protection against potential product liability claims, we could be exposed to significant liabilities, which may materially and adversely affect our business and financial position. These liabilities could prevent or interfere with our product development and commercialization efforts. If we are sued for any injury caused by our products or processes, our liability could exceed our product liability insurance coverage and our own financial resources and, consequently, could have a material adverse effect on our business, financial position, results of operations and future growth prospects.
Our future success depends on our ability to retain our management team and key employees.
We are highly dependent on the management, development, clinical, financial and business development expertise of our management team and key employees. Recruiting and retaining qualified scientific and clinical personnel will also be critical to our future success. The loss of the services of any of the members of our management team or key employees could impede the achievement of our development and commercialization objectives and seriously harm our ability to successfully implement our business strategy. Furthermore, replacing any of the members of our management team or key employees may be difficult and may take an extended period of time because of the limited number of individuals in our industry with the breadth of skills and experience required to successfully develop, gain regulatory approval for and commercialize drugs. Competition to hire from this limited pool is
19
intense, and we may be unable to hire, train, retain or motivate the members of our management team or key employees on acceptable terms given the competition among numerous pharmaceutical, biopharmaceutical and biotechnology companies for similar personnel. We also experience competition for the hiring of scientific and clinical personnel from universities and research institutions. If we are unable to continue to attract and retain high quality management and employees, our ability to pursue our growth strategy will be limited.
Our R&D activities could be affected or delayed as a result of possible restrictions on animal testing.
Certain laws and regulations require us to test our product candidates on animals before initiating clinical trials involving humans. Animal testing activities have been the subject of controversy and adverse publicity. Animal rights groups and other organizations and individuals have attempted to stop animal testing activities by pressing for legislation and regulation in these areas and by disrupting these activities through protests and other means. To the extent the activities of these groups are successful, our R&D activities may be interrupted, delayed or become more expensive.
If we fail to manage our growth effectively, our ability to develop and commercialize products could suffer.
We expect that if our drug discovery efforts continue to generate product candidates, our clinical product candidates continue to progress in development, and we continue to build our development and commercial organizations, we will require significant additional investment in personnel, management and resources. Our ability to achieve our research, development and commercialization objectives depends on our ability to respond effectively to these demands and expand our internal organization, systems, controls and facilities to accommodate additional anticipated growth. If we are unable to manage our growth effectively, our business could be harmed and our ability to execute our business strategy could suffer.
Our internal computer systems, or those of our collaboration partners or other contractors or consultants, may fail or suffer security breaches, which could result in a material disruption of our product development programs.
Our internal computer systems and those of our current and any future collaboration partners and other contractors or consultants are vulnerable to damage from cybersecurity breaches, computer viruses, unauthorized access, natural disasters, terrorism, war and telecommunication and electrical failures. While we do not believe that we have experienced any such material system failure, accident or security breach to date, if such an event were to occur and cause interruptions in our operations, it could result in a material disruption of our development programs and our business operations, whether due to a loss of our trade secrets or other proprietary information or other similar disruptions. For example, the loss of clinical trial data for our product candidates from completed or future clinical trials could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the data. To the extent that any disruption or security breach were to result in a loss of, or damage to, our data or applications or other data or applications relating to our technology or product candidates, or inappropriate disclosure of confidential or proprietary information, we could incur liabilities, our competitive position could be harmed and the further development and commercialization of our product candidates could be delayed.
Our operations as a global company subject us to various risks, and our failure to manage these risks could adversely affect our results of operations.
We face significant operational risks as a result of doing business internationally, such as:
- •
- fluctuations in foreign currency exchange rates (in particular, U.S. dollars, Euros and Danish kroner);
20
- •
- potentially adverse and/or unexpected tax consequences, including penalties due to the failure of tax planning or due to the challenge by tax
authorities on the basis of transfer pricing and liabilities imposed from inconsistent enforcement;
- •
- potential changes to the accounting standards, which may influence our financial situation and results;
- •
- becoming subject to the different, complex and changing laws, regulations and court systems of multiple jurisdictions and compliance with a
wide variety of foreign laws, treaties and regulations;
- •
- reduced protection of, or significant difficulties in enforcing, intellectual property rights in certain countries;
- •
- restrictions imposed by local labor practices and laws on our business and operations, including unilateral cancellation or modification of
contracts;
- •
- rapid changes in global government, economic and political policies and conditions, political or civil unrest or instability, terrorism,
epidemics or pandemics (such as the COVID-19 coronavirus), and other similar outbreaks or events, and potential failures in supply or demand for our products due to such changes or events or the fear
that they may occur; and
- •
- tariffs, trade protection measures, import or export licensing requirements, trade embargoes and other trade barriers.
Additionally, as a global company, we are subject to the Foreign Corrupt Practices Act, or the FCPA, which generally prohibits companies and their intermediaries from making or offering improper payments to non-U.S. officials for the purpose of obtaining or retaining business. The FCPA generally also requires companies listed on a U.S. stock exchange to maintain a system of adequate internal accounting controls and to make and keep books, records and accounts that accurately and fairly reflect transactions and dispositions of assets. Because of the predominance of government-sponsored health care systems around the world, many of our commercial relationships outside of the United States are with governmental entities, and personnel of such entities may be considered non-U.S. officials for purposes of the FCPA. Violations of the FCPA and other applicable anti-bribery laws are punishable by criminal fines and imprisonment, civil penalties, disgorgement of profits, injunctions, debarment from government contracts as well as other remedial measures. We have adopted a written code of business conduct and other policies and procedures to assist us and our personnel in complying with the FCPA and other applicable anti-bribery laws prior to completion of the offering. However, our personnel and others acting on our behalf could take actions that violate these requirements.
If we were to experience any of the foregoing events, it could adversely affect our reputation, business, financial condition and results of operations.
We have identified material weaknesses in our internal control over financial reporting. If we fail to maintain an effective system of internal control over financial reporting, we may not be able to accurately report our financial results in a timely manner or prevent fraud, which may adversely affect our business, investor confidence in our company and the market price of our shares and ADSs.
Our management concluded that we had not maintained effective internal control over financial reporting and disclosure controls and procedures as of December 31, 2019, 2018, 2017, 2016 and 2015 due to the material weaknesses we identified in the design and operating effectiveness of our internal controls over financial reporting, including lack of sufficient competencies related to IFRS and SEC reporting knowledge for the purposes of timely and reliable financial reporting in connection with our financial statement preparation process for the years ended December 31, 2019, 2018, 2017, 2016 and 2015. Under the standards established by the U.S. Public Company Accounting Oversight Board, or the
21
PCAOB, a material weakness is a deficiency, or a combination of deficiencies, that creates a reasonable possibility that a material misstatement of a company's annual financial statements will not be prevented or detected on a timely basis. The material weaknesses identified by us related to our processes to assess risk and to design and implement effective control activities over financial reporting. Previously we did not have formalized risk assessment, oversight and compliance processes or formalized control descriptions for all of our key controls. During 2018 and 2019 these areas were developed and implemented, and we are working in 2020 to properly integrate them further into our routines and practices in the organization. Where control descriptions existed, they need to be further developed to ensure that they include all relevant information to enable the operating effectiveness of such controls. We will also work to ensure that adequate controls are performed in all areas. Where control activities were dependent on certain information, which is referred to as our IUC, we did not perform or document controls to assess the completeness and accuracy of such information. Internal capabilities have not been sufficient around the special and unusual transactions where we are relying on external assistance for proper handling of related risks. We have not adequately monitored control activities and identified control deficiencies; thus, we have been unable to evaluate whether other deficiencies, individually or in combination, resulted in a reasonable possibility that a material misstatement of our annual financial statements would not be prevented or detected on a timely basis. Further, restatements resulting from the correction of certain misstatements in our quarterly and annual financial statements have been identified, including in the first quarter of 2019 related to the recognition of warrants. These restatements occurred due to the lack of sufficient overall review of the financial statements and lack of oversight of the application and implementation of accounting policies and accounting standards by the Company.
In addition to engaging external subject matter experts in key areas to improve our competencies with respect to IFRS and SEC reporting, in late 2018 and early 2019 we implemented a formalized process for risk assessment, completed the design and documentation of our key controls to respond to the identified risks, and initiated implementation of a formalized process for performing our evaluation of the effectiveness of our internal controls over financial reporting. We hired a new Chief Executive Officer in April 2019 and a new Chief Financial Officer in October 2019. In 2019, we also continued to focus on our accounting and reporting functions by replacing and adding more capable resources and working towards improving competencies and skills. In 2020, we expect to focus on utilizing our information technology infrastructure to drive efficiency and effectiveness in processes and internal controls through automation and standardization, while we continue to engage with external subject matter experts in key areas to improve our competencies with respect to IFRS and SEC reporting. We have hired, and plan to continue to hire, additional finance and accounting personnel with appropriate expertise to perform specific functions and to further assist in the implementation of improved processes and internal controls. We expect to continue to build, develop and invest in our financial management and reporting infrastructure and further evolve and document our accounting policies and financial reporting procedures, including ongoing senior management review and audit committee oversight.
While our management believes that the steps that we have taken and plans we continue to make will improve our overall system of internal control over financial reporting and will remediate identified material weaknesses, the material weaknesses cannot be considered remediated until the applicable relevant controls operate for a sufficient period of time and management has concluded, through testing, that these controls are operating effectively. No assurance can be provided at this time that the actions and remediation efforts will effectively remediate the material weaknesses described above or prevent the incidence of other material weaknesses in the Company's internal control over financial reporting in the future. Further, while our goal is to remediate the material weaknesses in 2020, we do not know the specific time frame needed to fully remediate the material weaknesses identified.
22
Additionally, even if the material weakness in the design and operating effectiveness of our internal controls over financial reporting that had been identified was remediated by our actions, we may discover future deficiencies or material weaknesses in our internal controls over financial reporting, including those identified through testing conducted by us pursuant to Section 404(a) of the Sarbanes-Oxley Act or subsequent testing by our independent registered public accounting firm when required pursuant to the Sarbanes-Oxley Act. Such deficiencies may be deemed to be significant deficiencies or material weaknesses and may require changes to our consolidated financial statements or identify other areas for further attention or improvement. Even if we are able to report our financial statements accurately and in a timely manner, if we do not make all necessary improvements to address any outstanding material weaknesses, continued disclosure of such significant deficiencies and material weaknesses may be required in future filings with the SEC, which may adversely affect our business, investor confidence in our company and the market price of our shares and ADSs.
The United Kingdom's withdrawal from the EU could result in increased regulatory and legal complexity, which may make it more difficult for us to do business in the EU and the rest of Europe and impose additional challenges in securing regulatory approval of our product candidates in the EU and the rest of Europe.
The United Kingdom is a major market for pharmaceutical products. Following the result of a referendum in 2016, the United Kingdom left the EU on January 31, 2020, commonly referred to as Brexit. Pursuant to the formal withdrawal arrangements agreed between the United Kingdom and the EU, the United Kingdom will be subject to a transition period until December 31, 2020, or the Transition Period, during which EU rules will continue to apply. Negotiations between the United Kingdom and the EU are expected to continue in relation to the customs and trading relationship between the United Kingdom and the EU following the expiry of the Transition Period.
In addition, as a result of Brexit, the EMA, formerly situated in London, relocated to Amsterdam. Following the Transition Period, there is a risk that the relocation will interrupt current administrative routines and occupy resources, which may generally adversely affect our dealings with the EMA. Further, there is considerable uncertainty resulting from a lack of precedent and the complexity of the United Kingdom and EU's intertwined legal regimes as to how Brexit (following the Transition Period) will impact the life sciences industry in Europe, including our company, including with respect to ongoing or future clinical trials.
The impact will largely depend on the model and means by which the United Kingdom's relationship with the EU is governed post-Brexit. For example, following the Transition Period, the United Kingdom will no longer be covered by the centralized procedures for obtaining EU-wide marketing authorization from the EMA and, unless a specific agreement is entered into, a separate process for authorization of drug products, including our product candidates, will be required in the United Kingdom, the potential process for which is currently unclear. As a result, we cannot predict the impact that Brexit will have on (i) the marketing of pharmaceutical products, (ii) the process to obtain regulatory approval in the United Kingdom for product candidates or (iii) the award of exclusivities that are normally part of the EU legal framework (for instance Supplementary Protection Certificates, Pediatric Extensions or Orphan exclusivity). Brexit may adversely affect and delay our ability to commercialize, market and sell our product candidates in the United Kingdom.
Brexit may also result in a reduction of funding to the EMA if the United Kingdom no longer makes financial contributions to European institutions, such as the EMA. If UK funding is so reduced, it could create delays in the EMA issuing regulatory approvals for our product candidates and, accordingly, have a material adverse effect on our business, financial condition, results of operations or prospects.
23
As a result of Brexit, other European countries may seek to conduct referenda with respect to their continuing membership with the EU. Given these possibilities and others we may not anticipate, as well as the absence of comparable precedent, it is unclear what financial, regulatory and legal implications the withdrawal of the United Kingdom from the EU would have and how such withdrawal would affect us, and the full extent to which our business could be adversely affected.
Risks Related to Our Intellectual Property
Our ability to compete may decline if we or our collaboration partners are unable to or do not adequately protect intellectual property rights or if our intellectual property rights are inadequate for our product candidates or future product candidates
Our commercial success and viability depends on our and our collaboration partners' ability to obtain and maintain patent protection in the United States, Europe and other countries with respect to our existing product candidates owned by us and to successfully defend these rights against third party challenges, as well as our ability to maintain adequate intellectual property protection for any future products. If we or our collaboration partners do not adequately protect our intellectual property, competitors may be able to use our technologies and erode or negate any competitive advantage we may have, which could materially harm our business, negatively affect our position in the marketplace, limit our ability to commercialize our product candidates and delay or render impossible our achievement of profitability.
Our strategy and future prospects are based, in particular, on our patent portfolio. We and our collaboration partners or licensees will best be able to protect our product candidates and their uses from unauthorized use by third parties to the extent that valid and enforceable patents, effectively protected trade secrets, or other regulatory exclusivities, cover them. Also, intellectual property rights have limitations and do not necessarily address all potential threats to our competitive advantage. Our ability to obtain patent protection for our product candidates is uncertain and the degree of future protection afforded by our intellectual property rights is uncertain due to a number of factors, including, but not limited to:
- •
- we or our collaboration partners may not have been the first to make the inventions covered by pending patent applications or issued patents;
- •
- we or our collaboration partners may not have been the first to file patent applications for our product candidates or the compositions we
developed or for their uses;
- •
- others may independently develop identical, similar or alternative products or compositions and uses thereof;
- •
- our or our collaboration partners' disclosures in patent applications may not be sufficient to meet the statutory requirements for
patentability;
- •
- any or all of our or our collaboration partners' pending patent applications may not result in issued patents;
- •
- we or our collaboration partners may not seek or obtain patent protection in countries that may eventually provide us with a significant
business opportunity;
- •
- any patents issued to us or our collaboration partners may not provide a basis for commercially viable products, may not provide any
competitive advantages, or may be successfully challenged by third parties;
- •
- our or our collaboration partners' compositions and methods may not be patentable;
- •
- others may design around our or our collaboration partners' patent claims to produce competitive products or uses which fall outside of the scope of our patents;
24
- •
- others may identify prior art or other bases which could result in the prohibition or limitation of our or our collaboration partners' patents;
- •
- our competitors might conduct R&D activities in the United States and other countries that provide a safe harbor from patent infringement
claims for certain R&D activities, as well as in countries where we or our collaboration partners do not have patent rights, and then use the information learned from such activities to develop
competitive products for sale in our major commercial markets; or
- •
- we may not develop additional proprietary technologies that are patentable.
Issued patents covering our product candidates could be found invalid or unenforceable if challenged in court.
Even if our patents do successfully issue and even if such patents cover our product candidates and methods of use, third parties may initiate interference, re-examination, post-grant review, inter partes review, or derivation actions in the U.S. Patent and Trademark Office, or the USPTO, may initiate third party oppositions in the European Patent Office, or the EPO, or similar actions challenging the validity, enforceability or scope of such patents in other patent administrative proceedings worldwide, which may result in our patent claims being narrowed or invalidated. Such proceedings could result in revocation or amendment of our patents in such a way that they no longer cover our product candidates or competitive products. Further, if we initiate legal proceedings against a third party to enforce a patent covering our product candidate or technology, the defendant could counterclaim that the patent covering our product candidate or technology is invalid or unenforceable. In patent litigation in the United States, certain European and other countries worldwide, it is commonplace for defendants to make counterclaims alleging invalidity and unenforceability in the same proceeding, or to commence parallel defensive proceedings such as patent nullity actions to challenge validity and enforceability of asserted patent claims.
In administrative and court actions, grounds for a patent validity challenge may include alleged failures to meet any of several statutory requirements, including lack of novelty, obviousness (lack of inventive step) and in some cases, lack of sufficiently teaching, or non-enablement of, the claimed invention. Grounds for unenforceability assertions include allegations that someone connected with prosecution of the patent withheld relevant information from the Examiner during prosecution in the USPTO, or made a misleading statement during prosecution in the USPTO, the EPO or elsewhere. Third parties may also raise similar claims before administrative bodies in the USPTO or the EPO, even outside the context of litigation. The outcome following legal assertions of invalidity and unenforceability is unpredictable. With respect to validity, for example, we cannot be certain that there is no invalidating prior art, of which we or the patent examiner were unaware during prosecution. Further, we cannot be certain that all of the potentially relevant art relating to our patents and patent applications has been cited in every patent office. If a defendant were to prevail on a legal assertion of invalidity or unenforceability, we would lose at least part, and perhaps all, of the patent protection on our product candidates.
We may become involved in lawsuits to protect or enforce our patents or other intellectual property, which could be expensive, time consuming and unsuccessful and have a material adverse effect on the success of our business.
Competitors may infringe our patents, trademarks, copyrights or other intellectual property. To counter infringement or unauthorized use, we may be required to file infringement claims on a country-by-country basis, which can be expensive, unpredictable, time consuming and divert the time and attention of our management and scientific personnel. Any claims we assert against perceived infringers could provoke these parties to assert counterclaims against us alleging that we infringe their patents, in addition to counterclaims asserting that our patents are invalid or unenforceable, or both. In
25
any patent infringement proceeding, there is a risk that a court will decide that a patent of ours is invalid or unenforceable, in whole or in part, and that we do not have the right to stop the other party from using the invention at issue. There is also a risk that, even if the validity of such patents is upheld, the court will construe the patent's claims narrowly or decide that we do not have the right to stop the other party from using the invention at issue on the grounds that our patent claims do not cover the invention. An adverse outcome in a litigation or proceeding involving one or more of our patents could limit our ability to assert those patents against those parties or other competitors, and may curtail or preclude our ability to exclude third parties from making and selling similar or competitive products. Similarly, if we assert trademark infringement claims, a court may determine that the marks we have asserted are invalid or unenforceable, or that the party against whom we have asserted trademark infringement has superior rights to the marks in question. In this case, we could ultimately be forced to cease use of such trademarks.
Even if we establish infringement, the court may decide not to grant an injunction against further infringing activity and instead award only monetary damages, which may or may not be an adequate remedy. Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during litigation. There could also be public announcements of the results of hearings, motions or other interim proceedings or developments. If securities analysts or investors perceive these results to be negative, it could adversely affect the market price of the ADSs. Moreover, there can be no assurance that we will have sufficient financial or other resources to file and pursue such infringement claims, which typically last for years before they are concluded. Even if we ultimately prevail in such claims, the monetary cost of such litigation, any award of damages or loss of profit and the diversion of the attention of our management and scientific personnel could outweigh any benefit we receive as a result of the proceedings.
Claims that our product candidates or their uses infringe the intellectual property rights of third parties could result in costly litigation, and unfavorable outcomes could require us to pay damages or royalties and could limit our R&D activities or our ability to commercialize certain products.
Even if we have or obtain patents covering our product candidates, compositions or uses, we may still be barred from making, using, importing or selling our product candidates or technologies because of the patent rights of others. Others have filed, and in the future may file, patent applications covering compositions or products and uses that are similar or identical to ours. There are many-issued U.S., European and other worldwide patents relating to therapeutic drugs, and some of these relate to compounds we intend to commercialize. Numerous worldwide patents and pending patent applications owned by others exist in the metabolic disease, gastrointestinal disease and cardiovascular disease field and cover, among others, GLP-2 product candidates that we are developing. To the extent that we identify any potential issue with third party patents that may affect our product candidates, we ensure that we have a strategy to deal with such third party patents, either by ensuring that we believe that such patents are invalid, not infringed or that we commercialize our products upon expiry of such patents. Such strategies can include seeking a judicial or administrative revocation of such patents, ensuring that we are in a position to defend a claim for infringement, or seeking a license where that is appropriate. We cannot guarantee that our products, compositions and their uses do not or will not infringe third party patent or other intellectual property rights. Because patent applications can take 18 months to publish and many years to issue, there may be currently pending applications with patent claims unknown to us or which will change over time and may later result in issued patents that purportedly cover our product candidates or compositions and uses. These patent applications may have been filed earlier than or have priority over patent applications filed by us. We may be required to develop or obtain alternative technologies, review product design or, in the case of claims concerning registered trademarks, rename our product candidates.
26
Claims that our or our collaboration partners' products, compositions or their uses infringe or interfere with the patent rights of third parties, or that we or our collaboration partners have misappropriated third party trade secrets, could result in costly litigation and could require substantial time and money to resolve, even if litigation were avoided. The basis of such litigation could be existing patents or patents that are granted in the future. If we or our collaboration partners were to face infringement claims or challenges by third parties, an adverse outcome could subject us or our collaboration partners to significant liabilities to such third parties. Litigation or threatened litigation could result in significant demands on the time and attention of our management team. A negative outcome could expose us or our collaboration partners to payment of costs, damages and other financial remedies, including in some jurisdictions, increased damages, such as treble damages and attorneys' fees, if found to have willfully infringed a patent. Litigation with third parties concerning alleged infringement of their intellectual property rights could require us and our collaboration partners to bear substantial costs and impose burdens on our and their management and personnel, even if we or our collaboration partners were to ultimately succeed in such proceedings. Costs of patent litigation and awards of damages in patent infringement cases can be significant, and equitable remedies such as temporary restraining orders and injunctions can negatively impact or prevent product development and commercialization. In light of these risks, settlements are often a preferred alternative, to avoid litigation uncertainties and costs, even when there are strong defenses to claims that are made. A negative outcome, potential or actual, could cause us or our collaboration partners to pursue contractual and other remedies against each other; in particular, our license agreements generally allow our collaboration partners to reduce amounts we are owed as royalties and/or milestones by amounts paid to third parties as a result of or in settlement of certain infringement claims, subject to contractual conditions and limitations. We or our collaboration partners could also face equitable remedies, such as being forced, including by court order, to cease developing, manufacturing, importing or commercializing an infringing product candidate or product in one or more jurisdictions. A negative outcome could also lead us or our collaboration partners to delay, curtail or cease the development and commercialization of some or all of our candidate drugs, or could cause us or our collaboration partners to seek legal or administrative actions against third parties. We or our collaboration partners may need to obtain licenses from third parties and such licenses may not be available on commercially reasonable terms, or at all. Even if we or our collaboration partners are able to obtain a license, it could be non-exclusive, thereby giving our competitors access to the same rights licensed to us. In addition, even if we or our collaboration partners were ultimately to succeed in asserting one or more patent defenses in an infringement suit, or to settle at an early stage to avoid litigation uncertainties and costs despite having strong patent defenses, such litigation could burden us and our collaboration partners with substantial unanticipated costs and damages. A negative outcome could cause us or our collaboration partners to pursue contractual remedies against each other, including, for example, over settlement or license related payments or royalty reductions.
Biopharmaceutical patents and patent applications involve highly complex legal and factual questions, which, if determined adversely to us, could negatively impact our patent position.
The patent positions of biopharmaceutical companies can be highly uncertain and involve complex legal and factual questions. The interpretation and breadth of claims allowed in some patents covering biopharmaceutical compositions may be uncertain and difficult to determine, and are often affected materially by the facts and circumstances that pertain to the patented compounds, compositions and related patent claims. The standards of the USPTO, the EPO and other international patent offices are evolving and could change in the future. Consequently, we cannot predict the issuance and scope of patents with certainty. Patents, if issued, may be challenged, invalidated or circumvented. European patents and patents in certain other jurisdictions are subject to third party opposition proceedings. U.S. patents and patent applications may also be subject to interference proceedings, and U.S. patents may be subject to reexamination proceedings, post-grant review and/or inter partes review in the USPTO.
27
The issuance of a patent is not conclusive as to its inventorship, scope, validity or enforceability and our patents or pending patent applications may be challenged in the courts or patent offices in the United States, Europe and elsewhere worldwide. There is no assurance that all of the potentially relevant prior art relating to our patents and patent applications has been found. For example, publications of discoveries in the scientific literature often lag behind the actual discoveries, and patent applications are typically not published until 18 months after filing, or in some cases not at all. Therefore, we cannot know with certainty whether we were the first to make the inventions claimed in our owned and licensed patents or pending patent applications, or that we were the first to file for patent protection of such inventions. If such prior art exists, it may be used to invalidate a patent, or may prevent a patent from issuing from a pending patent application. For example, such patent filings may be subject to a third party pre-issuance submission of prior art to the USPTO, EPO or to other patent offices around the world. As a result, the issuance, scope, validity, enforceability and commercial value of our patent rights may be uncertain. Our pending and future patent applications may not result in patents being issued that protect our technology or products, in whole or in part, or may not effectively prevent others from commercializing competitive technologies and products. For example, such patent filings may be subject to a third party preissuance submission of prior art to the USPTO, the EPO or to other patent offices around the world. Alternately or additionally, we may become involved in post-grant review procedures, oppositions, derivations, proceedings, reexaminations, inter partes review or interference proceedings, in the United States or elsewhere, challenging patents or patent applications in which we have rights, including patents on which we rely to protect our business. An adverse determination in any such challenges may result in loss of exclusivity or in patent claims being narrowed, invalidated or held unenforceable, in whole or in part, which could limit our ability to stop others from using or commercializing similar or identical technology and products, or limit the duration of the patent protection of our technology and products. European patents or patents in other jurisdictions may be subject also to administrative opposition or comparable proceedings in corresponding worldwide patent offices, which could result in either loss of the patent or denial of the patent application or loss or reduction in the scope of one or more of the claims of the patent or patent application. In addition, such interference, reexamination, post-grant review, inter partes review and opposition proceedings may be time consuming and costly. Also, given the amount of time required for the development, testing and regulatory review of new product candidates, patents protecting such candidates may expire before or shortly after such candidates are commercialized. Accordingly, rights under any issued patents may not provide us with sufficient protection against competitive products or processes.
In addition, changes in or different interpretations of patent laws in the United States, Europe and other countries worldwide may diminish the value of our patents or narrow the scope of our patent protection, while patent reform legislation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents. For example, changes in or different interpretations of patent laws in the United States, Europe and other countries worldwide may permit others to use our or our collaboration partners' discoveries or to develop and commercialize our technology and products without providing any compensation to us, or may limit the number of patents or claims we can obtain. The laws of some countries may not protect intellectual property rights to the same extent as the laws of the United States or Europe, and those countries may lack adequate rules and procedures for defending our intellectual property rights, or vice versa.
If we fail to obtain and maintain patent protection and trade secret protection for our product candidates, we could lose our competitive advantage and competition we face would increase, reducing any potential revenue and adversely affecting our ability to attain or maintain profitability.
28
If we are unable to protect the confidentiality of our trade secrets and know-how, our business and competitive position would be harmed.
In addition to seeking patent protection for our product candidates, we also rely on trade secrets, including unpatented know-how, technology and other proprietary information, to maintain our competitive position. We seek to protect these trade secrets, in part, by entering into non-disclosure and confidentiality agreements with parties who have access to them, such as our employees, collaboration partners, consultants, advisors, university and/or institutional researchers and other third parties. We also have entered or seek to enter into confidentiality and invention or patent assignment agreements with our employees, advisors and consultants. Despite these efforts, any of these parties may breach the agreements and disclose our proprietary information, including our trade secrets, and we may not be able to obtain adequate remedies for such breaches. Our trade secrets may also be obtained by third parties by other means, such as breaches of our physical or computer security systems. Enforcing a claim that a party illegally disclosed or misappropriated a trade secret is difficult, expensive and time consuming, and the outcome is unpredictable. In addition, some courts inside and outside the United States are less willing or unwilling to protect trade secrets. Moreover, if any of our trade secrets were to be lawfully obtained or independently developed by a competitor, we would have no right to prevent them, or those to whom they communicate it, from using that technology or information to compete with us. If any of our trade secrets were to be disclosed to, or independently developed by, a competitor, our competitive position would be harmed.
We will not seek to protect our intellectual property rights in all jurisdictions throughout the world, and we may not be able to adequately enforce our intellectual property rights even in the jurisdictions where we seek protection.
Obtaining and maintaining a patent portfolio entails significant expense and resources. Part of the expense includes periodic maintenance fees, renewal fees, annuity fees, various other governmental fees on patents or applications due in several stages over the lifetime of patents or applications, as well as the cost associated with complying with numerous procedural provisions during the patent application process. Filing, prosecuting and defending patents on our product candidates in all countries and jurisdictions throughout the world would be prohibitively expensive, and our intellectual property rights in some countries outside the United States and Europe could be less extensive than those in the United States and in Europe, assuming that rights are obtained in the United States or in Europe. We may choose not to pursue or maintain protection for particular inventions. In addition, there are situations in which failure to make certain payments or noncompliance with certain requirements in the patent process can result in abandonment or lapse of a patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. If we choose to forego patent protection or allow a patent application or patent to lapse purposefully or inadvertently, our competitive position could suffer. Competitors may use our technologies in jurisdictions where we do not pursue and obtain patent protection to develop their own products and further, may export otherwise infringing products to territories where we have patent protection, but enforcement is not as strong as that in the United States or in Europe. These products may compete with our product candidates and our patents or other intellectual property rights may not be effective or sufficient to prevent them from competing. Even if we pursue and obtain issued patents in particular jurisdictions, our patent claims or other intellectual property rights may not be effective or sufficient to prevent third parties from so competing.
In addition, the laws of some countries do not protect intellectual property rights to the same extent as the federal and state laws in the United States and Europe. Many companies have encountered significant problems in protecting and defending intellectual property rights in certain foreign jurisdictions. The legal systems of some countries, particularly developing countries, do not favor the enforcement of patents and other intellectual property protection, especially those relating to
29
biopharmaceuticals or biotechnologies. This could make it difficult for us to stop the infringement of our patents, if obtained, or the misappropriation of our other intellectual property rights. For example, many foreign countries have compulsory licensing laws under which a patent owner must grant licenses to third parties. In addition, many countries limit the enforceability of patents against third parties, including government agencies or government contractors. In these countries, patents may provide limited or no benefit. Patent protection must ultimately be sought on a country-by-country basis, which is an expensive and time consuming process with uncertain outcomes. Accordingly, we may choose not to seek patent protection in certain countries, and we will not have the benefit of patent protection in such countries. Proceedings and legal actions to enforce our patent rights in the United States or in Europe and in foreign jurisdictions can be expensive, could result in substantial costs, and could divert management time and our efforts and attention from other aspects of our business. In addition, such proceedings or legal actions could put our patents at risk of being invalidated, found unenforceable or interpreted narrowly, could put our patent applications at risk of not being issued and could provoke third parties to assert claims against us. We may not prevail in any lawsuits that we initiate and the damages or other remedies awarded, if any, may not be commercially meaningful. We may or may not choose to pursue litigation or other actions against those that have infringed our patents, or used them without authorization, due to the associated expense and time commitment of monitoring these activities. If we fail to protect or to enforce our intellectual property rights successfully, our competitive position could suffer, which could harm our results of operations.
In addition, changes in the law and legal decisions by courts in the United States, Europe and foreign countries may affect our ability to obtain adequate protection for our technology and the enforcement of intellectual property. Accordingly, our efforts to enforce our intellectual property rights around the world may be inadequate to obtain a significant commercial advantage from the intellectual property that we develop or license.
Patent terms and regulatory exclusivities may be inadequate to protect our competitive position on our product candidates for an adequate amount of time.
Given the amount of time required for the development, testing and regulatory review of new product candidates, patents protecting such candidates might expire before or shortly after such candidates are commercialized. We expect to seek extensions of patent terms in the United States and, if available, in other countries where we have patents.
Depending upon the timing and duration of the U.S. regulatory review process and patent life considerations, certain of our U.S. patents may be eligible for patent term extension under the Drug Price Competition and Patent Term Restoration Act of 1984, or the Hatch-Waxman Amendments. The Hatch-Waxman Amendments provide up to five years of patent term extension, or PTE, on a patent that covers an approved product or method of use as compensation for patent term lost during the FDA regulatory review process. Patent term restoration cannot extend the term of a patent beyond a total of 14 years from the product's approval date. Only one patent with a claim covering an approved drug or method is eligible for the extension, and the extension must be applied for prior to the patent expiration date (which due date may be extended by submission of one or more applications for interim extensions for periods of up to one year each and cannot be extended longer than the maximum period of patent term extension). The USPTO, in consultation with the FDA, reviews and approves a request for patent term extension or restoration and calculates the PTE period that will be awarded. PTE only extends patent coverage on the approved product or method of use.
In certain Member States of the EU, patent term extensions may be obtained through a Supplementary Protection Certificate to recover some of the time lost between the patent application filing date and the date of first regulatory approval, up to a maximum term of five years. Up to five years of patent term extension are also available in Japan for patent term recovery related to the pharmaceutical regulatory review and approval process.
30
Applicable authorities, including the FDA/USPTO in the United States, and comparable regulatory authorities and intellectual property offices in other EU countries and worldwide, may not agree with our assessment of whether such extensions are available, and may refuse to grant extensions to our patents, or may grant more limited extensions than we request. If this occurs, our competitors may be able to take advantage of our investment in development and clinical trials by referencing our clinical and preclinical data and launch their product earlier than might otherwise be the case.
Third parties may challenge the inventorship of our patent filings and other intellectual property or may assert ownership or commercial rights to inventions we develop.
Third parties may in the future make claims challenging the inventorship or ownership of our intellectual property. We have written agreements with collaboration partners that provide for the ownership of intellectual property arising from our collaborations. These agreements provide that we or our licensees must negotiate certain commercial rights with collaboration partners with respect to joint inventions or inventions made by our collaboration partners that arise from the results of the collaboration. In addition, our standard employment contracts ensure that any inventions are ours by right and this contractual position is in addition to our rights to any invention by the operation of law where we conduct research and development ourselves.
In some instances, there may not be, or parties may dispute that there are, adequate written provisions to address clearly the resolution of intellectual property rights that may arise from collaboration. If we or our licensees cannot successfully negotiate sufficient ownership and commercial rights to the inventions that result from our use of a third party collaboration partner's materials where required, or if disputes otherwise arise with respect to the intellectual property developed with the use of a collaboration partner's samples, we may be limited in our ability to capitalize on the market potential of these inventions. In addition, we may face claims by third parties that our agreements with employees, contractors or consultants obligating them to assign intellectual property to us are ineffective, or in conflict with prior or competing contractual obligations of assignment, which could result in ownership disputes regarding intellectual property we have developed or will develop and interfere with our ability to capture the commercial value of such inventions. Litigation may be necessary to resolve an ownership dispute, and if we are not successful, we may be precluded from using certain intellectual property, or may lose our exclusive rights in that intellectual property. Either outcome could have an adverse impact on our business, financial position, results of operations and future growth prospects.
Third parties may assert that our employees or consultants or we have wrongfully used or disclosed confidential information or misappropriated trade secrets, or claim ownership of what we regard as our own intellectual property.
We employ individuals who were previously employed at universities or other biotechnology or pharmaceutical companies, including our competitors or potential competitors. Although we try to ensure that our employees and consultants do not use the proprietary information or know-how of others in their work for us, and no such claims against us are currently pending, we may be subject to claims that we or our employees, consultants or independent contractors have used or disclosed intellectual property, including trade secrets or other proprietary information, of a former employer or other third parties. Litigation may be necessary to defend against these claims. If we fail in defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights or personnel. Even if we are successful in defending against such claims, litigation could result in substantial costs and be a distraction to management and other employees.
31
Obtaining and maintaining patent protection depends on compliance with various procedural, document submission, fee payment and other requirements imposed by governmental patent agencies, and our patent protection could be reduced or eliminated for non-compliance with these requirements.
Periodic maintenance fees, renewal fees, annuity fees and various other governmental fees on patents and applications are required to be paid to the USPTO and various governmental patent agencies outside the United States in several stages over the lifetime of the patents and applications. The USPTO and various non-U.S. governmental patent agencies require compliance with a number of procedural, documentary, fee payment and other similar provisions during the patent application process and after a patent has issued. There are situations in which non-compliance can result in abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction.
Risks Related to Government Regulation
Government restrictions on pricing and reimbursement, as well as other healthcare payor cost-containment initiatives, may negatively impact our ability to generate revenue.
Sales of certain of our out-licensed products and our product candidates, if and when approved for marketing, has and will depend, in part, on the extent to which our products will be covered by third party payors, such as government health care programs like Medicare and Medicaid, commercial insurance and managed healthcare organizations. These third party payors play an important role in determining the extent to which new drugs, biologics and medical devices will be covered. The Medicare and Medicaid programs increasingly are used as models for how private payors and other governmental payors develop their coverage and reimbursement policies for drugs, biologics and medical devices. It is difficult to predict at this time what third party payors will decide with respect to the coverage and reimbursement for our product candidates. The primary trend in the U.S. healthcare industry and elsewhere has been cost containment, including price controls, restrictions on coverage and reimbursement and requirements for substitution of generic products and/or biosimilars. Adoption of price controls, cost containment measures and adoption of more restrictive policies in jurisdictions with existing controls and measures, could limit our net revenue and results.
Government authorities and third-party payors have attempted to control costs by limiting coverage and the amount of reimbursement for medical products, drugs and services. Increasingly, third-party payors are requiring that drug companies provide them with predetermined discounts from list prices and are challenging the prices charged for drugs. We cannot be sure that coverage will be available for any product candidate that we commercialize and, if coverage is available, the level of reimbursement. Because coverage and reimbursement determinations are made on a payor-by-payor basis, obtaining coverage and adequate reimbursement from a third party payor does not guarantee that we will obtain similar coverage or reimbursement from another third party payor. Reimbursement may impact the demand for, or the price of, any product candidate for which we obtain marketing approval. If reimbursement is not available or is available only to limited levels, we may not be able to successfully commercialize any product candidate for which we obtain marketing approval. Decreases in third party reimbursement for our product candidates or a decision by a third party payor not to cover our product candidates or provide only limited reimbursement for our product candidates could reduce physician usage of our products once approved and have a material adverse effect on our sales, results of operations and financial condition. Further, the adoption and implementation of any future governmental cost containment or other health reform initiative may result in additional downward pressure on the price that we may receive for any approved product.
Outside the United States, international operations are generally subject to extensive governmental price controls and other market regulations, and we believe the increasing emphasis on cost-containment initiatives in Europe, Canada, and other countries has and will continue to put
32
pressure on the pricing and usage of our product candidates. In many countries, the prices of medical products are subject to varying price control mechanisms as part of national health systems. Other countries allow companies to fix their own prices for medical products, but monitor and control company profits. Additional foreign price controls or other changes in pricing regulation could restrict the amount that we are able to charge for our product candidates. Accordingly, in markets outside the United States, the reimbursement for our products may be reduced compared with the United States and may be insufficient to generate commercially reasonable revenue and profits.
Moreover, increasing efforts by governmental and third party payors in the United States and abroad to cap or reduce healthcare costs may cause such organizations to limit both coverage and the level of reimbursement for new products approved and, as a result, they may not cover or provide adequate payment for our product candidates. We expect to experience pricing pressures in connection with the sale of any of our product candidates due to the trend toward managed healthcare, the increasing influence of health maintenance organizations, and additional legislative changes. The downward pressure on healthcare costs in general, particularly prescription drugs, medical devices and surgical procedures and other treatments, has become very intense. As a result, increasingly high barriers are being erected to the successful commercialization of new products.
We may face difficulties from changes to current regulations and future legislation.
Existing regulatory policies may change and additional government regulations may be enacted that could prevent, limit or delay regulatory approval of our product candidates. We cannot predict the likelihood, nature or extent of government regulation that may arise from future legislation or administrative action, either in the United States or abroad. If we are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we are not able to maintain regulatory compliance, we may lose any marketing approval that we may have obtained and we may not achieve or sustain profitability.
The current presidential administration and U.S. Congress have also attempted to repeal or "repeal and replace" the Affordable Care Act, or the ACA. Although those efforts did not succeed, the presidential administration may continue to seek to modify, repeal, or otherwise invalidate all, or certain provisions of, the ACA. There is still uncertainty with respect to the impact President Trump's administration and the U.S. Congress may have on the ACA, if any, and any changes will likely take time to unfold. Additionally, there remain judicial and Congressional challenges to certain aspects of the ACA. For example, on December 14, 2018, a U.S. District Court Judge in the Northern District of Texas, or Texas District Court Judge, ruled that the entire ACA is invalid based primarily on the fact that the Tax Cuts and Jobs Act of 2017 repealed the tax-based shared responsibility payment imposed by the ACA, on certain individuals who fail to maintain qualifying health coverage for all or part of a year, which is commonly referred to as the "individual mandate". Additionally, on December 18, 2019, the U.S. Court of Appeals for the 5th Circuit upheld the District Court ruling that the individual mandate was unconstitutional and remanded the case back to the District Court to determine whether the remaining provisions of the PPACA are invalid as well. It is unclear how this decision, future decisions, and subsequent appeals will impact the law and the effect such impact could have on coverage and reimbursement for healthcare items and services covered by plans that were authorized by the ACA.
In addition, other legislative changes have been proposed and adopted since the ACA was enacted. These changes included aggregate reductions to Medicare payments to providers of up to 2% per fiscal year, effective April 1, 2013, which, due to subsequent legislative amendments, will stay in effect through 2029 unless additional Congressional action is taken. In January 2013, President Obama signed into law the American Taxpayer Relief Act of 2012, which, among other things, reduced Medicare payments to several providers and increased the statute of limitations period for the U.S. government to recover overpayments to providers from three to five years. These laws may result in additional reductions in Medicare and other healthcare funding, which could have a material adverse effect on customers for our out-licensed products and product candidates (if and when approved) and accordingly, our financial results.
33
Likewise, the annual Medicare Physician Fee Schedule update, which, until recently, was based on a target-setting formula system called the Sustainable Growth Rate, or SGR, , was adjusted to reflect the comparison of actual expenditures to target expenditures. Because one of the factors for calculating the SGR was linked to the growth in the U.S. gross domestic product, or GDP, the SGR formula often resulted in a negative payment update when growth in Medicare beneficiaries' use of services exceeded GDP growth. Congress repeatedly intervened to delay the implementation of negative SGR payment updates. However, the Medicare Access and CHIP Reauthorization Act of 2015, or MACRA, ended the use of the statutory formula and established a quality payment program, also referred to as the Quality Payment Program. . The quality payment program has two tracks, one known as the merit based incentive payment system for providers in the fee-for service Medicare program, and the advanced alternative payment model for providers in specific care models, such as accountable care organizations. In November 2019, CMS issued a final rule finalizing the changes to the Quality Payment Program. At this time it is unclear how the introduction of the Quality Payment Program will impact overall physician reimbursement under the Medicare program. .
There have been several recent U.S. Congressional inquiries and proposed and enacted federal and state legislation designed to, among other things, bring more transparency to drug pricing, review the relationship between pricing and manufacturer patient programs, reduce the cost of drugs under Medicare, and reform government program reimbursement methodologies for drugs. At the federal level, the Trump administration's budget proposal for fiscal year 2021 includes a $135 billion allowance to support legislative proposals seeking to reduce drug prices, increase competition, lower out-of-pocket drug costs for patients, and increase patient access to lower-cost generic and biosimilar drugs Further, the Trump administration previously released a "Blueprint" to lower drug prices and reduce out of pocket costs of drugs that contains additional proposals to increase drug manufacturer competition, increase the negotiating power of certain federal healthcare programs, incentivize manufacturers to lower the list price of their products, and reduce the out of pocket costs of drug products paid by consumers. The Department of Health and Human Services, or HHS, has solicited feedback on some of these measures and has implemented others under its existing authority. Congress and the Trump administration have each indicated that it will continue to seek new legislative and/or administrative measures to control drug costs. At the state level, legislatures are increasingly passing legislation and implementing regulations designed to control pharmaceutical and biological product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures, and, in some cases, such measures are designed to encourage importation from other countries and bulk purchasing. We expect that additional state and federal healthcare reform measures will be adopted in the future, any of which could limit the amounts that federal and state governments will pay for healthcare products and services, which could result in reduced demand for our product candidates or additional pricing pressures.
We expect more rigorous coverage criteria in the future in the U.S. healthcare market and an additional downward pressure on the prices that we receive for approved products. Any reduction in reimbursement from Medicare or other government programs may result in a similar reduction in payments from private payors. The implementation of cost containment measures or other healthcare reforms may prevent us from being able to generate revenue, attain profitability or commercialize our out-licensed products and product candidates.
In addition, it is not currently possible to predict how, if at all, the FDA's approved process or regulation will change as a result of the Trump Administration or what impact any such changes will have on us.
34
Our operations involve hazardous materials and we and third parties with whom we contract must comply with environmental and safety laws and regulations, which can be expensive and restrict how we do business.
As a pharmaceutical company, we are subject to environmental and safety laws and regulations, including those governing the use of hazardous materials. The cost of compliance with health and safety regulations is substantial. Our business activities involve the controlled use of hazardous materials. Our R&D activities involve the controlled storage, use and disposal of hazardous materials, including the components of our product candidates and other hazardous compounds. We and manufacturers and suppliers with whom we may contract are subject to laws and regulations governing the use, manufacture, storage, handling and disposal of these hazardous materials. In some cases, these hazardous materials and various wastes resulting from their use are stored at our and our manufacturers' facilities pending their use and disposal. We cannot eliminate the risk of accidental contamination or injury from these materials, which could cause an interruption of our commercialization efforts, R&D efforts and business operations, environmental damage resulting in costly clean-up and liabilities under applicable laws and regulations governing the use, storage, handling and disposal of these materials and specified waste products. We cannot guarantee that that the safety procedures utilized by third party manufacturers and suppliers with whom we may contract will comply with the standards prescribed by laws and regulations or will eliminate the risk of accidental contamination or injury from these materials. In such an event, we may be held liable for any resulting damages and such liability could exceed our resources and European, U.S. federal and state or other applicable authorities may curtail our use of certain materials and/or interrupt our business operations. Furthermore, environmental laws and regulations are complex, change frequently and have tended to become more stringent. We cannot predict the impact of such changes and cannot be certain of our future compliance. We do not currently carry biological or hazardous waste insurance coverage. In the event of an accident or environmental discharge, we may be held liable for any consequential damage and any resulting claims for damages, which may exceed our financial resources and may materially adversely affect our business, results of operations and prospects, and the value of our shares.
We are subject to healthcare laws and regulations, which may require substantial compliance efforts and could expose us to criminal sanctions, civil penalties, exclusion from government healthcare programs, contractual damages, reputational harm and diminished profits and future earnings, among other penalties.
Our current and future operations may directly, or indirectly through our prescribers, customers and purchasers, expose us to broadly applicable fraud and abuse and other healthcare laws and regulations that may constrain the business or financial arrangements and relationships through which we research, market, sell and distribute our products. Restrictions under applicable U.S. federal, state, local and non-U.S. healthcare laws and regulations include, but are not limited to, the following:
- •
- the federal Anti-Kickback Statute, which prohibits, among other things, persons or entities from knowingly and willfully soliciting, offering,
receiving or providing remuneration, including any kickback, bribe or rebate, directly or indirectly, in cash or in kind, to induce or reward, or in return for, either the referral of an individual
for, or the purchase or lease, order or recommendation of, any item, good, facility or service, for which payment may be made under federal healthcare programs such as Medicare and Medicaid;
- •
- the federal Beneficiary Inducement Statute, which prohibits giving anything of value to a government insurance beneficiary that could influence
the choice of provider or reimbursable covered product;
- •
- federal civil and criminal false claims laws, including the civil False Claims Act, and civil monetary penalties laws, which impose criminal and civil penalties, including those from civil whistleblower or qui tam actions, against individuals or entities for, among other things, knowingly presenting, or causing to be presented, claims for payment that are false or fraudulent
35
- •
- the anti-inducement law prohibits, among other things, the offering or giving of remuneration, which includes, without limitation, any transfer
of items or services for free or for less than fair market value (with limited exceptions), to a Medicare or Medicaid beneficiary that the person knows or should know is likely to influence the
beneficiary's selection of a particular supplier of items or services reimbursable by a federal or state governmental program;
- •
- the federal Health Insurance Portability and Accountability Act of 1996, or HIPAA, which created additional federal criminal statutes that
impose criminal and civil liability for, among other things, executing or attempting to execute a scheme to defraud any healthcare benefit program or knowingly and willingly falsifying, concealing or
covering up a material fact or making false statements relating to healthcare matters;
- •
- HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act and its implementing regulations, which impose
certain requirements on covered entities and their business associates, including mandatory contractual terms, with respect to safeguarding the privacy, security and transmission of individually
identifiable health information;
- •
- the federal transparency requirements under the Physician Payments Sunshine Act, enacted as part of the Patient Protection and ACA that require
applicable manufacturers of covered drugs, devices, biologics and medical supplies for which payment is available under Medicare, Medicaid, or the Children's Health Insurance Program, with specific
exceptions, to track and annually report to the Centers for Medicare & Medicaid Services, or CMMS, payments and other transfers of value provided to physicians, as defined by such law, and
teaching hospitals, and certain ownership and investment interests held by physicians or their immediate family members;
- •
- analogous state, local and non-U.S. laws and regulations, such as state anti-kickback and false claims laws, which may apply to items or
services reimbursed by any third party payor, including commercial insurers; state, local and non-U.S. marketing and/or transparency laws applicable to manufacturers that may be broader in scope than
the federal requirements; state laws that require biopharmaceutical companies to comply with the biopharmaceutical industry's voluntary compliance guidelines, relevant compliance guidance promulgated
by the federal government, implementation of compliance programs, and compliance with the state's code of conduct; state and local laws that require a pharmaceutical company's sales representatives to
be registered or licensed by the state or local governmental entity; and state and non-U.S. laws governing the privacy and security of health information in certain circumstances, many of which differ
from each other in significant ways and may be more stringent than HIPAA, thus complicating compliance efforts; and
- •
- rules or legislation covering more or less the same subject matter are found in numerous other countries, including in Denmark, which sometimes result in lower or higher exposures in those countries than in the United States.
or making a false statement to avoid, decrease or conceal an obligation to pay money to the federal government;
Ensuring that our business arrangements with third parties comply with applicable healthcare laws and regulations will likely be costly. It is possible that governmental authorities will conclude that our business practices do not comply with current or future statutes, regulations or case law involving applicable fraud and abuse or other healthcare laws and regulations. If our operations were found to be in violation of any of these laws or any other governmental regulations that may apply to us, we may be subject to significant civil, criminal and administrative penalties, damages, fines, disgorgement, imprisonment, possible exclusion from government funded healthcare programs, such as Medicare and Medicaid, integrity obligations, contractual damages, reputational harm, diminished profits and future
36
earnings, and curtailment of our operations, any of which could substantially disrupt our operations. If the physicians or other providers or entities with whom we expect to do business are found not to be in compliance with applicable laws, they may be subject to significant criminal, civil or administrative sanctions, including exclusion from government funded healthcare programs.
The risk of us being found in violation of these laws is increased by the fact that many of them have not been fully interpreted by the regulatory authorities or the courts, and their provisions are open to a variety of interpretations. For example, the definition of the "remuneration" under the federal Anti-Kickback Statute has been interpreted to include anything of value. Further, courts have found that if "one purpose" of remuneration is to induce referrals, the federal Anti-Kickback Statute is violated.
Additionally, recent healthcare reform legislation has strengthened federal and state healthcare fraud and abuse laws. For example, the ACA amends the intent requirement of the federal Anti-Kickback Statute and criminal healthcare fraud statutes to clarify that liability under these statutes does not require a person or entity to have actual knowledge of the statutes or a specific intent to violate them. Moreover, the ACA provides that the government may assert that a claim that includes items or services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the civil False Claims Act. Because of the breadth of these laws and the narrowness of the statutory exceptions and safe harbors available, it is possible that some of our business activities could be subject to challenge under one or more of such laws.
Our employees and collaboration partners may engage in misconduct or other improper activities, including violating applicable regulatory standards and requirements or engaging in insider trading, which could significantly harm our business.
We are exposed to the risk of employee fraud or other misconduct and the fraud and misconduct of our collaboration partners. Misconduct by our employees or our collaboration partners could include intentional failures to:
- •
- comply with legal requirements or the requirements of the FDA, the EMA, the CMMS and other comparable regulatory authorities;
- •
- provide accurate information to applicable government authorities;
- •
- comply with fraud and abuse and other healthcare laws and regulations in the United States, or similar laws in Denmark and elsewhere;
- •
- comply with the FCPA and other applicable anti-bribery laws;
- •
- report financial information or data accurately; or
- •
- disclose unauthorized activities to us.
In particular, sales, marketing and business arrangements in the healthcare industry are subject to extensive laws and regulations intended to prevent fraud, misconduct, kickbacks, self-dealing, bribery and other abusive practices. These laws and regulations restrict or prohibit a wide range of pricing, discounting, marketing and promotion, sales commission, customer incentive programs and other business arrangements. Employee or collaboration partner misconduct could also involve the improper use of, including trading on, information obtained in the course of clinical trials, which could result in regulatory sanctions and serious harm to our reputation. We have adopted a code of conduct, but it is not always possible to identify and deter employee misconduct, and the precautions we take to detect and prevent this activity may be ineffective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to comply with these laws or regulations. If any such actions are instituted against us, and we or such collaboration are not successful in defending ourselves or asserting our rights, those actions could have
37
a significant impact on our business, including the imposition of significant fines or other sanctions. Further, if any actions are instituted against any of our collaboration partners and such partner fails to defend itself or assert its rights and as a result, is subjected to criminal, civil or administrative sanctions, including exclusion from government funded healthcare programs, such actions and outcomes could have a significant impact on our business.
If we are a passive foreign investment company, there could be adverse U.S. federal income tax consequences to U.S. Holders.
Under the Internal Revenue Code of 1986, as amended, or the Code, we will be a passive foreign investment company, or PFIC, for any taxable year in which (1) 75% or more of our gross income consists of passive income or (2) 50% or more of the average quarterly value of our assets consists of assets that produce, or are held for the production of, passive income, including cash. For purposes of these tests, passive income includes dividends, interest, gains from the sale or exchange of investment property and certain rents and royalties. In addition, for purposes of the above calculations, a non-U.S. corporation that directly or indirectly owns at least 25% by value of the shares of another corporation is treated as if it held its proportionate share of the assets and received directly its proportionate share of the income of such other corporation. If we are a PFIC for any taxable year during which a U.S. Holder (as defined in Item 10.E. "Taxation—Material U.S. Federal Income Tax Considerations for U.S. Holders") holds the ADSs, the U.S. Holder may be subject to adverse tax consequences regardless of whether we continue to qualify as a PFIC, including ineligibility for any preferred tax rates on capital gains or on actual or deemed dividends, interest charges on certain taxes treated as deferred, and additional reporting requirements.
We do not believe we were a PFIC for our taxable year ended December 31, 2019. We have not yet made any determination as to our expected PFIC status for our taxable year ended December 31, 2020 and, accordingly, any such expectation would be subject to change based on, among other factors, our use of cash, the source and nature of our income, and the price of our ordinary shares or ADSs. No assurances regarding our PFIC status can be provided for any past, current or future taxable years. The determination of whether we are a PFIC is a fact-intensive determination made on an annual basis and the applicable law is subject to varying interpretation. In particular, the characterization of our assets as active or passive may depend in part on our current and intended future business plans, which are subject to change. In addition, for our current and future taxable years, the total value of our assets for PFIC testing purposes may be determined in part by reference to the market price of our ordinary shares or ADSs from time to time, which may fluctuate considerably. Under the income test, our status as a PFIC depends on the composition of our income which will depend on the transactions we enter into in the future and our corporate structure. The composition of our income and assets is also affected by how, and how quickly, we spend the cash we raise in any offering. Our U.S. counsel expresses no opinion with respect to our PFIC status for our taxable year ended December 31, 2019, and also expresses no opinion with regard to our expectations regarding our PFIC status in the future.
If we are a PFIC, U.S. Holders (as defined in Item 10.E. "Taxation—Material U.S. Federal Income Tax Considerations for U.S. Holders) of the ADSs would be subject to adverse U.S. federal income tax consequences, such as ineligibility for any preferred tax rates on capital gains or on actual or deemed dividends, interest charges on certain taxes treated as deferred, and additional reporting requirements under U.S. federal income tax laws and regulations. For further discussion of the PFIC rules and the adverse U.S. federal income tax consequences in the event we are classified as a PFIC, see Item 10.E. "Taxation—Material U.S. Federal Income Tax Considerations for U.S. Holders."
38
If a United States person is treated as owning at least 10% of our ordinary shares, including ordinary shares represented by ADSs, such holder may be subject to adverse U.S. federal income tax consequences.
If a U.S. Holder is treated as owning (directly, indirectly or constructively through the application of attribution rules) at least 10% of the value or voting power of our ordinary shares, including ordinary shares represented by ADSs, such U.S. Holder may be treated as a "United States shareholder" with respect to each "controlled foreign corporation" in our group (if any). Because our group includes at least one U.S. subsidiary (Zealand Pharma US, Inc.), certain of our non-U.S. subsidiaries may be treated as controlled foreign corporations (regardless of whether Zealand Pharma A/S is treated as a controlled foreign corporation). A United States shareholder of a controlled foreign corporation may be required to annually report and include in its U.S. taxable income its pro rata share of "Subpart F income," "global intangible low-taxed income" and investments in U.S. property by controlled foreign corporations, regardless of whether we make any distributions. An individual that is a United States shareholder with respect to a controlled foreign corporation generally would not be allowed certain tax deductions or foreign tax credits that would be allowed to a United States shareholder that is a U.S. corporation. We cannot provide any assurances that we will assist investors in determining whether any of our non-U.S. subsidiaries, if any, are treated as a controlled foreign corporation or whether such investor is treated as a United States shareholder with respect to any of such controlled foreign corporations. Further, we cannot provide any assurances that we will furnish to any U.S. shareholder information that may be necessary to comply with the reporting and tax paying obligations discussed above. Failure to comply with these reporting obligations may subject you to significant monetary penalties and may prevent the statute of limitations with respect to your U.S. federal income tax return for the year for which reporting was due from starting. U.S. Holders should consult their tax advisors regarding the potential application of these rules to their investment in the ADSs.
Tax authorities may disagree with our positions and conclusions regarding certain tax positions, resulting in unanticipated costs, taxes or non-realization of expected benefits.
A tax authority may disagree with tax positions that we have taken, which could result in increased tax liabilities. For example, the U.S. Internal Revenue Service or another tax authority could challenge our allocation of income by tax jurisdiction and the amounts paid between our affiliated companies pursuant to our intercompany arrangements and transfer pricing policies, including amounts paid with respect to our intellectual property development. Similarly, a tax authority could assert that we are subject to tax in a jurisdiction where we believe we have not established a taxable connection, often referred to as a "permanent establishment" under international tax treaties, and such an assertion, if successful, could increase our expected tax liability in one or more jurisdictions. A tax authority may take the position that material income tax liabilities, interest and penalties are payable by us, in which case, we expect that we might contest such assessment. Contesting such an assessment may be lengthy and costly and if we were unsuccessful in disputing the assessment, the implications could increase our anticipated effective tax rate, where applicable.
Changes in Danish, U.S. or other foreign tax laws or compliance requirements, or the practical interpretation and administration thereof, could have a material adverse effect on our business, financial condition and results of operations.
We are affected by various Danish, U.S. and foreign taxes, including direct and indirect taxes imposed on our global activities, such as corporate income, withholding, customs, excise/energy, value added, sales, environmental and other taxes. Significant judgment is required in determining our provisions for taxes and there are many transactions and calculations where the ultimate tax determination is uncertain.
39
In recent years, tax authorities around the world have increased their scrutiny of company tax filings, and have become more rigid in exercising any discretion they may have. As part of this, the Organization for Economic Co-operation and Development, or OECD, has proposed a number of tax law changes under its Base Erosion and Profit Shifting, or BEPS, Action Plans to address issues of transparency, coherence and substance.
At the same time, the European Commission is finalizing its Anti-Tax Avoidance Directive, which seeks to prevent tax avoidance by companies and to ensure that companies pay appropriate taxes in the markets where profits are effectively made and business is effectively performed. The European Commission also continues to extend the application of its policies seeking to limit fiscal aid by Member States to particular companies, and the related investigation of the Member States' practices regarding the issuance of rulings on tax matters relating to individual companies.
These OECD and EU tax reform initiatives also need local country implementation, including in our home country of Denmark, which may result in significant changes to established tax principles. Although we have taken steps to be in compliance with the evolving OECD and EU tax initiatives, and will continue to do so, significant uncertainties remain as to the outcome of these efforts.
In general, such tax reform efforts, including with respect to tax base or rate, transfer pricing, intercompany dividends, cross border transactions, controlled corporations, and limitations on tax relief allowed on the interest on intercompany debt, will require us to continually assess our organizational structure against tax policy trends, and could lead to an increased risk of international tax disputes and an increase in our effective tax rate, and could adversely affect our financial results.
Changes in Danish or foreign direct or indirect tax laws or compliance requirements, including the practical interpretation and administration thereof, including in respect to market practices, or otherwise, could have a material adverse effect on our business, financial position, results of operations and future growth prospects.
PCAOB inspection of our independent auditors
With Zealand Pharma A/S being a public company listed in the United States, our independent public accounting firm, Deloitte Statsautoriseret Revisionspartnerselskab (CVR no 33 96 35 56), is registered with the PCAOB and therefore required to undergo regular PCAOB inspections to assess the registered accounting firm's compliance with United States law and professional standards in connection with its audits of financial statements filed with the SEC.
ITEM 4 INFORMATION ON THE COMPANY
A. HISTORY AND DEVELOPMENT OF THE COMPANY
We were founded in 1998 and are a biotechnology company focused on the discovery, design and development of innovative peptide-based medicines. We intend to be a leader in specialty medicines focusing on metabolic and gastrointestinal diseases and other specialty disease areas with significant unmet medical needs.
Our current pipeline of internal product candidates focuses on specialty gastrointestinal and metabolic diseases where we believe that the present standard of care is inadequate. In addition, we are looking to focus our efforts on drug candidates that may qualify for orphan/rare disease status.
40
Our shares are listed on Nasdaq Copenhagen (ZEAL). Our American Depositary Shares (ADS) are listed on the Nasdaq Global Select Market in the United States (ZEAL).
| Legal name: | Zealand Pharma A/S | |
| Commercial name: | Zealand Pharma | |
| Domicile: | Sydmarken 11 2860 Søborg (Copenhagen), Denmark | |
| Tel: | +45 8877 3600 | |
| Fax: | +45 8877 3898 | |
| Website: | zealandpharma.com |
The SEC maintains an Internet site at www.sec.gov that contains reports, proxy and information statements, and other information regarding issuers that file electronically with the SEC. Our website address is www.zealandpharma.com.
(The contents of this website are not incorporated by reference into this Annual Report on Form 20-F.)
| Date of incorporation: | December 22, 1998 (formerly incorporated beginning on April 1, 1997 as a private limited liability company) | |
| Legal form of the Company: | A Danish public limited liability company | |
| Legislation under which the Company operates: | Danish law | |
| Country of incorporation: | Denmark |
Emerging Growth Company
As a company with less than $1.07 billion in revenue during our last fiscal year, we qualify as an "emerging growth company," as defined in the Jumpstart Our Business Startups Act, or the JOBS Act. An emerging growth company may take advantage of specified reduced reporting and other burdens that are otherwise applicable generally to public companies. These provisions include:
- •
- a requirement to have only two years of audited financial statements and only two years of related Management's Discussion and Analysis of
Financial Condition and Results of Operations disclosure; and
- •
- an exemption from the auditor attestation requirement in the assessment of our internal control over financial reporting pursuant to the Sarbanes-Oxley Act.
We may take advantage of these provisions for up to five years or such earlier time that we are no longer an emerging growth company. We would cease to be an emerging growth company as of the end of any fiscal year following the fifth anniversary of our initial public offering or if, prior to that date, we have more than $1.07 billion in annual revenue or more than $700 million in market value of the equity securities held by non-affiliates as of the end of our second fiscal quarter, or on the date that we have issued more than $1 billion of non-convertible debt over a three-year period. We may choose to take advantage of some but not all of these reduced burdens, and therefore the information that we provide holders of shares and ADSs may be different than the information available from other public companies. In addition, Section 107 of the JOBS Act also provides that an emerging growth company can take advantage of an extended transition period for complying with new or revised accounting standards applicable to public companies. We currently prepare our consolidated financial statements in accordance with IFRS as issued by the IASB, which do not have separate provisions for publicly traded and private companies. However, in the event that we convert to accounting principles generally accepted in the United States (which we do not currently intend to do) while we remain an emerging growth company, we have irrevocably elected to opt out of such extended transition period.
41
Important events
Sanofi License Agreement and Royalty Pharma Agreement
Since 2003 we have had a license collaboration with Sanofi, or the Sanofi License Agreement, in the diabetes field pertaining to the development and commercialization of lixisenatide, both as a standalone therapy and as a combination therapy. Pursuant to the Sanofi License Agreement, we were entitled to receive certain royalties and commercial milestones in respect of global net-sales of Soliqua® 100/33/ Suliqua® and Lyxumia®/Adlyxin®. In September 2018 we, together with two of our wholly-owned subsidiaries, entered into a purchase and sale agreement with Royalty Pharma, or the Royalty Pharma Agreement, pursuant to which we sold and transferred our and our subsidiaries' respective rights to receive royalties and $85 million of potential commercial milestones in respect of global net sales of Soliqua® 100/33/ Suliqua® and Lyxumia®/Adlyxin® from and after July 1, 2018. In September 2018 we received DKK 1,310.2 million, or $205.0 million, upon closing of the transactions contemplated by the Royalty Pharma Agreement. The net gain from the transaction amounted to DKK 1,098.9 million, or $170.6 million, a portion of which was used to redeem outstanding notes we had issued in 2014 and related debt. In addition, we also remained eligible for a payment from Sanofi of potentially up to $15.0 million related to an offset from a settlement of an intellectual property dispute. However, we cannot be certain with regards to the timing and final amount of this payment, as both are dependent on factors that are outside of our control.
Under the terms of the Royalty Pharma Agreement, we remain required to perform certain of our obligations under the Sanofi License Agreement and to perform certain administrative obligations under the Royalty Pharma Agreement, which include that retain the underlying intellectual property and pay the relevant renewals thereon in a timely manner. A failure by us to comply with the terms of the Royalty Pharma Agreement or the Sanofi License Agreement may place us in breach of our contractual obligations with either party, expose us to liability for indemnification of either party and/or may result in arbitration and/or litigation against us that may lead, in the event of an adverse finding against us, to an award of significant damages, loss of profits and/or award of attorney fees against us. For a further discussion of the Royalty Pharma Agreement and the Sanofi License Agreement, see Note 2 'Revenue' and Note 7 'Other operating income' to the consolidated financial statements on pages 73-74 in our Annual Report 2019.
Alexion Collaboration
On March 20, 2019, we announced a collaboration with Alexion to discover and develop novel peptide therapies for complement-mediated diseases. The Collaborative Research and License Agreement, dated as of March 20, 2019, by and between Zealand Pharma A/S and Alexion Pharma Holding Unlimited Company, or the Alexion Agreement, provides Alexion with exclusive worldwide licenses for one preclinical target, with an option for up to three additional targets, in the complement pathway. We received an upfront payment of $25 million and an equity investment of $15 million (pursuant to Alexion's subscription for 802,859 of our ordinary shares), with potential for additional milestone-dependent and royalty payments. The accounting treatment of this upfront payment is outlined in Note 2 'Revenue' to the consolidated financial statements in our Annual Report 2019.
For the lead target, the Alexion Agreement provides the potential for development-related milestones of up to $115 million, as well as up to $495 million in sales-related milestones and the potential for royalty payments in the high single digits to low teens. Each of the three subsequent targets can be selected for an option fee of $15 million and has the potential for additional development-related milestones and sales-related milestones and royalty payments at a reduced price to the lead target.
As part of the equity investment from Alexion, our shares in ZP General Partner 3 ApS and ZP SPV 3 K/S were in January 2020 pledged to Alexion Pharma International Operation Unlimited
42
Company, or Alexion Pharma. Alexion Pharma is required to consent to any transfer of our shares in ZP General Partner 3 ApS and ZP SPV 3 K/S and Alexion Pharma also has a right to acquire such shares under certain conditions. Further, pursuant to the Alexion Agreement, in January 2020 we placed the relevant intellectual property to be developed under the Alexion Agreement in ZP SPV 3 K/S, our wholly owned subsidiary.
Encycle Acquisition
On October 22, 2019, we announced the acquisition of Encycle Therapeutics, Inc., or Encycle, pursuant to that certain Purchase Agreement, dated as of October 22, 2019, by and between the Zealand Pharma A/S, Encycle Therapeutics Inc. and certain other parties thereto, or the Encycle Agreement. The acquisition is determined to be an asset acquisition and is accounted for under IAS 38 'Intangible Assets'. The acquisition is centered on a pre-clinical lead asset that complements Zealand's focus on developing next-generation peptide therapeutics for gastrointestinal diseases. There were no upfront payments associated with the transaction. Future milestone payments could reach up to $80 million upon the completion of certain events, including $10 million upon the successful completion of a Phase 2 study. There is also a potential mid-single digit royalty on global net sales from the lead asset. The lead asset, ET3764, is being developed as an orally-delivered peptide drug to target integrin alpha-4-beta-7, which is involved in the pathogenesis of inflammatory bowel disease, or IBD. The target's mode of action has been clinically validated in IBD by vedolizumab, an approved, infusion-only alpha-4-beta-7 integrin inhibitor.
Bid for Valeritas Assets
On February 10, 2020, we announced a bid to acquire substantially all assets from Valeritas Holdings, Inc. (Nasdaq: VLRX), or Valeritas, for a total cash consideration of $23 million and the assumption of certain liabilities related to the ongoing business (including up to approximately $13.3 million related to open purchase orders, license payments and cure costs relating to prepetition contracts that will be assumed by Valeritas under the U.S. Bankruptcy Code upon exiting Chapter 11 proceedings), pursuant to the terms of the "stalking horse" asset purchase agreement entered into with Valeritas. On February 9, 2020, Valeritas and its subsidiaries filed voluntary petitions under Chapter 11 of the U.S. Bankruptcy Code in the U.S. Bankruptcy Court for the District of Delaware. At that time, we entered into a definitive agreement to acquire substantially all assets from Valeritas. Under the terms of the agreement, we serve as the stalking horse bidder in a sale process.
The proposed sale is to be conducted through a Court-supervised sale process under Section 363 of the Bankruptcy Code and will be subject to Court-approved bidding procedures and receipt of competing offers at auction. A hearing was held before the Bankruptcy Court on March 6, 2020, during which the Bankruptcy Court determined the procedures for the bidding and potential auction of Valeritas, which is expected to occur in March 2020. If our bid is selected, the sale will be subject to approval by the Bankruptcy Court and certain other closing conditions, including a condition relating to manufacturing performance to address supply disruptions experienced by Valeritas in December 2019. There can be no certainty that the transaction will be concluded.
Valeritas is a U.S.-based commercial-stage medical technology company focused on improving health and simplifying life for people with diabetes. Valeritas' product, the V-Go® Wearable Insulin Delivery device, is a simple, affordable, all-in-one basal-bolus insulin delivery device option for adult patients requiring insulin that is worn like a patch and can eliminate the need for taking multiple daily shots. Valeritas reported revenue of $22.4 million and loss before income taxes of $41.9 million for the nine months ended September 30, 2019. Valeritas reported in December 2019 that it experienced certain manufacturing performance issues that resulted in supply disruptions and are expected to have a material negative impact on its revenue and loss before income taxes. Although we expect that, if the Valeritas transaction is consummated, we may be able to reduce costs in the Valeritas business
43
associated with some aspects of how the business is run (for instance, rationalizing some of the sites and removing duplicative costs like insurance policies) relative to historical amounts, there can be no such assurance that any such cost reductions will be realized on a timely basis or at all. Even if such cost reductions are realized, the Company expects that the Valeritas acquisition will materially increase its operating loss and cash used in operations in the near term. The foregoing information concerning Valeritas Holdings, Inc. is taken from reports filed by such company with the SEC and has not been independently verified by us.
Reference is also made to '2019 Achievements' on page 8 in our Annual Report 2019 for a description of important events in 2019.
Capital expenditure
Capital expenditures primarily relate to building improvements and purchase of lab equipment at our headquarters in Søborg, Denmark. All capital expenditures are expected to be financed internally through cash on hand.
Public takeover offers in respect of the Company's shares
No such offers occurred during 2019 or 2020 to date.
B. BUSINESS OVERVIEW
We are a biotechnology company focused on the discovery, design and development of innovative peptide-based medicines. Our current pipeline of internal product candidates focus on specialty gastrointestinal and metabolic diseases where we believe that the present standard of care is inadequate. In addition, we are looking to focus our efforts on drug candidates that may qualify for orphan/rare disease status. We have the following programs in late clinical development:
- •
- Glepaglutide, a long-acting GLP-2 analog in development for the treatment of short bowel
syndrome, or SBS. Orphan drug designation has been granted in the U.S. We have published the results of a Phase 2 trial where glepaglutide
was dosed for three weeks in 18 patients with SBS. The trial demonstrates significant positive effects on gastrointestinal absorption and other efficacy parameters with the two highest doses, whilst
the lowest dose was non-effective. Based on the findings of this trial, the pivotal Phase 3 trial in 129 SBS patients was initiated in the fourth quarter of 2018 with patient enrollment
expected to be completed in 2020 and results from the trial expected in the first half of 2021. This study will evaluate the ability to reduce patient dependency on parenteral (intravenous) support
when treated with glepaglutide over 26 weeks. We believe glepaglutide may have the potential to offer patients with SBS a convenient once-weekly treatment alternative.
- •
- Dasiglucagon HypoPal® Rescue Pen for severe hypoglycemia. Ready-to-use dasiglucagon may offer diabetes patients and their families a fast treatment solution for severe hypoglycemia that is easier to use than currently marketed glucagon kits and offer a different mode of administration than the recently approved nasal administered glucagon powder Baqsimi®. Severe hypoglycemia is an acute, life-threatening condition resulting from a critical drop in blood glucose levels associated primarily with insulin therapy. Two Phase 3 efficacy trials with dasiglucagon for the treatment of severe hypoglycemia have been completed with good results in 2018 and 2019. A pediatric Phase 3 trial was initiated in the end of 2018, with positive results announced in September 2019. An NDA submission to the FDA is planned for early 2020.
44
- •
- Dasiglucagon dual-hormone artificial pancreas for automated diabetes
management. In a non-exclusive collaboration with Beta Bionics, we are developing dasiglucagon for use in an artificial pancreas device
containing both insulin and dasiglucagon. Breakthrough Device designation was received from the FDA in December 2019. Guided by an algorithm, this device is designed to maintain and control blood
glucose levels with minimal patient intervention. We have already reported positive results from two Phase 2a trials during the second quarter of 2017, and top-line results from a small
home-use Phase 2 trial in iLet™ gen 3.2 dual-hormone artificial pancreas system were announced in June 2019. We and Beta Bionics are in discussion with the FDA and expect to
initiate the pivotal Phase 3 trial in late 2020.
- •
- Dasiglucagon for congenital
hyperinsulinism. Congenital hyperinsulinism, or CHI, is an ultra-rare but devastating disease caused by inappropriately elevated insulin
secretion irrespective of glucose levels. This leads to frequent and often severe hypoglycemia and long-term irreversible damage to health. In 2017, the FDA and the European Commission both granted
orphan drug designation to dasiglucagon for the treatment of CHI. In January 2018, the FDA issued a safe-to-proceed letter, and the first Phase 3 trial with 32 pediatric patients (ages three
months to 12 years) with CHI started in the first quarter of 2019 with results expected in 2020. In May 2019, we enrolled the first children in the long-term Phase 3 extension study. In
December 2019, we initiated a second Phase 3 trial with 12 pediatric patients (ages seven days to one year) with CHI and we are currently awaiting patient recruitment. If results from these
studies are positive, we anticipate submitting an NDA to the FDA for treatment of CHI in 2021.
- •
- Dasiglucagon for post bariatric surgery hypoglycemia. In October 2019, we initiated a Phase 2 dose-finding clinical proof of concept trial to explore potential benefit of mini-doses of dasiglucagon in correcting serious hypoglycemic events following meal ingestions in some patients who have undergone bariatric surgery. The results of this trial are expected in the first half of 2020.
In addition to the late stage clinical programs, our portfolio includes ZP 7570, a potential once-weekly GLP-1-GLP-2 agonist for treatment of SBS in phase 1 development, two clinical license collaborations with Boehringer Ingelheim targeting treatment of type 2 diabetes and/or obesity, and a pre-clinical license collaboration with Alexion targeting Complement C3. The complement system is part of the immune system that protects the body against, among other things, bacterial infection. In certain diseases, this system can become dysregulated and lead to certain auto- immune diseases. Peptide-based therapeutics may offer an opportunity to treat some of these diseases. We also have a pipeline of other pre-clinical programs with the potential to enter into the clinic in 2020 and the years to come.
Furthermore, as we approach commercialization of certain of our product candidates, we are building a fully integrated commercial organization with U.S. operations to market our own therapies for rare diseases in the United States. During 2019, we took the decision to accelerate the development of our commercial organization to prepare to independently launch the Dasiglucagon HypoPal® Rescue Pen.
Our Product Pipeline
We operate within the global market for peptide-based medicines. All revenues are generated from milestone and royalty payments related to our license agreements. See Note 2 'Revenue' to the consolidated financial statements on pages 61-63 in our Annual Report 2019 for disclosure of revenue by category. We do not sell products to specific geographic markets.
45
The chart below summarizes the development stage and status of our portfolio of product candidates:
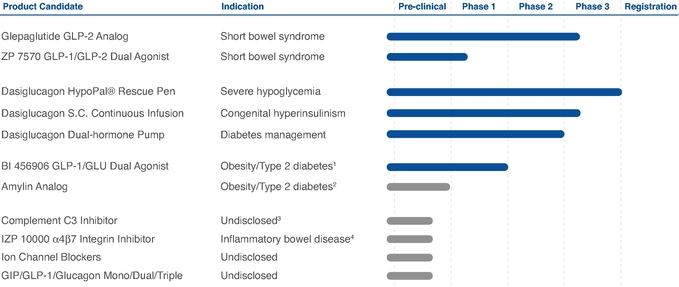
- (1)
- Partnered
with Boehringer Ingelheim. Zealand eligible for EUR 365 million in outstanding milestones. The initial EUR 20 million milestone
will become payable upon initiation of Phase 2 clinical testing of BI456906 GLP-1/GLU Dual Agonist. That Phase 2 clinical trial has not yet been initiated, but is expected to be
initiated, and as a result, the initial EUR 20 million milestone is expected to be received in 2020 rather than in late 2019 as previously anticipated.
- (2)
- Partnered
with Boehringer Ingelheim. Zealand eligible for EUR 283 million in outstanding milestones.
- (3)
- Partnered
with Alexion. Zealand eligible for USD 610 million in outstanding milestones.
- (4)
- Acquired Encycle Therapeutics, Inc. Future potential earn-outs of up to USD 80 million contingent on successful achievement of development, regulatory and commercial milestones; payable in cash and/or ZEAL equity at Zealand's discretion.
Overview of Short Bowel Syndrome (SBS)
SBS is a complex chronic and severe condition associated with reduced or complete loss of intestinal function. Many patients have to be connected to infusion lines and pumps every day, which pose significant restrictions on their ability to engage in daily activities. In addition, they are at risk of experiencing a number of serious and life-threatening complications such as sepsis, blood clots, liver damage and renal impairment.
GLP-2 treatment of SBS
In 2012 the FDA approved teduglutide as a novel treatment for the treatment of adult patients with SBS who are dependent on parenteral support. Teduglutide has a mean terminal half-life of approximately 1.3 hours in SBS subjects and has to be administered daily vial a syringe, following reconstitution of the lyophilized power. In 2018, Shire reported global sales of teduglutide of approximately $450 million.
46
Glepaglutide
Glepaglutide is a long-acting GLP-2 analogue with an effective half-life of approximately 50 hours with potential for once-weekly dosing in an auto-injector pen. In our pre-clinical studies, we observed that glepaglutide was effective in increasing intestinal weight and length. A Phase 2 clinical proof-of-concept, dose-finding Phase 2 trial was completed in 2017. Eighteen patients with SBS were enrolled in the trial and the primary result demonstrated that treatment with 1 mg and 10 mg glepaglutide reduced mean fecal output by 592 g/d (p=0.002) and 833 g/d (p=0.0002), respectively, and increased intestinal wet weight absorption. No changes were observed in the 0.1 mg dose group. Common adverse events were stoma complications, injection site reactions, peripheral edema, polyuria, nausea, and abdominal pain.
Key results from Phase 2 trial with glepaglutide in SBS are as follows:
Changes from baseline* (Adjusted means [95% CI]) |
0.1 mg (N=10) | 1 mg (N=11) | 10 mg (N=11) | |||
|---|---|---|---|---|---|---|
| Primary Endpoint | ||||||
Absolute Faecal Output (g/d) |
173 [–160, 506] p=0.274 |
–592 [–913, –272] p=0.002 |
–833 [–1152, –515] p=0.0002 |
|||
Secondary Endpoints |
||||||
Relative Faecal Output (%) |
10 [–3, 22] p=0.113 |
–23 [–35, –11] p=0.002 |
–30 [–42, –19] p=0.0002 |
|||
Absolute Urine Weight (g/d) |
90 [–208, 389] p=0.515 |
530 [245, 816] p=0.002 |
368 [82, 654] p=0.017 |
|||
Relative Urine Weight (%) |
11 [–11, 34] p=0.293 |
40 [18, 61] p=0.002 |
32 [11, 54] p=0.008 |
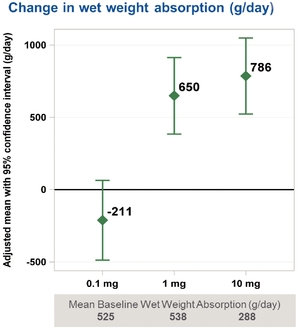
47
Common related adverse events as sorted by number of patients in each treatment period in the glepaglutide Phase 2 trial.
| |
0.1 mg N=12 | 1 mg N=12 | 10 mg N=12 | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
Any related adverse events |
10 | 11 | 11 | |||||||
Stoma complications |
3 | 6 | 10 | |||||||
Any injection site reaction |
2 | 7 | 8 | |||||||
Peripheral oedema |
2 | 6 | 5 | |||||||
Polyuria |
1 | 3 | 3 | |||||||
Abdominal pain |
5 | 1 | 1 | |||||||
Nausea |
4 | 0 | 3 | |||||||
Abdominal distension |
3 | 2 | 2 | |||||||
Fatigue |
1 | 1 | 3 | |||||||
Dizziness |
2 | 0 | 2 | |||||||
Vomiting |
3 | 0 | 1 | |||||||
Following the End-of-Phase 2 meeting with the FDA and scientific advice from the European Medicines Agency, we initiated the glepaglutide Phase 3 trial in patients with SBS in October 2018. The Phase 3 trial seeks to demonstrate efficacy and safety of once- and twice-weekly subcutaneous injections of 10 mg glepaglutide in SBS patients on parenteral support. We expect to enroll 129 patients at sites across the United States, Canada and Europe with enrollment expected to be completed in 2020. The trial will be placebo-controlled, randomized, parallel-group, double-blind, and with fixed dose injection. The primary objective will be to evaluate the efficacy of glepaglutide in reducing parenteral support volume in SBS patients. The secondary objectives will be to evaluate additional efficacy endpoints, as well as safety and tolerability. Results from the trial are expected in the first half of 2021.
Diabetes and severe hypoglycemia
All people with type 1 diabetes and most people with severely affected type 2 diabetes must constantly monitor and adjust their blood glucose levels to remain in proper glycemic control, as both high and low blood glucose may affect their health, both in the short and long term.
Severe hypoglycemia is an acute, life-threatening condition resulting from a critical drop in blood glucose levels associated primarily with insulin therapy. Severe hypoglycemic is one of the biggest concerns for insulin-dependent patients their families. It is a condition characterized by confusion, seizures and, often, loss of consciousness which, if left untreated, can result in death.
People require assistance from another person to treat severe hypoglycemia. Powder formulations of glucagon that require reconstitution immediately before administration, due to their poor drug stability, are available on the market. Studies have suggested that up to 85% of trained caregivers fail to give the full dose of glucagon when using these glucagon kits in a simulated emergency situation, due to complexity of use. Recently, Baqsimi®, a nasally-administered dry powder glucagon product, was approved by the FDA as an alternative treatment of severe hypoglycemia, which does not require mixing before administration. In addition to Baqsimi®, Xeris Pharmaceuticals Inc. has developed a ready use glucagon injection, Gvoke™, which is designed to be delivered in a pre-filled syringe, or PFS, or an auto-injector. Gvoke™ was approved by the FDA in September 2019.
Dasiglucagon HypoPal® PFS and rescue pen for severe hypoglycemia
Our dasiglucagon HypoPal® is a glucagon analog which is stable in liquid formulation. It is being developed in a PFS and auto-injector rescue pen presentations for treatment of severe hypoglycemia.
The HypoPal® rescue pen may offer diabetes patients and their families a fast treatment solution for severe hypoglycemia that is easier to use than currently marketed glucagon kits. Two Phase 3
48
efficacy trials with dasiglucagon for the treatment of severe hypoglycemia have been completed in 2018 and 2019. A pediatric Phase 3 trial was initiated at the end of 2018, with positive results announced in September 2019. The NDA submission to the FDA is planned for early 2020.
The pivotal Phase 3 trial demonstrated that a single dose of dasiglucagon administered via the PFS rapidly increased blood glucose levels in patients with type 1 diabetes following insulin-induced hypoglycemia. The trial compared the glycemic response observed after administration of dasiglucagon with that of placebo and that of currently marketed glucagon, in powder form for reconstitution prior to injection. The primary endpoint was time to plasma glucose recovery, which was defined as first increase in plasma glucose of at least 20 mg/dL (1.1 mmol/L) from baseline without administration of rescue intravenous glucose. The trial enrolled 168 total patients, including 82 in the dasiglucagon arm, 43 in the placebo arm and 43 in the GlucaGen® arm. GlucaGen® is a glucagon currently marketed by Novo Nordisk A/S.
- •
- The primary result demonstrates that the median time to blood glucose recovery was 10 minutes for dasiglucagon, which was superior to
placebo (median: 40 min; p<0.001). The median time to recovery for GlucaGen® was 12 minutes.
- •
- 99% of subjects were recovered from the insulin-induced hypoglycemia within 15 minutes following dosing with dasiglucagon, versus 2% with placebo and 95% with GlucaGen®.
Overall, no safety concerns were raised for dasiglucagon within the trial. Nausea and vomiting were reported with similar numbers for dasiglucagon and GlucaGen® (nausea: 55% and 53%, vomiting: 23% and 19%, respectively).
The confirmatory Phase 3 trial demonstrated that a single dose of dasiglucagon administered via the HypoPal® rescue pen rapidly increased blood glucose levels in patients with type 1 diabetes following insulin-induced hypoglycemia. The trial compared the glycemic response observed after dosing of dasiglucagon with that of placebo. The primary endpoint was time to plasma glucose recovery, which was defined as first increase in plasma glucose of ³20 mg/dL (1.1 mmol/L) from baseline without administration of rescue intravenous glucose. 45 subjects were included in the trial. The primary result demonstrated that the median time to blood glucose recovery was 10 minutes for dasiglucagon, which was superior to placebo (median: 35 min; p<0.001). The dasiglucagon pharmacokinetic profiles were consistent between the two phase 3 trials. Overall, no safety concerns were raised for dasiglucagon within the trial. Nausea and vomiting were reported with dasiglucagon (nausea: 62% and vomiting: 29%).
The pediatric Phase 3 trial compared the glycemic response observed after induction of hypoglycemia and administration of dasiglucagon (0.6 mg) with that of placebo and that of GlucaGen® (1 mg) in powder form for reconstitution prior to injection. The primary endpoint was time to plasma glucose recovery, which was defined as first increase in plasma glucose of at least 20 mg/dL (1.1 mmol/L) from baseline without administration of rescue intravenous glucose. The trial enrolled 42 pediatric patients (divided into age groups of 6 to 11 and 12 to 17 years old), including 21 in the dasiglucagon arm, 11 in the placebo arm and 10 in the GlucaGen® arm. The primary result demonstrated that the median time to blood glucose recovery was 10 minutes for dasiglucagon, which was superior to placebo (median: 30 min; p<0.001). The median time to recovery for GlucaGen® was 12 minutes. Overall, no safety concerns were raised for dasiglucagon within the trial. Nausea and vomiting were reported with dasiglucagon in both age groups (6 to 11 years; nausea: 25% and vomiting:
49
25%; 12 to 17 years; nausea: 92% and vomiting: 67%). For GlucaGen® (6 to 11 years; nausea: 50% and vomiting: 25%; 12 to 17 years; nausea: 17% and vomiting: 0%).
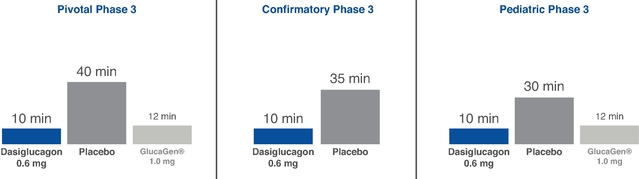
Dual-hormone artificial pancreas for automated diabetes management
Despite availability of modern insulins and improved insulin pump systems and glucose sensors, management of type 1 diabetes remains a significant patient burden and data suggest that only 20 to 30% of patients in the United States are below the glucose targets recommended by the American Diabetes Association. Maintaining tight glycemic control with infusion or injection with insulin is difficult due to delays in uptake and constant changes in insulin requirements. Therefore, several academic researchers and a few device companies are working on glucose-sensor guided dual-hormone pumps that injects insulin when glucose is high and glucagon with glucose is low, and thus holds the potential to remove the burden of marinating tight glucose control from the patients.
Dasiglucagon for dual-hormone artificial pancreas for automated diabetes management
Dasiglucagon is being developed in a 1ml pre-filled cartridge for use in dual-hormone artificial pancreas device systems, with insulin being the other hormone. We have already reported positive results from two Phase 2a trials during the second quarter of 2017. In our Phase 2a microdose trial, 17 patients with type 1 diabetes received four different doses of dasiglucagon, ranging from 0.03 mg to 0.6 mg, under euglycemia (normal blood glucose level) and hypoglycemia conditions. A dose-response with increases in blood glucose levels was observed across the dose range tested in this trial. In our Phase 2a pump trial, 10 adult patients with type 1 diabetes received dosing of dasiglucagon under challenging conditions, including fasting, a high basal insulin rate and exercise to stimulate the administration of glucagon by the iLet™ algorithms. No severe hypoglycemic episodes were observed, time below 60 mg/dl glucose was approximately 13% and 18% for dasiglucagon and recombinant glucagon, respectively, and time with 70 to 180 mg/dl glucose was approximately 71% and 65% for dasiglucagon and recombinant glucagon, respectively. Nausea and vomiting were reported in a similar number of patients for both dasiglucagon and the recombinant glucagon (nausea was reported in 55% and 53% of patients, respectively, and vomiting was reported in 23% and 19% of patients, respectively), but dasiglucagon was observed to be well-tolerated in the trial, with no injection site reactions noted. Furthermore, chronic toxicology studies support human testing of long-term usage of dasiglucagon, as dasiglucagon was observed to demonstrate similar physiological effects to native glucagon. Data from our Phase 2a trials and the toxicology studies provided the foundation for further clinical development of dasiglucagon in the iLet™ pump system.
We have a non-exclusive collaboration with Beta Bionics who is the developer of an artificial pancreas device containing both insulin and dasiglucagon, the iLet™. Breakthrough Device designation
50
was received from the FDA in December 2019. Guided by an algorithm, this device is designed to maintain and control blood glucose levels with minimal patient intervention.

Top-line results from a small home-use Phase 2 trial in iLet™ gen 3.2 dual-hormone artificial pancreas system were announced in June 2019. The trial compared operational performance of the iLet in the insulin-only configuration for one week versus the bihormonal configuration for one week in 10 adult participants with T1D. The iLet operated as expected, meeting the primary aim of the study. Preliminary data analysis demonstrated that the bihormonal iLet using dasiglucagon provided superior glycemic control over the insulin-only iLet (see table below).
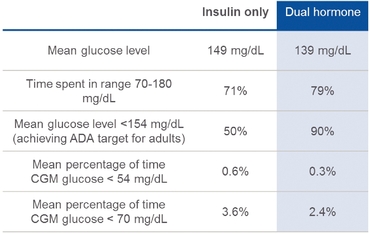
We and Beta Bionics are in discussion with the FDA and expect to initiate the pivotal Phase 3 trial in late 2020.
Congenital hyperinsulinism
CHI is an ultra-rare but devastating disease caused by inappropriately elevated insulin secretion irrespective of glucose levels. This leads to frequent and often severe hypoglycemia and long-term irreversible damage to health. Researchers have demonstrated the potential using chronic low-dose glucagon infusions to improve management of nine children with CHI. Reduction or discontinuation of I.V. glucose infusion was seen in all children. Six children were discharged to home during treatment. In five children, pancreatectomy or re-operation was avoided. In three children, glucagon was administered for one to four years leading to stable euglycemia.
Dasiglucagon for congenital hyperinsulinism
In 2017, the FDA and the European Commission both granted orphan drug designation to dasiglucagon for the treatment of CHI. In January 2018, the FDA issued a safe-to-proceed letter, and the first Phase 3 trial with 32 pediatric patients (ages three months to 12 years) with CHI started in the first quarter of 2019 with results expected in 2020. In December 2019, we initiated a second Phase 3 trial with 12 pediatric patients (ages seven days to one year) with CHI. If results from these studies are positive, we anticipate submitting an NDA to the FDA for treatment of CHI in 2021.
51
Post bariatric surgery hypoglycemia
Post-gastric bypass hypoglycemia, or PGBH, occurs following a meal and starting approximately 6 months or later following surgery for gastric bypass (called Roux-en-Y gastric bypass, or RYGB). Although it is rare, severe PGBH can be life-threatening with reports of associated seizure, syncope and motor vehicle accidents. The mechanisms underlying PGBH are not completely understood but may include inappropriate secretion of insulin and gut hormones, increased beta cell response to oral stimuli, increased glucose effectiveness, dysfunction of counter-regulatory hormones such as glucagon, and rapid post-weight loss improvement in insulin sensitivity.
Dasiglucagon for post bariatric surgery hypoglycemia
In October 2019, we initiated a Phase 2 dose-finding clinical proof of concept trial to explore potential benefit of mini-doses of dasiglucagon in correcting serious hypoglycemic events following meal ingestions in some patients who have undergone bariatric surgery. The results of this trial are expected in the first half of 2020.
Recent Developments
Reference is also made to the section 'Transforming peptides' on pages 12-25 in our Annual Report 2019.
Segment information
The Group is managed by a corporate management team reporting to the Chief Executive Officer. The corporate management team, including the Chief Executive Officer, represents the chief operating decision maker (CODM). No separate business areas or separate business units have been identified in connection with product candidates or geographical markets. As a consequence of this, no segment reporting is made concerning business areas or geographical areas.
Seasonality
The Company's financial performance, financial position and cash flows are not subject to significant seasonality.
Raw materials
A number of raw materials are used to produce our proprietary product candidates. The bulk of the raw materials are items that are also used by other pharmaceutical producers, so are generally not difficult for us to obtain. We are dependent only on suppliers of raw materials solely for use in the preclinical and clinical development stages of our product candidates. The raw materials have relatively low price volatility.
Market and competition
The pharmaceutical and biotechnology industries are characterized by intense competition and significant and rapid technological change as researchers learn more about diseases and develop new technologies and treatments. Significant competitive factors in our industry include: (i) product safety and efficacy; (ii) quality and breach of an organization's technology; (iii) skill of an organization's employees and its ability to recruit and retain key employees; (iv) timing and scope of regulatory approvals; (v) government reimbursement rates for, and the average settling price of, products; (vi) the availability of raw materials and qualified manufacturing capacity; (vii) manufacturing costs; (viii) intellectual property and patent rights and their protection; and (ix) sales and marketing capabilities. While we believe that our product and product candidate platform, development expertise
52
and scientific knowledge provide us with competitive advantages, we face potential competition from many different sources, including major pharmaceutical, specialty pharmaceutical and biotechnology companies, academic institutions and governmental agencies and public and private research institutions.
Any product candidates that we successfully develop and commercialize will compete with existing drugs and new drugs that may become available in the future.
We compete with companies that are producing drugs for, among other disease indications, SBS, such as Takeda plc which currently markets and distributes Gattex, and hypoglycemia, such as Novo Nordisk, Eli Lilly and Xeris Pharmaceuticals, Inc., which each market and distribute glucagon rescue kits.
Our competitors may also succeed in obtaining FDA, EMA or other regulatory approvals more rapidly than us, which could place us at a significant competitive disadvantage or deny us marketing exclusivity rights. Market acceptance of our product candidates will depend on a number of factors, including:
- •
- potential advantages over existing or alternative therapies or tests;
- •
- the actual or perceived safety of similar classes of products;
- •
- the effectiveness of our sales, marketing and distribution capabilities; and
- •
- the scope of any approval provided by the FDA, the EMA or other comparable regulatory authorities.
Although we believe our drugs and product candidates possess attractive attributes, we cannot ensure that our product candidates will achieve regulatory or market acceptance, or that we will be able to compete effectively in the market.
If our product candidates fail to gain regulatory approvals and acceptance in their intended markets, we may not generate meaningful revenue or achieve profitability.
In addition, many of our competitors have significantly greater financial resources and expertise in R&D, manufacturing, preclinical studies, conducting clinical trials, obtaining regulatory approvals and marketing drugs. Mergers and acquisitions in the pharmaceutical and biotechnology industries may result in even more resources being concentrated among a smaller number of competitors, particularly through partnership arrangements with large established companies. These companies also compete with us in recruiting and retaining qualified scientific and management personnel and establishing clinical trial sites and patient registration for clinical trials, as well as in acquiring technologies complementary to, or necessary for, our programs.
Patents
Patent Strategy
Our strategy for filing patent applications is to file early in the drug discovery process, typically before a lead compound has been selected. Before filing an initial patent application, we conduct searches of patents and publications based on keywords, patent classification codes and/or sequences to verify patentability of the compounds identified to date. A more focused, structure-based search is conducted once a lead compound is selected.
Patent applications are generally prepared by our in-house patent professionals in collaboration with outside patent counsel. Patent applications drafted before a lead compound is chosen typically disclose a large number of structurally related compounds. Our patent applications cover compositions of matter and methods of use, and may additionally cover dosing regimens and methods of making the
53
compounds. Later-filed patent applications typically cover next-generation compounds having for example, structural differences that might confer improved properties. Initially, we file one or more patent applications that establish priority to be claimed in later-filed applications. For most patent families, we file a patent application under the International Patent System, or PCT, which can be entered for examination into the patent office in any of the countries that are signatories to the PCT. In some cases, we file national applications directly in the major jurisdictions, which include Europe, the United States and Japan. For certain patent families, we file in parallel an application under the PCT and national applications in certain jurisdictions, such as the United States. Our patent strategy includes an evaluation of the of third party patents that may be infringed by our drug candidate products and development programs, and we prepare our development and commercialization plans to avoid claims of infringement. To the extent that we identify any potential issue with third party patents that may affect any of our product candidates, we develop a strategy to deal with such third party patents by ensuring that we are satisfied that such patents are invalid, not infringed, or that we commercialize our products after the expiry of such patents. Such strategies can include seeking a judicial or administrative revocation of such patents, ensuring that we are in a position to defend a claim for infringement, or seeking a license where that is appropriate.
We or our outside patent counsel handle the prosecution of our patent applications. If we enter into a licensing arrangement with a collaboration partner, we typically retain ultimate control of patent prosecution of patent applications for our inventions. For new inventions arising from collaboration under the license agreement, the collaboration partner may, depending on the identity of the inventors, file patent applications that are owned either by the collaboration partner alone or jointly with us.
Patent and Patent Application Portfolio
We own one patent family covering lixisenatide. This family includes approximately 60 granted patents and one patent application. This includes 59 granted non-U.S. patents in 10 non-U.S. jurisdictions and one issued U.S patent. This entire family is all licensed exclusively to Sanofi. We own five patent families covering two related proprietary GLP-2 analogs, glepaglutide and elsiglutide, or backup candidates. These patent families include 67 granted patents and 36 patent applications. Of these, 59 patents are non-U.S. patents in 16 non-U.S. jurisdictions. Although the disclosures of one of these patent families encompass both elsiglutide and glepaglutide, it has been possible to claim the subject matter relating to elsiglutide and glepaglutide in separate patents in the United States. For our internal compound dasiglucagon, a glucagon analog that has a favorable stability profile, we own three patent families including approximately 29 pending non-U.S. patent applications in 18 non-U.S. jurisdictions.
We also possess certain technologies we employ when designing novel peptide drug candidates. An example of one of our internal peptide enhancing technologies is the SIP technology. The SIP technology adds a number of specific amino acids to a peptide thereby strengthening or tightening the molecular structure to make the peptide less susceptible to biological degradation. The SIP technology can assist to maintain the peptide in the blood for a longer period of time before the peptide is degraded and may permit less frequent dosing of the peptide. The SIP technology has been employed for the development of lixisenatide, glepaglutide and elsiglutide. In addition to these we possess other proprietary technologies which involve the addition of a fatty acid to the amino acid chain of a given peptide as another technique to increase the half-life of the peptide in the blood stream.
Although specific reference is made to the status of patents granted or pending in the U.S. Patent and Trademark Office, or USPTO, the European Patent Office, or EPO, and Japan, in many cases the patent families also include patents or applications in a number of additional jurisdictions, including Australia, Canada, China, and India. Nominal expiration dates mentioned below are based on the statutory patent terms. Upon marketing approval, patent term extensions or supplementary protection certificates may be obtainable in various jurisdictions, including the United States, certain European
54
jurisdictions, and Japan, with respect to certain patents claiming compositions of matter, methods of use or methods of manufacturing the products, with a maximum of five years of extension potentially available. U.S. patents may also be entitled to adjustments to their statutory patent term depending on the length of the delay to the issuance of the patents caused by the USPTO.
We (or our wholly owned subsidiary, ZP Holding SPV K/S (a limited partnership), or ZP Holding, in the case of lixisenatide) own all patents and applications described below, as relating to lixisenatide, elsiglutide, glepaglutide and dasiglucagon.
Lixisenatide
Our wholly owned subsidiary, ZP Holding, owns one patent family covering the lixisenatide compound and compositions. This patent family includes granted patents in Europe (one European patent validated in 25 countries and a second European patent validated in 18 countries), the United States, Australia (two), Canada, China, Hong Kong (two), India, Israel (three), Japan (three), New Zealand and South Africa. The patents include composition of matter claims covering both the peptide and composition of lixisenatide, originally known as ZP10. The granted U.S. patent originally claimed lixisenatide and closely related analogs of lixisenatide, but was reissued with claims covering lixisenatide only. The closely related analogs may be pursued in one or more continuing applications. The granted European patents include claims drawn to the composition of matter of a genus of compounds that encompasses lixisenatide and of lixisenatide, compositions comprising lixisenatide, and uses of lixisenatide to treat various diseases, including type 2 diabetes, type 1 diabetes and obesity. The patents in this patent family have a nominal expiration date in July 2020 and some may be eligible for up to five years of regulatory patent term extension in several jurisdictions, including certain European jurisdictions, Japan, Israel and the United States. This entire family is all licensed exclusively to Sanofi.
Elsiglutide and glepaglutide
We own one patent family that covers both elsiglutide and glepaglutide, which includes 66 granted patents and 8 patent applications. This patent family discloses both the elsiglutide compound and the glepaglutide compound, which are structurally-related GLP-2 receptor agonists developed using our SIP technology. These two compounds therefore share one patent family in common.
This patent family includes granted patents in Australia, Canada, China (two), Eurasia (designated in nine countries), Hong Kong, Israel (three), India (two), Japan (three), South Korea (three), Mexico, New Zealand (three), Ukraine and South Africa (two). The granted U.S. patents include composition of matter claims covering both the peptide and composition of elsiglutide and glepaglutide, and claims drawn to a method of treating CID by administering elsiglutide and a method of treating inflammatory bowel disease by administering glepaglutide. The granted European patent includes claims drawn to the composition of matter of a genus of compounds that encompasses elsiglutide and glepaglutide and of elsiglutide and glepaglutide, as well as analogs thereof and methods of using elsiglutide and glepaglutide for the preparation of a medicament to treat or prevent side effects of chemotherapy, including diarrhea. Twelve applications are pending in this family in the United States and non-U.S. jurisdictions, including Brazil, Europe, Hong Kong, India, Japan, Norway and South Africa. The patents in this patent family have a nominal expiration date in May 2026, with the exception of one of the granted U.S. patents, which has an adjusted expiration date in June 2026.
The patents in this patent family were licensed to Helsinn for elsiglutide until June 2017. With the termination of the license agreement with Helsinn, we are now responsible for the maintenance of this patent family.
The granted European patent in this first patent family was opposed in the EPO by a competitor. The Opposition Division of the EPO maintained all original claims at a hearing in June 2014. The decision to maintain the patent with narrow claims covering glepaglutide and elsiglutide was made final at an oral hearing in November 2018 before the Technical Board of Appeals of the EPO. These proceedings are now closed.
55
In addition, glepaglutide has a total of five other patent families that are at an early stage of prosecution. These consist of pending patent applications in the United States and at the EPO.
Dasiglucagon
We own three patent families covering dasiglucagon, which include 25 granted non-U.S. patents and 21 patent applications in the United States and non-U.S. jurisdictions, including Brazil, Japan and India. The pending claims in one family cover the dasiglucagon compound and a group of structurally related compounds having glucagon agonist activity and increased solubility and/or stability relative to the native glucagon, as well as pharmaceutical compositions comprising such compounds and related uses for treating a variety of diseases including hypoglycemia, type 1 and 2 diabetes and other metabolic conditions, and nucleic acid molecules for expression of the compounds in host cells. The patent applications in the family that protect the compound itself, when issued, will have a nominal expiration date in July 2033. A United States patent in this family granted with claims covering the dasiglucagon compound. This patent received a patent term adjustment of 560 days, and is scheduled to expire in February 2035.
ZP 10,000 a4b7 Integrin Inhibitor
This is an asset that was acquired with our acquisition of Encycle. This includes the acquisition of six patent families consisting of 20 granted patents and 33 patent applications in various territories, including two patent families that are co-owned with the University of Montreal and two patent families that are licensed from the University of Toronto. The remaining two families are wholly owned by Encycle, including the family that includes the composition of matter patent application.
Ion Channel Blockers
We own one unpublished patent application in relation to this pre-clinical asset. This is at a very early stage of prosecution.
Government Regulation
The FDA and comparable regulatory authorities in state and local jurisdictions and in other countries impose substantial and burdensome requirements upon companies involved in the clinical development, manufacture, marketing and distribution of drugs, such as those we are developing. These agencies and other federal, state and local entities regulate, among other things, the R&D, testing, manufacture, quality control, safety, effectiveness, labeling, storage, record keeping, approval, advertising and promotion, distribution, post-approval monitoring and reporting, sampling and export and import of our product candidates.
U.S. Government Regulation
In the United States, the FDA regulates drugs under the Federal Food, Drug, and Cosmetic Act and its implementing regulations. The process of obtaining regulatory approvals and the subsequent compliance with applicable federal, state, local and foreign statutes and regulations requires the expenditure of substantial time and financial resources. Failure to comply with the applicable U.S. requirements at any time during the product development process, approval process or after approval, may subject an applicant to a variety of administrative or judicial sanctions, such as the FDA's refusal to approve pending NDAs, withdrawal of an approval, imposition of a clinical hold, issuance of warning letters, product recalls, product seizures, total or partial suspension of production or distribution, injunctions, fines, refusals of government contracts, restitution, disgorgement or civil or criminal penalties.
56
The process required by the FDA before a drug may be marketed in the United States generally involves the following:
- •
- completion of preclinical laboratory studies, animal studies and formulation studies in compliance with the FDA's good laboratory practice
regulations;
- •
- submission to the FDA of an IND, which must become effective before human clinical trials may begin;
- •
- approval by the institutional review board, or IRB, at each clinical site before each trial may be initiated;
- •
- performance of adequate and well-controlled human clinical trials in accordance with applicable IND and other clinical trial-related
regulations, sometimes referred to as good clinical practices to establish the safety and efficacy of the proposed product candidate for its proposed indication;
- •
- submission to the FDA of an NDA;
- •
- satisfactory completion of an FDA pre-approval inspection of the production facility or facilities where the product is produced to assess
compliance with the FDA's cGMP requirements to assure that the facilities, methods and controls are adequate to preserve the product's identity, strength, quality, purity and potency;
- •
- potential FDA audit of the preclinical and/or clinical trial sites that generated the data in support of the NDA; and
- •
- FDA review and approval of the NDA prior to any commercial marketing or sale of the product in the United States.
Preclinical Studies
Preclinical studies include laboratory evaluation of product chemistry, toxicity and formulation, as well as animal studies to assess potential safety and efficacy. An IND sponsor must submit the results of the preclinical studies, together with manufacturing information, analytical data and any available clinical data or literature, among other things, to the FDA as part of an IND. Some preclinical studies may continue even after the IND is submitted. An IND automatically becomes effective 30 days after receipt by the FDA, unless before that time the FDA raises concerns or questions related to one or more proposed clinical trials and places the clinical trial on a clinical hold. In such a case, the IND sponsor and the FDA must resolve any outstanding concerns before the clinical trial can begin. As a result, submission of an IND may not result in the FDA allowing clinical trials to commence.
Clinical Trials
Clinical trials involve the administration of the IND to human patients under the supervision of qualified investigators in accordance with GCP requirements, which include the requirement that all research patients provide their informed consent in writing for their participation in any clinical trial. Clinical trials are conducted under protocols detailing, among other things, the objectives of the trial, the parameters to be used in monitoring safety and the effectiveness criteria to be evaluated. A protocol for each clinical trial and any subsequent protocol amendments must be submitted to the FDA as part of the IND. In addition, an IRB at each institution participating in the clinical trial must review and approve the plan for any clinical trial before it commences at that institution. Information about certain clinical trials must be submitted within specific timeframes to the National Institutes of Health, or NIH, for public dissemination on their www.clinicaltrials.gov website.
57
Human clinical trials are typically conducted in three sequential phases, which may overlap or be combined:
- •
- Phase 1 clinical trial: the product candidate is initially introduced into healthy human patients or patients with the target disease or
condition and tested for safety, dosage tolerability, absorption, metabolism, distribution, excretion and, if possible, to gain an early indication of its effectiveness;
- •
- Phase 2 clinical trial: the product candidate is administered to a limited patient population to identify possible adverse effects and
safety risks, to preliminarily evaluate the efficacy of the product for specific targeted diseases and to determine dosage tolerability and optimal dosage; and
- •
- Phase 3 clinical trial: the product candidate is administered to an expanded patient population, generally at geographically dispersed clinical trial sites, in well-controlled clinical trials to generate enough data to statistically evaluate the efficacy and safety of the product for approval, to establish the overall risk-benefit profile of the product, and to provide adequate information for the labeling of the product.
Progress reports detailing the results of the clinical trials must be submitted at least annually to the FDA and more frequently if serious adverse events occur. Each of Phase 1, Phase 2 and Phase 3 clinical trials may not be completed successfully within any specified period, or at all. Furthermore, the FDA or the sponsor may suspend or terminate a clinical trial at any time on various grounds, including a finding that the research patients are being exposed to an unacceptable health risk. Similarly, an IRB can suspend or terminate approval of a clinical trial at its institution if the clinical trial is not being conducted in accordance with the IRB's requirements or if the drug has been associated with unexpected serious harm to patients.
Marketing Approval
Assuming successful completion of the required clinical testing, the results of the preclinical studies and clinical trials, together with detailed information relating to the product's chemistry, manufacture, controls and proposed labeling, among other things, are submitted to the FDA as part of an NDA requesting approval to market the product for one or more indications. In most cases, the submission of an NDA is subject to a substantial application user fee. Under the Prescription Drug User Fee Act, or PDUFA, guidelines that are currently in effect, the FDA has a goal of ten months from the date of "filing" of a standard NDA for a new molecular entity to review and act on the submission. This review typically takes twelve months from the date the NDA is submitted to the FDA because the FDA has approximately two months to make a "filing" decision.
In addition, under the Pediatric Research Equity Act of 2003, or PREA, as amended and reauthorized, certain NDAs or supplements to an NDA must contain data that are adequate to assess the safety and effectiveness of the drug for the claimed indications in all relevant pediatric subpopulations, and to support dosing and administration for each pediatric subpopulation for which the product is safe and effective. The FDA may, on its own initiative or at the request of the applicant, grant deferrals for submission of some or all pediatric data until after approval of the product for use in adults, or full or partial waivers from the pediatric data requirements.
The FDA also may require submission of a Risk Evaluation and Mitigation Strategies, or REMS, plan to ensure that the benefits of the drug outweigh its risks. The REMS plan could include medication guides, physician communication plans, assessment plans, or elements to assure safe use, such as restricted distribution methods, patient registries, or other risk minimization tools.
The FDA conducts a preliminary review of all NDAs within the first 60 days after submission, before accepting them for filing, to determine whether they are sufficiently complete to permit
58
substantive review. The FDA may request additional information rather than accept an NDA for filing. In this event, the application must be resubmitted with the additional information. The resubmitted application is also subject to review before the FDA accepts it for filing.
Once the submission is accepted for filing, the FDA begins an in-depth substantive review. The FDA reviews an NDA to determine, among other things, whether the drug is safe and effective and whether the facility in which it is manufactured, processed, packaged or held meets standards designed to assure the product's continued safety, quality and purity.
The FDA may refer an application for a novel drug to an advisory committee. An advisory committee is a panel of independent experts, including clinicians and other scientific experts, that reviews, evaluates and provides a recommendation as to whether the application should be approved and under what conditions. The FDA is not bound by the recommendations of an advisory committee, but it considers such recommendations carefully when making decisions.
Before approving an NDA, the FDA typically will inspect the facility or facilities where the product is manufactured. The FDA will not approve an application unless it determines that the manufacturing processes and facilities are in compliance with cGMP requirements and adequate to assure consistent production of the product within required specifications. Additionally, before approving an NDA, the FDA may inspect one or more clinical trial sites to assure compliance with GCP requirements.
After evaluating the NDA and all related information, including the advisory committee recommendation, if any, and inspection reports regarding the manufacturing facilities and clinical trial sites, the FDA may issue an approval letter, or, in some cases, a complete response letter. A complete response letter generally contains a statement of specific conditions that must be met in order to secure final approval of the NDA and may require additional clinical or preclinical testing in order for the FDA to reconsider the application. Even with submission of this additional information, the FDA ultimately may decide that the application does not satisfy the regulatory criteria for approval. If and when those conditions have been met to the FDA's satisfaction, the FDA will typically issue an approval letter. An approval letter authorizes commercial marketing of the drug with specific prescribing information for specific indications.
Even if the FDA approves a product, it may limit the approved indications for use of the product, require that contraindications, warnings or precautions be included in the product labeling, require that post-approval studies, including Phase 4 clinical trials, be conducted to further assess a drug's safety after approval, require testing and surveillance programs to monitor the product after commercialization, or impose other conditions, including distribution and use restrictions or other risk management mechanisms under a REMS, which can materially affect the potential market and profitability of the product. The FDA may prevent or limit further marketing of a product based on the results of post-marketing studies or surveillance programs. After approval, some types of changes to the approved product, such as adding new indications, manufacturing changes, and additional labeling claims, are subject to further testing requirements and FDA review and approval.
Post-Approval Requirements
Drugs manufactured or distributed pursuant to FDA approvals are subject to pervasive and continuing regulation by the FDA, including, among other things, requirements relating to recordkeeping, periodic reporting, product sampling and distribution, advertising and promotion and reporting of adverse experiences with the product. After approval, most changes to the approved product, such as adding new indications or other labeling claims are subject to prior FDA review and approval. There also are continuing, annual user fee requirements for any marketed products and the establishments at which such products are manufactured, as well as new application fees for supplemental applications with clinical data.
59
The FDA may impose a number of post-approval requirements as a condition of approval of an NDA. For example, the FDA may require post-marketing testing, including Phase 4 clinical trials, and surveillance to further assess and monitor the product's safety and effectiveness after commercialization. In addition, drug manufacturers and other entities involved in the manufacture and distribution of approved drugs are required to register their establishments with the FDA and state agencies, and are subject to periodic unannounced inspections by the FDA and these state agencies for compliance with cGMP requirements. Changes to the manufacturing process are strictly regulated and often require prior FDA approval before being implemented. FDA regulations also require investigation and correction of any deviations from cGMP requirements and impose reporting and documentation requirements upon the sponsor and any third party manufacturers that the sponsor may decide to use. Accordingly, manufacturers must continue to expend time, money, and effort in the area of production and quality control to maintain cGMP compliance.
Once an approval is granted, the FDA may withdraw the approval if compliance with regulatory requirements and standards is not maintained or if problems occur after the product reaches the market. Later discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with manufacturing processes, or failure to comply with regulatory requirements, may result in mandatory revisions to the approved labeling to add new safety information; imposition of post-market studies or clinical trials to assess new safety risks; or imposition of distribution or other restrictions under a REMS program. Other potential consequences include, among other things:
- •
- restrictions on the marketing or manufacturing of the product, complete withdrawal of the product from the market or product recalls;
- •
- fines, warning letters or holds on post-approval clinical trials;
- •
- refusal of the FDA to approve pending NDAs or supplements to approved NDAs, or suspension or revocation of product approvals;
- •
- product seizure or detention, or refusal to permit the import or export of products; or
- •
- injunctions or the imposition of civil or criminal penalties.
The FDA strictly regulates marketing, labeling, advertising and promotion of products that are placed on the market. Drugs may be promoted only for the approved indications and in accordance with the provisions of the approved label. The FDA and other agencies actively enforce the laws and regulations prohibiting the promotion of off-label uses, and a company that is found to have improperly promoted off-label uses may be subject to significant liability.
Combination Products
Certain of our product candidates are designed to be delivered to patients by dedicated medical devices. In the United States, products composed of components that would normally be regulated by different centers at the FDA are known as combination products. Typically, the FDA's Office of Combination Products assigns a combination product to a specific center as the lead reviewer based upon the product's primary mode of action. Depending on the type of combination product, its approval, clearance or licensure may usually be obtained through the submission of a single marketing application. However, the FDA sometimes will require separate marketing applications for individual constituent parts of the combination product which may require additional time, effort, and information, For example, delivery devices require Human Factors testing and their manufacture is subject to FDA's Quality System Regulation. Even when a single marketing application is required for a combination product, such as an NDA for a combination drug and delivery device, both the FDA's Center for Drug Evaluation and Research and the FDA's Center for Devices and Radiological Health may participate in the review.
60
Other U.S Healthcare Laws
Our current and future operations may directly, or indirectly through our prescribers, customers and purchasers, expose us to broadly applicable fraud and abuse and other healthcare laws and regulations that may constrain the business or financial arrangements and relationships through which we research, market, sell and distribute our products. Restrictions under applicable U.S. federal, state, local and non-U.S. healthcare laws and regulations include, but are not limited to, the following:
- •
- the federal Anti-Kickback Statute, which prohibits, among other things, persons or entities from knowingly and willfully soliciting, offering,
receiving or providing remuneration, including any kickback, bribe or rebate, directly or indirectly, in cash or in kind, to induce or reward, or in return for, either the referral of an individual
for, or the purchase or lease, order or recommendation of, any item, good, facility or service, for which payment may be made under federal healthcare programs such as Medicare and Medicaid;
- •
- the federal Beneficiary Inducement Statute, which prohibits giving anything of value to a government insurance beneficiary that could influence
the choice of provider or reimbursable covered product;
- •
- federal civil and criminal false claims laws, including the civil False Claims Act, and civil monetary penalties laws, which impose criminal
and civil penalties, including those from civil whistleblower or qui tam actions, against individuals or entities for, among other things, knowingly presenting, or causing to be presented, claims for
payment that are false or fraudulent or making a false statement to avoid, decrease or conceal an obligation to pay money to the federal government;
- •
- the anti-inducement law prohibits, among other things, the offering or giving of remuneration, which includes, without limitation, any transfer
of items or services for free or for less than fair market value (with limited exceptions), to a Medicare or Medicaid beneficiary that the person knows or should know is likely to influence the
beneficiary's selection of a particular supplier of items or services reimbursable by a federal or state governmental program;
- •
- the federal Health Insurance Portability and Accountability Act of 1996, or HIPAA, which created additional federal criminal statutes that
impose criminal and civil liability for, among other things, executing or attempting to execute a scheme to defraud any healthcare benefit program or knowingly and willingly falsifying, concealing or
covering up a material fact or making false statements relating to healthcare matters;
- •
- HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act and its implementing regulations, which impose
certain requirements on covered entities and their business associates, including mandatory contractual terms, with respect to safeguarding the privacy, security and transmission of individually
identifiable health information;
- •
- the federal transparency requirements under the Physician Payments Sunshine Act, enacted as part of the Patient Protection and ACA that require
applicable manufacturers of covered drugs, devices, biologics and medical supplies for which payment is available under Medicare, Medicaid, or the Children's Health Insurance Program, with specific
exceptions, to track and annually report to the Centers for Medicare & Medicaid Services, or CMMS, payments and other transfers of value provided to physicians, as defined by such law, and
teaching hospitals, and certain ownership and investment interests held by physicians or their immediate family members;
- •
- analogous state, local and non-U.S. laws and regulations, such as state anti-kickback and false claims laws, which may apply to items or services reimbursed by any third party payor, including commercial insurers; state, local and non-U.S. marketing and/or transparency laws applicable to
61
- •
- rules or legislation covering more or less the same subject matter are found in numerous other countries, including in Denmark, which sometimes result in lower or higher exposures in those countries than in the United States.
manufacturers that may be broader in scope than the federal requirements; state laws that require biopharmaceutical companies to comply with the biopharmaceutical industry's voluntary compliance guidelines, relevant compliance guidance promulgated by the federal government, implementation of compliance programs, and compliance with the state's code of conduct; state and local laws that require a pharmaceutical company's sales representatives to be registered or licensed by the state or local governmental entity; and state and non-U.S. laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and may be more stringent than HIPAA, thus complicating compliance efforts; and
Additionally, recent healthcare reform legislation has strengthened federal and state healthcare fraud and abuse laws. For example, the ACA amends the intent requirement of the federal Anti-Kickback Statute and criminal healthcare fraud statutes to clarify that liability under these statutes does not require a person or entity to have actual knowledge of the statutes or a specific intent to violate them. Moreover, the ACA provides that the government may assert that a claim that includes items or services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the civil False Claims Act. Because of the breadth of these laws and the narrowness of the statutory exceptions and safe harbors available, it is possible that some of our business activities could be subject to challenge under one or more of such laws.
Ensuring that our business arrangements with third parties comply with applicable healthcare laws and regulations will likely be costly. It is possible that governmental authorities will conclude that our business practices do not comply with current or future statutes, regulations or case law involving applicable fraud and abuse or other healthcare laws and regulations. If our operations were found to be in violation of any of these laws or any other governmental regulations that may apply to us, we may be subject to significant civil, criminal and administrative penalties, damages, fines, disgorgement, imprisonment, possible exclusion from government funded healthcare programs, such as Medicare and Medicaid, integrity obligations, contractual damages, reputational harm, diminished profits and future earnings, and curtailment of our operations, any of which could substantially disrupt our operations. If the physicians or other providers or entities with whom we expect to do business are found not to be in compliance with applicable laws, they may be subject to significant criminal, civil or administrative sanctions, including exclusion from government funded healthcare programs.
Healthcare Reform
Existing regulatory policies may change and additional government regulations may be enacted that could prevent, limit or delay regulatory approval of our product candidates. We cannot predict the likelihood, nature or extent of government regulation that may arise from future legislation or administrative action, either in the United States or abroad. For example, in the United States the current presidential administration and U.S. Congress have attempted to repeal or "repeal and replace" the Affordable Care Act, or the ACA. Although those efforts did not succeed, the presidential administration may continue to seek to modify, repeal, or otherwise invalidate all, or certain provisions of, the ACA. Additionally, there remain judicial and Congressional challenges to certain aspects of the ACA. For example, on December 14, 2018, a U.S. District Court Judge in the Northern District of Texas, or Texas District Court Judge, ruled that the entire ACA is invalid based primarily on the fact that the Tax Cuts and Jobs Act of 2017 repealed the tax-based shared responsibility payment imposed by the ACA, on certain individuals who fail to maintain qualifying health coverage for all or part of a year, which is commonly referred to as the "individual mandate". Additionally, on December 18, 2019, the U.S. Court of Appeals for the 5th Circuit upheld the District Court ruling that the individual
62
mandate was unconstitutional and remanded the case back to the District Court to determine whether the remaining provisions of the ACA are invalid as well. It is unclear how this decision, future decisions, and subsequent appeals will impact the law and the effect such impact could have on coverage and reimbursement for healthcare items and services covered by plans that were authorized by the ACA.
Other legislative changes have been proposed and adopted since the ACA was enacted. These changes included aggregate reductions to Medicare payments to providers of up to 2% per fiscal year, effective April 1, 2013, which, due to subsequent legislative amendments, will stay in effect through 2029 unless additional Congressional action is taken. In January 2013, President Obama signed into law the American Taxpayer Relief Act of 2012, which, among other things, reduced Medicare payments to several providers and increased the statute of limitations period for the U.S. government to recover overpayments to providers from three to five years. The Medicare Access and CHIP Reauthorization Act of 2015, or MACRA, ended the use of the statutory formula and established a quality payment program, also referred to as the Quality Payment Program. The quality payment program has two tracks, one known as the merit based incentive payment system for providers in the fee-for service Medicare program, and the advanced alternative payment model for providers in specific care models, such as accountable care organizations. In November 2019, CMS issued a final rule finalizing the changes to the Quality Payment Program. At this time it is unclear how the introduction of the Quality Payment Program will impact overall physician reimbursement under the Medicare program. These new laws may result in additional reductions in Medicare and other healthcare funding.
There have been several recent U.S. Congressional inquiries and proposed and enacted federal and state legislation designed to, among other things, bring more transparency to drug pricing, review the relationship between pricing and manufacturer patient programs, reduce the cost of drugs under Medicare, and reform government program reimbursement methodologies for drugs. In addition, the Trump administration's budget proposal for fiscal year 2021 includes a $135 billion allowance to support legislative proposals seeking to reduce drug prices, increase competition, lower out-of-pocket drug costs for patients, and increase patient access to lower-cost generic and biosimilar drugs. Further, the Trump administration previously released a "Blueprint" to lower drug prices and reduce out of pocket costs of drugs that contains additional proposals to increase drug manufacturer competition, increase the negotiating power of certain federal healthcare programs, incentivize manufacturers to lower the list price of their products, and reduce the out of pocket costs of drug products paid by consumers. The Department of Health and Human Services, or HHS, has solicited feedback on some of these measures and has implemented others under its existing authority. Congress and the Trump administration have each indicated that it will continue to seek new legislative and/or administrative measures to control drug costs. At the state level, legislatures are increasingly passing legislation and implementing regulations designed to control pharmaceutical and biological product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures, and, in some cases, such measures are designed to encourage importation from other countries and bulk purchasing.
Coverage and Reimbursement
Sales of certain of our out-licensed products and our product candidates, if and when approved for marketing, has and will depend, in part, on the extent to which our products will be covered by third party payors, such as government health care programs like Medicare and Medicaid, commercial insurance and managed healthcare organizations. These third party payors play an important role in determining the extent to which new drugs, biologics and medical devices will be covered. The Medicare and Medicaid programs increasingly are used as models for how private payors and other governmental payors develop their coverage and reimbursement policies for drugs, biologics and medical devices. It is difficult to predict at this time what third party payors will decide with respect to
63
the coverage and reimbursement for our product candidates. The primary trend in the U.S. healthcare industry and elsewhere has been cost containment, including price controls, restrictions on coverage and reimbursement and requirements for substitution of generic products and/or biosimilars. Adoption of price controls, cost containment measures and adoption of more restrictive policies in jurisdictions with existing controls and measures, could limit our net revenue and results.
Government authorities and third-party payors have attempted to control costs by limiting coverage and the amount of reimbursement for medical products, drugs and services. Increasingly, third-party payors are requiring that drug companies provide them with predetermined discounts from list prices and are challenging the prices charged for drugs. We cannot be sure that coverage will be available for any product candidate that we commercialize and, if coverage is available, the level of reimbursement. Because coverage and reimbursement determinations are made on a payor-by-payor basis, obtaining coverage and adequate reimbursement from a third party payor does not guarantee that we will obtain similar coverage or reimbursement from another third party payor. Reimbursement may impact the demand for, or the price of, any product candidate for which we obtain marketing approval. If reimbursement is not available or is available only to limited levels, we may not be able to successfully commercialize any product candidate for which we obtain marketing approval. Decreases in third party reimbursement for our product candidates or a decision by a third party payor not to cover our product candidates or provide only limited reimbursement for our product candidates could reduce physician usage of our products once approved and have a material adverse effect on our sales, results of operations and financial condition. Further, the adoption and implementation of any future governmental cost containment or other health reform initiative may result in additional downward pressure on the price that we may receive for any approved product.
Outside the United States, international operations are generally subject to extensive governmental price controls and other market regulations, and we believe the increasing emphasis on cost-containment initiatives in Europe, Canada, and other countries has and will continue to put pressure on the pricing and usage of our product candidates. In many countries, the prices of medical products are subject to varying price control mechanisms as part of national health systems. Other countries allow companies to fix their own prices for medical products, but monitor and control company profits. Additional foreign price controls or other changes in pricing regulation could restrict the amount that we are able to charge for our product candidates. Accordingly, in markets outside the United States, the reimbursement for our products may be reduced compared with the United States and may be insufficient to generate commercially reasonable revenue and profits.
C. ORGANIZATIONAL STRUCTURE
For information regarding our organizational structure, please see Exhibit 8.1 to this Annual Report on Form 20-F.
D. PROPERTY, PLANT AND EQUIPMENT
We lease of approximately 7,181 square meters of office and laboratory space at Sydmarken 11, 2860 Søborg, Denmark, where all our activities, including R&D, are currently conducted. The lease is interminable for 13 years for Zealand and 15 years for the landlord, Ejendomsselskabet Sydmarken 5 A/S, from the date when Zealand moved into the facilities, which was September 1, 2019. After said periods Zealand can, without restrictions, and the landlord, subject to certain restrictions under Danish law, terminate the lease upon 12 months written notice.
ITEM 4A UNRESOLVED STAFF COMMENTS
Not applicable.
64
ITEM 5 OPERATING AND FINANCIAL REVIEW AND PROSPECTS
Critical accounting estimates
Reference is made to Note 1 'Significant accounting policies, and significant accounting estimates and assessments' to the consolidated financial statements on pages 54-60 in our Annual Report 2019.
New accounting pronouncements
Reference is made to Note 1 'Significant accounting policies, and significant accounting estimates and assessments' to the consolidated financial statements on pages 54-60 in our Annual Report 2019.
A. OPERATING RESULTS
Reference is made to the section 'Financial review' contained on pages 40-42 in our Annual Report 2019 and "Item 3—Key Information—D. Risk Factors". Reference is further made to 'Risk management and internal control' on pages 38-39 in our Annual Report 2019.
The financial condition of the Group and its development is described in our Annual Report 2019. The information in this section is based on, and should be read in conjunction with, our Annual Report 2019. The analysis and discussion included in our Annual Report 2019 is primarily based on the consolidated financial statements, which are prepared in accordance with IFRS as issued by the IASB.
2019 compared with 2018
The section 'Financial review' contained on pages 40-42 in our Annual Report 2019 constitutes the Board of Directors' and Executive Management's discussion and analysis of results of operations.
2018 compared with 2017
The following financial review is based on the Company's consolidated financial information for the year ended December 31, 2018, as compared to the financial information for the year ended December 31, 2017. The below financial information has been restated. Please see Note 1 'Significant accounting policies, and significant accounting estimates and assessments' on pages 54-60 in our Annual Report 2019 for information regarding the restatement.
Revenue
Revenue in 2018 amounted to DKK 38.0 million, as compared to DKK 136.3 million in 2017.
Revenue from milestone payments amounted to DKK 13.1 million in 2018, as compared to DKK 101.0 in 2017, corresponding to an 87% decrease. The milestone payments in 2018 comprised a payment of DKK 9.8 million from an undisclosed counterpart in connection with a material transfer agreement and a payment of DKK 3.3 million from a license agreement with Protagonist Therapeutics Inc.
Total royalty revenue in 2018 amounted to DKK 24.9 million, as compared to DKK 35.3 million in 2017, a decrease of 29%. The decrease in 2018 is a consequence of the sale of future Sanofi royalties and milestones, the effect of which was that only royalties earned before June 30, 2018 were included in the 2018 income statement. Royalty revenue in 2018 from sales of Lyxumia®/ Adlyxin® amounted to DKK 7.1 million (as compared to DKK 16.7 in 2017) and from sales of Soliqua® 100/33 amounted to DKK 17.8 million (as compared to DKK 18.7 million in 2017).
65
Royalty expenses
Royalty expenses for 2018 amounted to DKK 3.4 million, as compared to DKK 14.2 million for 2017, and related to royalties paid to third parties on milestone payments received and royalty income relating to the license agreement with Sanofi.
Research and development expenses
R&D expenses amounted to DKK 438.2 million in 2018, as compared to DKK 323.9 million in 2017. The increase in R&D expenses for the year ended December 31, 2018 was primarily related to external costs of DKK 79.6 million from accelerated development activities. This 2018 figure comprises costs for three dasiglucagon programs, including the Phase 3 trials relating to the rescue pen for severe hypoglycemia, and clinical costs for dasiglucagon to be used in a dual-hormone artificial pancreas and to treat CHI. It also includes costs for initiating the Phase 3 trial with glepaglutide, as well as costs relating to pre-clinical activities.
The R&D share of personnel expenses for the year ended December 31, 2018 was DKK 153.6 million, as compared to DKK 118.6 million for the year ended December 31, 2017. The increase was mainly related to an increase in the number of employees in the clinical development organization.
Administrative expenses
Administrative expenses amounted to DKK 43.5 million in 2018, as compared to DKK 47.3 million in 2017. The decrease was due to a change in the composition of employees working in R&D and administration in comparison to 2017.
Other operating income
Other operating income amounted to DKK 1,099.5 million in 2018, as compared to DKK 0.6 million in 2017, and mainly consisted of the net effect from the Royalty Pharma Agreement to sell future royalty streams and $85 million of potential commercial milestones for Soliqua® 100/33/ Suliqua® and Lyxumia®/Adlyxin® to Royalty Pharma. Zealand received DKK 1,310.2 million, or $205 million, in September 2018 at closing of the transaction. Costs directly related to the transaction amounted to DKK 211.3 million and consisted of DKK 176.9 million paid to third parties plus other transaction costs of DKK 34.5 million.
Other operating income in 2018 in 2017 also consisted of government grants of DKK 0.6 million in each year.
Operating result
The operating result for 2018 was DKK 652.4 million, as compared to DKK –248.5 million in 2017.
Net financial items (financial income less financial expenses)
Net financial items amounted to DKK –27.3 million in 2018, as compared to DKK –31.4 million in 2017. The decrease was mainly due to positive exchange rate adjustments in 2018 compared to negative exchange rate adjustments in 2017 and decreased interest expenses, as Zealand redeemed a royalty bond in September 2018 in connection with the closing of the Royalty Pharma Agreement. Net financial items consist of interest income and expenses, amortized costs relating to the royalty bond financing, banking fees and exchange rate adjustments. DKK 15.1 million of the net financial items in 2018 (as compared to DKK 18.9 million in 2017) related to interest expense on the royalty bond, and DKK 18.3 million in 2018 (as compared to DKK 5.7 million in 2017) related to amortized costs of the
66
royalty bond financing. The increased amortized costs were a result of the repayment of the outstanding royalty bond in September 2018 as all remaining capitalized financing costs had been expensed.
Result before tax
Result before tax in 2018 was DKK 625.1 million, as compared to DKK –279.9 million in 2017.
Income tax
Income tax amounted to DKK –43.8 million in 2018, as compared to DKK 5.5 million in 2017. The income tax in 2018 was a consequence of the positive result before tax for the year stemming mainly from the net effect from our entry into the Royalty Pharma Agreement described above. No deferred tax asset was recognized in the statement of financial position in 2018 due to uncertainty as to when and whether tax losses could be utilized.
Net result and comprehensive result
The net result and comprehensive result for the year ended December 31, 2018 both amounted to DKK 581.3 million, as compared to DKK –274.4 million for the year ended December 31, 2017. The increased result in 2018 was mainly a consequence of an increase in other operating income as a result of the sale of future milestones and royalties relating to the Sanofi license having a net impact of DKK 1,098.9 million. This increase was partly offset by a decrease in revenue and an increase in research and development expenses.
Allocation of result
No dividend was proposed for 2018 or 2017. The net result for 2018 of DKK 581.3 million and the net result for 2017 of DKK –274.4 million was transferred to retained loss.
Segment information
The Group is managed by a Corporate Management Team reporting to the Chief Executive Officer. The Corporate Management Team, including the Chief Executive Officer, represents the chief operating decision maker (CODM). No separate business areas or separate business units have been identified in connection with product candidates or geographical markets. As a consequence of this, no segment reporting is made concerning business areas or geographical areas.
Inflation
Inflation for the fiscal years ended December 31, 2019, 2018 and 2017 has not had a material impact on the Group's revenue or net loss.
Foreign currencies
Reference is made to Note 26 'Financial risks—Exchange rate risk' to the consolidated financial statements on page 84 in our Annual Report 2019.
Governmental policies
Please refer to "Item 4—Information on the Company—Government Regulation".
67
B. LIQUIDITY AND CAPITAL RESOURCES
It is our aim to have an adequate capital structure in relation to the underlying operating results and research and development projects, so that we have sufficient capital to support operations and our long-term growth targets.
The Board of Directors finds that the current capital and share structure is appropriate for the shareholders and for the Company's present requirements.
As of December 31, 2019, our cash and cash equivalents were DKK 1,081.1 million, and we had marketable securities of DKK 299.5 million. We require cash to meet our operating expenses and capital expenditures. We have funded our cash requirements since our incorporation primarily with equity financing, milestone and royalty payments from our collaboration partners, and sales of future royalty streams.
The overall objectives and policies for our financial risk management are outlined in the Finance Policy, which is approved by the Board of Directors. For further information, reference is made to "Item 11—Qualitative and Quantitative Disclosures about Market Risks".
Financial resources
Reference is made to 'Consolidated statements of financial position' on page 51 and 'Consolidated statements of cash flows' on page 52 in our Annual Report 2019.
We believe our financial resources are sufficient to meet our requirements for at least the next 12 months.
Cash flow in 2019, 2018 and 2017
Reference is made to 'Consolidated statements of cash flows' on page 52 in our Annual Report 2019. The total cash flow for 2019, 2018 and 2017 amounted to DKK 213.4 million, DKK 266.1 million and DKK 280.5 million, respectively.
There are no material restrictions on the ability of subsidiaries with material cash amounts to transfer funds to the parent company Zealand Pharma A/S.
Cash outflow/inflow from operating activities
Cash flow from operating activities amounted to DKK –409.5 million, DKK –461.4 million and DKK –278.7 million in 2019, 2018 and 2017, respectively. Cash outflow from operating activities for 2019 was mainly due to the loss for the year adjusted for the upfront cash and equity investment from the Alexion agreement. Cash outflow from operating activities for 2018 was mainly a result of the net profit for the year adjusted for the net effect from the Royalty Pharma Agreement to sell future royalty streams and $85 million of potential commercial milestones for Soliqua® 100/33/ Suliqua® and Lyxumia®/Adlyxin® and for other non-cash items. Cash outflow from operating activities for 2017 was mainly a result of the net loss for the year adjusted for non-cash items.
Cash outflow/inflow from investing activities
Cash flow from investing activities amounted to DKK –51.7 million, DKK 882.9 million and DKK 221.4 million in 2019, 2018 and 2017, respectively. Cash outflow from investing activities in 2019 mainly comprised purchase of other investment and property, plant and equipment. Cash inflow from investing activities in 2018 mainly comprised the net effect from the Royalty Pharma Agreement described above. Cash inflow from investing activities in 2017 reflected milestone payments received from Sanofi during the year being transferred from restricted cash to cash and cash equivalents in conjunction with the repayment of a portion of the royalty bond.
68
Net investments in securities for 2018 amounted to DKK 225.6 million, as compared to DKK 75.0 for 2017. No additional investments in marketable securities were made in 2019. Zealand's marketable securities portfolio comprises listed short-term bonds in Danish kroner.
Cash flows related to other investments for 2019, 2018 and 2017 amounted to DKK 22.8 million, DKK 0.0 million and DKK 9.3 million, respectively. Zealand invested in Beta Bionics, Inc. in 2018, but payment related to such investment did not occur until 2019.
Investments in plant and equipment in 2019 and 2018 amounted to DKK 21.0 million and DKK 4.0 million, respectively mainly related to improvement of the new leased headquarters in Denmark paid in cash. Investments in plant and equipment for 2017 amounted to DKK 7.2 million, mainly related to new laboratory equipment.
Cash outflow/inflow from financing activities
Cash flow from financing activities amounted to DKK 674.5 million (–155.4), mainly related to capital contribution from Van Herk Investments, Alexion Pharmaceuticals and proceeds from employees exercising warrants. 2018 mainly related to the repayment of the royalty bond.
Cash flow from financing activities amounted to DKK 674.5 million in 2019, mainly related to capital contribution from Van Herk Investments, Alexion Pharmaceuticals and proceeds from employees exercising warrants. Cash flow from financing activities in 2018 amounted to DKK –155.4 million, related to the repayment of the royalty bond of DKK –158.3 million and proceeds from issuance of shares related to exercise of warrants of DKK 2.9 million. Cash flow from financing activities in 2017 amounted to DKK 337.9 million and included the net proceeds from the U.S. IPO of DKK 507.5 million, DKK –176.4 million related to repayment of the royalty bond and proceeds from issuance of shares related to exercise of warrants of DKK 6.8 million.
Debt financing
In connection with the closing of the Royalty Pharma Agreement in September 2018, we redeemed outstanding notes we had issued in 2014 and related debt. No long-term loans were outstanding as of December 31, 2019 or 2018. Reference is made to 'Consolidated statements of financial position' on page 51 and Note 22 'Royalty bond' to the consolidated financial statements on page 82 in our Annual Report 2019 for information on debt financing.
Financial instruments
Reference is made to Note 26 'Financial risks—Contractual maturity (liquidity risk)' to the consolidated financial statements on page 85 in our Annual Report 2019.
Commitments for capital expenditure etc.
Contractual obligations for capital expenditure and other contingent liabilities as of December 31, 2019 are shown in Note 25 'Contingent assets, liabilities and other contractual obligations' on page 83 to the consolidated financial statements in our Annual Report 2019.
The Executive Management of the Group believes that the obligations are covered by the Group's financial resources as well as expected future cash flows from operating activities.
C. RESEARCH AND DEVELOPMENT, PATENTS AND LICENSES, ETC.
We currently focus on gastrointestinal, metabolic and other specialty diseases where we believe that the present standard of care is inadequate and where we believe that we have the resources to advance our peptide-based product candidates into the later stages of clinical development, including
69
registration and, potentially, commercialization, while opportunistically considering partnership relationships that may arise. In addition, we are looking to focus our efforts on drug candidates that may qualify for orphan/rare disease status. Our R&D organization is structured to enable dynamic collaboration across various functions and project teams at each stage of discovery and development, allowing us to advance promising opportunities quickly and take advantage of our extensive knowledge of peptide design and product development. Our research and development expenses were DKK 561.4 million, DKK 438.2 million and DKK 323.9 million in 2019, 2018 and 2017, respectively. See Note 4 'Research, development and administration expenses' on pages 64-65 to the consolidated financial statements in our Annual Report 2019.
Information related to selected research and development projects can be found under 'Transforming peptides' on pages 12-25 in our Annual Report 2019.
Reference is made to "Item 3—Key Information—D. Risk Factors".
D. TREND INFORMATION
We do not currently produce, hold inventory or sell any products by ourselves or through partnerships.
Information about expectations for the financial year 2020 can be found on page 11 in the subsection '2020 Outlook and objectives' in our Annual Report 2019.
E. OFF-BALANCE SHEET ARRANGEMENTS
Reference is made to Note 25 'Contingent assets, liabilities and other contractual obligations' to the consolidated financial statements page 83 in our Annual Report 2019.
F. TABULAR DISCLOSURE OF CONTRACTUAL OBLIGATIONS
2019
DKK thousand
|
Within 1 year |
1 - 3 years | 3 - 5 years | More than 5 years |
Total | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Lease commitments (recognized in statement of financial position) |
7,692 | 11,621 | 11,738 | 54,709 | 85,760 | |||||||||||
Contractual obligations related to agreements with contract research organizations (not recognized in statement of financial position) |
230,162 | 88,784 | — | — | 318,946 | |||||||||||
| | | | | | | | | | | | | | | | | |
Total contractual obligations |
237,854 | 100,405 | 11,738 | 54,709 | 404,706 | |||||||||||
| | | | | | | | | | | | | | | | | |
As of January 1, 2019 leases have been recognized in the statement of financial position. Reference is made to Note 1 'Significant accounting policies, and significant accounting estimates and assessments' to the consolidated financial statements on pages 54-60 in our Annual Report 2019.
Reference is also made to Note 25 'Contingent assets, liabilities and other contractual obligations' to the consolidated financial statements on page 83 in our Annual Report 2019.
G. SAFE HARBOR
Forward-looking information discussed in this Item 5 includes assumptions, expectations, projections, intentions and beliefs about future events. These statements are intended as "forward-looking statements". We caution that assumptions, expectations, projections, intentions and beliefs about future events may and often do vary from actual results and the differences can be material.
70
Please see the section entitled "Forward-Looking Statements" at the beginning of this Annual Report on Form 20-F.
ITEM 6 DIRECTORS, EXECUTIVE MANAGEMENT AND EMPLOYEES
A. DIRECTORS AND EXECUTIVE MANAGEMENT
Reference is made to 'Board of Directors and Corporate Management' on pages 45-47 in our Annual Report 2019 for information about the members of our Board of Directors and Corporate Management, as well as their activities outside of the Company.
The Board of Directors has the overall responsibility for the affairs of the Company.
There are no family relationships between the Board of Directors and Corporate Management. No Director or member of Corporate Management have been elected according to an arrangement or understanding with shareholders, customers, suppliers or others.
As required by the Danish Companies Act, directors are elected at General Meetings by simple majority vote. In addition, three employee representatives are elected for four-year terms by the employees of Zealand Pharma A/S.
B. COMPENSATION
Reference is made to Note 6 'Information on staff and remuneration' to the consolidated financial statements on pages 65-73 in our Annual Report 2019.
C. BOARD PRACTICES
Reference is made to 'Corporate governance' on pages 32-34 in our Annual Report 2019 regarding board practices. The year of election for each member of the Board of Directors and the year of appointment for each member of Corporate Management is included in 'Board of Directors and Corporate Management' on pages 45-47 in our Annual Report 2019.
The terms of office of all the members of our Board of Directors elected by the general meeting expire at the next annual general meeting to be held in April 2020. All members of the Board of Directors elected by the general meeting are eligible for re-election. Employee elected board members are elected for a period of four years.
None of our directors has a service contract with us or any of our subsidiaries providing for benefits upon termination of employment.
D. EMPLOYEES
At December 31, 2019, 2018 and 2017 we had 179, 149 and 133 full-time employees, and during 2019, 2018 and 2017 we had an average of 173, 146 and 128 full-time employees. Labor unions currently representing our employees include HK it, medie & Industri Hovedstaden. We negotiate a collective agreement in good faith every three years, with the next negotiation scheduled to take place in 2020.
E. SHARE OWNERSHIP
For information on the Board of Directors' and Corporate Management's individual holdings of shares and warrants as of December 31, 2019 and change in these holdings during 2019, reference is made to the sections 'Board of Directors and Corporate Management' on pages 45-47, and Note 6 'Information on staff and remuneration' to the consolidated financial statements on pages 65-73 in our
71
Annual Report 2019. The members of our Board of Directors and Corporate Management in the aggregate hold 41,045 shares, representing less than 1% of the beneficial ownership of the Company.
In the period from January 1, 2020 until March 9, 2020, no shares were sold or purchased by the members of the Board of Directors or Executive Management. The internal rules on trading in our shares by members of the Board of Directors and Executive Management only permit trading in the four-week period commencing at the beginning of the third trading day following the date of public disclosure of interim financial reports or the annual report. Under special circumstances, the trading window may be derogated from.
We granted warrants to our executive management and selected employees of the company in 2005, 2007 and in each of the years between 2009 and 2019. Since our Annual General Meeting in 2012, it has been part of our remuneration policy that members of the Board of Directors are not permitted to participate in the warrant incentive program in their capacity as board members.
Warrants are, and have been, granted pursuant to shareholder authorizations provided to our Board of Directors under our Articles of Association. The detailed terms of the warrants, including the exact exercise price and the size of the grants, have been set our Board of Directors. Warrants are granted for employee services and will typically become exercisable between approximately one to five years after the date of grant and may be exercised in a pre-defined exercise period to subscribe for shares in a number of pre-defined exercise windows against payment of the exercise price. Unexercised warrants will lapse. Granted warrants are generally subject to provisions which allow for the forfeiture of unexercised warrants if the grantee separates from the company or one of our subsidiaries under circumstances in which the warrant holder is considered a "bad-leaver," understood as, for example, being dismissed for cause or resigning without us having materially breached the employment contract. Warrant holders may generally maintain all granted warrants if they separate from the company or one of our subsidiaries under circumstances where they are considered as "good-leavers," such as dismissal without cause, leaving us pursuant to an agreed severance agreement or retirement, warrant holder's resignation due to our material breach of contract or the warrant holder's death.
Reference is made to Note 6 'Information on staff and remuneration' to the consolidated financial statements on pages 65-73 in our Annual Report 2019.
ITEM 7 MAJOR SHAREHOLDERS AND RELATED PARTY TRANSACTIONS
A. MAJOR SHAREHOLDERS
As of November 2, 2010, we had one class of shares (prior to this date we had multiple classes of shares). As of March 9, 2020, our registered, issued and outstanding share capital was DKK 36,054,661 distributed into 36,054,661 shares of nominal value DKK 1 each.
Below is information as of March 9, 2020 with respect to (a) any shareholder who is known to the Company to be the owner of more than 5% of the Company's shares and (b) the total amount of any class owned by Zealand Pharma A/S and its affiliates (treasury shares) and by the Board of Directors and Executive Management as a group:
Identify of person or group
|
Shares owned |
Percent of class |
Percent of total votes |
|||||||
|---|---|---|---|---|---|---|---|---|---|---|
Van Herk Investments, Rotterdam, Netherlands |
6,943,329 | 19.3 | % | 19.3 | % | |||||
Wellington Management Company LLP, Boston, U.S. |
3,529,116 | 9.8 | % | 9.8 | % | |||||
Sunstone Capital A/S, Copenhagen, Denmark |
2,153,118 | 6.0 | % | 6.0 | % | |||||
Zealand Pharma A/S and subsidiaries (treasury shares) |
64,223 | 0.2 | % | 0.2 | % | |||||
Board of Directors and Executive Management |
41,045 | 0.1 | % | 0.1 | % | |||||
72
As of March 9, 2020, approximately 33.7% of our share capital was held in Denmark and approximately 19.7% of our share capital was held in North America. The total number of shareholders is more than 15,100 of whom more than 96.8% are estimated to be Danish residents and 0.6% are estimated to be resident in the United States.
Since August 9, 2017, which is the date that ADSs representing our ordinary shares were first listed on The Nasdaq Global Select Market, there has been no significant change in the percentage ownership held by any major shareholder except as described below:
- •
- As of September 5, 2019, the holdings of Van Herk Investments B.V. (Dutch registration no. 59055057) increased nominally from DKK 1,962,535 shares, corresponding to approximately 6.4% of the total share capital and total voting rights of Zealand Pharma A/S at such time, to DKK 6,714,730 shares, corresponding to approximately 18.8% of the total share capital and total voting rights of Zealand Pharma A/S at such time.
No shareholders of the Company have different voting rights from any other shareholder.
For further information reference is made to 'Shareholder information' on pages 43-44 in our Annual Report 2019.
B. RELATED PARTY TRANSACTIONS
We have no related parties with controlling interest.
Our other related parties comprise of the Company's Board of Directors and Corporate Management.
For further information reference is made to Note 27 'Related parties' to the consolidated financial statements on page 86 in our Annual Report 2019.
There have not been and there are no loans to members of the Board of Directors or Corporate Management in 2019, 2018 or 2017.
C. INTERESTS OF EXPERTS AND COUNSEL
Not applicable.
A. CONSOLIDATED STATEMENTS AND OTHER FINANCIAL INFORMATION
The consolidated financial statements required by this item accompany this Annual Report on Form 20-F in the form of our Annual Report 2019 (see Exhibit 15.1).
Legal proceedings
There are no material legal proceedings at December 31, 2019 or December 31, 2018.
Dividends
We have never declared or paid any cash dividends on our shares and we do not anticipate paying any cash dividends on our shares in the foreseeable future. We intend to retain all available funds and any future earnings to fund the development and expansion of our business. Any future determination related to our dividend policy and the declaration of any dividends will be made at the discretion of our Board of Directors and will depend on a number of factors, including our results of operations, financial condition, future prospects, contractual restrictions, restrictions imposed by applicable law and other factors our Board of Directors deems relevant.
73
In accordance with the Danish Companies Act ("DCA") ordinary dividends, if any, are declared with respect to a financial year at the annual general meeting of shareholders in the following year, where the statutory annual report (which includes the audited financial statements) for that financial year is approved. Further, our shareholders may resolve at a general meeting to distribute extraordinary dividends and our shareholders may also at a general meeting grant our Board of Directors an authorization to distribute extraordinary dividends. Any resolution to distribute extraordinary dividends within six months of the date of the statement of financial position as set out in our latest adopted annual report must be accompanied by the statement of financial position from our latest annual report or an interim statement of financial position which must be reviewed by our auditor. If the decision to distribute extraordinary dividends is passed more than six months after the date of the statement of financial position as set out in our latest adopted annual report, an interim statement of financial position must be prepared and reviewed by our auditor. The statement of financial position or the interim statement of financial position, as applicable, must show that sufficient funds are available for distribution. Our general meeting of shareholders cannot resolve to distribute dividends at an amount exceeding the amount recommended or approved by our Board of Directors. Moreover, ordinary dividends and extraordinary dividends may only be made out of distributable reserves and may not exceed what is considered sound and adequate with regard to our financial condition or be to the detriment of our creditors.
In accordance with the DCA, share buybacks, if any, may only be carried out by the board of directors using funds that could have been distributed as dividends at the latest annual general meeting of shareholders. Any share buyback must generally only be conducted in accordance with an authorization obtained at a general meeting of our shareholders. The authorization must be granted for a defined period of time not exceeding five years. In addition, the authorization must specify the maximum permitted value of treasury shares as well as the minimum and maximum amount that we may pay as consideration for such shares. A decision by our Board of Directors to engage in share buybacks, if any, will be made in accordance with the factors applicable to dividend payments set forth above.
For further information reference is made to 'Shareholder information', on pages 43-44 in our Annual Report 2019.
B. SIGNIFICANT CHANGES
On February 10, 2020, we announced a bid to acquire substantially all assets from Valeritas for a total cash consideration of $23 million and the assumption of certain liabilities related to the ongoing business (including up to approximately $13.3 million related to open purchase orders, license payments and cure costs relating to prepetition contracts that will be assumed by Valeritas under the U.S. Bankruptcy Code upon exiting Chapter 11 proceedings), pursuant to the terms of the "stalking horse" asset purchase agreement entered into with Valeritas.
No other significant events have occurred since December 31, 2019. Reference is made to Note 30 'Significant events after the balance sheet date' to the consolidated financial statements on page 86 in our Annual Report 2019.
A. OFFER AND LISTING DETAILS
The Company's shares are listed in Denmark on Nasdaq Copenhagen, and traded under the symbol "ZEAL." The Company's ADRs are traded on the Nasdaq Global Select Market under the symbol "ZEAL."
74
See Exhibit 2.1 to this Annual Report on Form 20-F for a description of the Company's ordinary shares. Reference is also made to 'Shareholder information' on pages 43-44 in our Annual Report 2019.
B. PLAN OF DISTRIBUTION
Not applicable.
C. MARKETS
The shares have been publicly traded since 2010 and have been listed on Nasdaq Copenhagen since that time.
ADSs representing the shares, as evidenced by American Depository Shares issued by The Bank of New York Mellon, as the Depository, have been listed on the Nasdaq Global Select Market since August 2017.
D. SELLING SHAREHOLDERS
Not applicable.
E. DILUTION
Not applicable.
F. EXPENSES OF THE ISSUE
Not applicable.
ITEM 10 ADDITIONAL INFORMATION
A. SHARE CAPITAL
Not applicable.
B. MEMORANDUM AND ARTICLES OF ASSOCIATION
This section summarizes certain material provisions of our Articles of Association, certain other constitutive documents and relevant Danish corporate law.
General
We are a public limited liability company organized under the laws of Denmark and registered with the Danish Business Authority under CVR number 20045078. We have been established with the objective of engaging in research, manufacture trade and related activities primarily within the pharmaceutical industry. Our objectives are set out in Article 2 of our Articles of Association.
Powers of the Board of Directors
Unless otherwise directed by the Board of Directors all members of the Board of Directors have equal voting rights, and all resolutions are passed by a simple majority of votes. In the event of a tie, the Chairman, and in his/her absence the Deputy Chairman, shall have the casting vote. The Board of Directors forms a quorum when at least a majority of its members is present.
According to the Danish Companies Act, no member of the Board of Directors or the Executive Management may take part in the consideration of any business involving agreements between any member of the Group and himself, legal actions brought against himself, or any business involving
75
agreements between any member of the Group and any third party or legal actions brought against any third party, if he has a major interest therein that might conflict with our interests.
The Danish Companies Act sets specific requirements for granting loans or providing security to any member of the Board of Directors and anyone particularly close to such a member of the Board of Directors
The remuneration of the Board of Directors must be approved by our shareholders at the Annual General Meeting.
Rights, restrictions and preferences attaching to the shares
No share carries any special rights. Each share confers the right to cast one vote at the general meeting of shareholders, unless the Articles of Association provide otherwise. Each holder of shares may cast as many votes as it holds shares. Voting instructions may be given only in respect of a number of ADSs representing an integral number of shares or other deposited securities. Shares that are held by the company or direct or indirect subsidiaries do not confer the right to vote.
ADS holders may only exercise voting rights with respect to the shares underlying their respective ADSs. In accordance with the provisions of the deposit agreement, which provides that a holder may vote the shares underlying any ADSs for any particular matter to be voted on by our shareholders either by withdrawing the shares underlying the ADSs or, to the extent permitted by applicable law and as permitted by the depositary, by requesting temporary registration as shareholder and authorizing the depositary to act as proxy. The depositary will try, as far as practical, to vote the shares underlying the ADSs as instructed by the ADS holders.
Changes to Shareholders' rights
Any change to the rights of the shareholders would require an amendment to the Articles of Association.
Where we elect to pay a dividend these are accrued to the company where they have not been claimed after 3 years.
General Meetings
General Meetings of the company are held in the greater Copenhagen area and must be held not later than four months from the closing of the financial year. Notice of the date of Annual General Meeting shall be sent to shareholders not later than eight weeks before the date of that meeting together with a date by which any shareholders wishing to have any specific item included on the agenda of the meeting should submit that item. Our general meetings of our shareholders are convened with a maximum notice of five weeks and a minimum notice of three weeks.
Ownership restrictions
There are no limitations on the rights of non-resident or foreign owners to hold or vote the shares imposed by the laws of Denmark, our Articles of Association, or any other of our constitutive documents.
Change of control
There is no provision in the Articles of Association, nor any other constituent document, that would have an effect of delaying, deferring or preventing a change in control of Zealand Pharma A/S and that would operate only with respect to a merger, acquisition or corporate restructuring involving the company (or any of its subsidiaries).
76
Ownership disclosure
Pursuant to chapter 7 of the Danish Capital Markets Act (Kapitalmarkedsloven), shareholders in a company incorporated in Denmark with its shares admitted to trading and official listing on a regulated market are required to immediately (normally within the same trading day as the triggering transaction), and simultaneously notify the company and the Danish Financial Supervisory Authority, when the shareholder's direct or indirect stake (i) represents 5% or more of the voting rights in the company or the nominal value of its share capital, and (ii) when a change in a holding already notified implies that the limits of 5%, 10%, 15%, 20%, 25%, 50% or 90% and the limits of one-third and two-thirds of the voting rights or the nominal value of its share capital are reached.
This duty to notify also applies to anyone, who directly or indirectly holds (a) financial instruments that afford the holder an unconditional right to purchase existing shares, such as share options and/or (b) financial instruments based on existing shares and with an economic effect equal to that of the financial instruments mentioned under (a), regardless of them not affording the right to purchase existing shares, such as ADSs or, depending on the circumstances, cash-settled derivatives linked to the value of our shares or ADSs. Holding these kinds of financial instruments counts towards the thresholds mentioned above and may thus trigger a duty to notify by themselves or when accumulated with a holding of shares or ADSs.
The notifications must comply with, among others, the requirements for the contents thereof set out in sections 15, 16 and 19 of the Danish executive order on major shareholders (Storaktionærbekendtgørelsen), including the identity of the shareholder and the date when a limit is reached or no longer reached. Failure to comply with the duties of disclosure is punishable by fine or suspension of voting rights in instances of gross or repeated non-compliance. The Danish Financial Supervisory Authority will in certain cases publish information concerning reprimands or sanctions imposed, including, as a general rule, the name of the shareholder in question, as a consequence of non-compliance with the above rules. When we receive a notification pursuant to chapter 7 of the Danish Capital Markets Act, we must publish its contents. Publication must occur after the receipt of the notification and no later than three weekdays thereafter.
Furthermore, the general duty of notification pursuant to the DCA applies, which implies that shareholders must notify the company when the limit of 100% of the voting rights or nominal value of the shares is reached or no longer reached. This also applies to holders of the ADSs.
Changes in capital
Our Articles of Association do not contain conditions governing changes in the capital more stringent than those contained in the Danish Companies Act.
C. MATERIAL CONTRACTS
We have not entered into any material contracts other than in the ordinary course of business, except for the Alexion Agreement and the Encycle Agreement, each as described in "Item 4—Information on the Company—Important events". See also Exhibit 4.1 and Exhibit 4.2, respectively, to this Annual Report on Form 20-F.
D. EXCHANGE CONTROLS
There are no governmental laws, decrees, or regulations in Denmark (including, but not limited to, foreign exchange controls) that restrict the export or import of capital, or that affect the remittance of dividends, interest or other payments to non-resident holders of the shares or the ADSs. There are no limitations on the right of non-resident or foreign owners to hold or vote the shares or the ADSs imposed by the laws of Denmark or our Articles of Association.
77
E. TAXATION
The following is a description of the material U.S. federal income tax consequences to the U.S. Holders described below of owning and disposing of the ADSs. It is not a comprehensive description of all tax considerations that may be relevant to a particular person's decision to acquire securities. This discussion applies only to a U.S. Holder that holds the ADSs as capital assets for tax purposes (generally, property held for investment). In addition, it does not describe all of the tax consequences that may be relevant in light of a U.S. Holder's particular circumstances, including foreign, state and local tax consequences, U.S. federal gift, estate and alternative minimum tax consequences, the potential application of the Medicare contribution tax, and tax consequences applicable to U.S. Holders subject to special rules, such as:
- •
- banks, insurance companies, and certain other financial institutions;
- •
- brokers, dealers or traders in securities who use a mark-to-market method of tax accounting;
- •
- persons holding the ADSs as part of a hedging transaction, "straddle," wash sale, conversion transaction or integrated transaction or persons
entering into a constructive sale with respect to the ADSs;
- •
- regulated investment companies;
- •
- real estate investment trusts, grantor trusts or other trusts;
- •
- persons whose "functional currency" for U.S. federal income tax purposes is not the U.S. dollar;
- •
- expatriates of the United States;
- •
- tax exempt entities, including "individual retirement accounts" and "Roth IRAs";
- •
- entities classified as partnerships or other pass-through entities for U.S. federal income tax purposes (and investors therein);
- •
- persons that own or are deemed to own shares (including shares represented by the ADSs) representing ten percent or more of our vote or value;
- •
- persons who acquired the ADSs in exchange for services or otherwise as compensation; and
- •
- persons holding the ADSs in connection with a trade or business conducted outside the United States.
If an entity that is classified as a partnership for U.S. federal income tax purposes holds the ADSs, the U.S. federal income tax treatment of a partner generally will depend on the status of the partner and the activities of the partnership. Partnerships holding the ADSs and partners in such partnerships are encouraged to consult their own tax advisers as to the particular U.S. federal income tax consequences of holding and disposing of the ADSs.
For U.S. federal income tax purposes, U.S. Holders of ADSs will be treated as the beneficial owners of the underlying shares represented by the ADSs and an exchange of ADSs for our shares generally will not be subject to U.S. federal income tax.
The discussion is based on the Code, administrative pronouncements, judicial decisions, and final, temporary and proposed U.S. Treasury Regulations, and the income tax treaty between Denmark and the United States, or the Treaty, all as of the date hereof, changes to any of which may affect the tax consequences described herein—possibly with retroactive effect.
A "U.S. Holder" is a holder who, for U.S. federal income tax purposes, is a beneficial owner of the ADSs who is eligible for the benefits of the Treaty and is:
- (1)
- an individual who is a citizen or resident of the United States;
78
- (2)
- a corporation, or other entity taxable as a corporation, created or organized in or under the laws of the United
States, any state therein or the District of Columbia;
- (3)
- an estate the income of which is subject to U.S. federal income tax regardless of its source; or
- (4)
- a trust, if (A) a U.S. court is able to exercise primary supervision over the trust's administration and one or more United States persons (as such term is defined under the Code) have authority to control all substantial decisions of the trust, or (B) the trust has a valid election in place under all applicable U.S. Treasury regulations to treat the trust as a United States person (as such term is defined under the Code).
U.S. Holders are encouraged to consult their own tax advisers concerning the U.S. federal, state, local and foreign tax consequences of owning and disposing of the ADSs in their particular circumstances.
Taxation of distributions
Subject to the Passive Foreign Investment Company, or PFIC, rules described below, distributions paid on the ADSs, other than certain pro rata distributions of the ADSs, generally will be treated as dividends to the extent paid out of our current or accumulated earnings and profits (as determined under U.S. federal income tax principles). Because we do not maintain calculations of our earnings and profits under U.S. federal income tax principles, we expect that distributions generally will be reported to U.S.
Holders as dividends. Subject to applicable limitations, dividends paid to certain non-corporate U.S. Holders may be taxable at preferential rates applicable to long-term capital gain. However, if we are a PFIC for the taxable year in which the dividend is paid or the preceding taxable year (see discussion below under "—Passive Foreign Investment Company rules"), the preferential rate will not apply. Each U.S. Holder is advised to consult its tax advisors regarding the availability of the reduced tax rate on dividends with regard to its particular circumstances.
The amount of a dividend will include any amounts withheld by us in respect of Danish taxes. The amount of the dividend will be treated as foreign-source dividend income to U.S. Holders and will not be eligible for the dividends-received deduction generally available to U.S. corporations under the Code. Dividends will be included in a U.S. Holder's income on the date of the U.S. Holder's receipt of the dividend. The amount of any dividend income paid in DKK will be the U.S. dollar amount calculated by reference to the exchange rate in effect on the date of actual or constructive receipt, regardless of whether the payment is in fact converted into U.S. dollars. If the dividend is converted into U.S. dollars on the date of receipt, a U.S. Holder should not be required to recognize foreign currency gain or loss in respect of the dividend income. A U.S. Holder may have foreign currency gain or loss if the dividend is converted into U.S. dollars after the date of receipt.
Subject to applicable limitations, some of which vary depending upon the U.S. Holder's particular circumstances, Danish taxes withheld from dividends on the ADSs (or shares underlying the ADSs) will be creditable against the U.S. Holder's U.S. federal income tax liability. The rules governing foreign tax credits are complex and U.S. Holders should consult their tax advisers regarding the creditability of foreign taxes in their particular circumstances. In lieu of claiming a foreign tax credit, U.S. Holders may, at their election, deduct foreign taxes, including any Danish tax, in computing their taxable income, subject to generally applicable limitations under U.S. law. An election to deduct foreign taxes instead of claiming foreign tax credits applies to all foreign taxes paid or accrued in the taxable year.
Sale or other taxable disposition of the ADSs
Subject to the PFIC rules described below, gain or loss realized on the sale or other taxable disposition of the ADSs will be capital gain or loss, and will be long-term capital gain or loss if the
79
U.S. Holder held the ADSs for more than one year. The amount of the gain or loss will equal the difference between the U.S. Holder's tax basis in the ADSs disposed of and the amount realized on the disposition, in each case as determined in U.S. dollars. This gain or loss generally will be U.S.-source gain or loss for foreign tax credit purposes. Capital gain from the sale, exchange or other disposition of ADSs by a non-corporate U.S. Holder generally is eligible for preferential rates of taxation if the non-corporate U.S. Holder's holding period for such ADSs determined at the time of such sale, exchange, or other taxable disposition exceeds one year (i.e., such gain is long-term capital gain). The deductibility of capital losses is subject to limitations.
If the consideration received by a U.S. Holder is not paid in U.S. dollars, the amount realized will be the U.S. dollar value of the payment received determined by reference to the spot rate of exchange on the date of the sale or other disposition. However, if the ADSs are treated as traded on an "established securities market" and a U.S. Holder is either a cash basis taxpayer or an accrual basis taxpayer that has made a special election (which must be applied consistently from year to year and cannot be changed without the consent of the IRS), such U.S. Holder will determine the U.S. dollar value of the amount realized in a non-U.S. dollar currency by translating the amount received at the spot rate of exchange on the settlement date of the sale. If a U.S. Holder is an accrual basis taxpayer that is not eligible to or does not elect to determine the amount realized using the spot rate on the settlement date, such U.S. Holder will recognize foreign currency gain or loss to the extent of any difference between the U.S. dollar amount realized on the date of sale or disposition and the U.S. dollar value of the currency received at the spot rate on the settlement date.
Passive Foreign Investment Company rules
Under the Code, we will be a PFIC for any taxable year in which, after the application of certain "look-through" rules with respect to subsidiaries, either (i) 75% or more of our gross income consists of "passive income," or (ii) 50% or more of the average quarterly value of our assets, including cash, consist of assets that produce, or are held for the production of, "passive income." Passive income generally includes interest, dividends, rents, certain non-active royalties and capital gains. Whether we will be a PFIC in any year depends on the composition of our income and assets, and the relative fair market value of our assets from time to time, which we expect may vary substantially over time.
Based on certain estimates of our gross income and gross assets, the latter determined by reference to the value of the ADSs and shares, we do not believe that we were a PFIC for the taxable year ending December 31, 2019. We have not yet made any determination as to our expected PFIC status for the current year and, accordingly, any such expectation would be subject to change based on, among other factors, our use of cash, the source and nature of our income, and the price of our ordinary shares or ADSs. No assurances can be provided with respect to our PFIC status for our taxable year ended December 31, 2019 or with regard to our PFIC status for the current year or any future taxable year. The determination of whether we are a PFIC is made annually for each of our taxable years. As a result, our PFIC status may change. In particular, for purposes of the asset test described above, the total value of our assets will be treated as equal to the sum of the aggregate value of the ADSs and shares plus the Company's liabilities. Therefore, for purposes of the asset test, the total value of our assets will depend on the market price of the ADSs. However, the value of our passive assets generally will be equal to the actual fair market value of such assets. A decrease in the market price of the ADSs would cause a decrease in the deemed total value of our assets for purposes of the asset test but generally would not cause a corresponding decrease in the actual value of our passive assets. Accordingly, fluctuations in the market price of the ADSs may result in us being a PFIC.
For purposes of the income test, we believe that we are engaged in an active trade or business of discovering and developing peptide drugs and that the royalties and milestone payments we receive from unrelated parties should be treated as derived in the active conduct of a trade or business and not characterized as passive income. However, we have no assurance that these anticipated milestone
80
payments and royalties will be paid when expected. If any such payments are delayed or not received then, depending on the amount of passive income we receive from other sources, the relative percentage of our income that is passive could increase and potentially cause us to be classified as a PFIC. There can be no assurances that we will not be classified as a PFIC for the current taxable year or for any future taxable year.
If we are a PFIC for any year during which a U.S. Holder holds the ADSs, we generally would continue to be treated as a PFIC with respect to that U.S. Holder for all succeeding years during which the U.S. Holder holds the ADSs, even if we ceased to meet the threshold requirements for PFIC status in any particular year unless (i) we cease to be a PFIC and the U.S. Holder has made a "deemed sale" election under the PFIC rules, (ii) we cease to be a PFIC and the U.S. Holder has a valid mark-to-market election in effect (as described below) or (iii) the U.S. Holder makes a Qualified Electing Fund Election, or QEF Election, with respect to all taxable years during which such U.S. Holders holding period in which we are a PFIC. However, a U.S. Holder may make a QEF Election with respect to our ADSs only if we annually provide such U.S. Holder with certain tax information, and we currently do not intend to prepare or provide such information. As a result, the QEF Election is not expected to be available to a U.S. Holder and the remainder of this discussion assumes that such election will not be available. If the "deemed sale" election is made, a U.S. Holder will be deemed to have sold the ADSs the U.S. Holder holds at their fair market value and any gain from such deemed sale would be subject to the rules described below. After the deemed sale election, so long as we do not become a PFIC in a subsequent taxable year, the U.S. Holder's ADSs with respect to which such election was made will not be treated as shares in a PFIC and the U.S. Holder will not be subject to the rules described below with respect to any "excess distribution" the U.S. Holder receives from us or any gain from an actual sale or other disposition of the ADSs. U.S. Holders should consult their tax advisors as to the possibility and consequences of making a deemed sale election if we cease to be a PFIC and such election becomes available.
If we are a PFIC for any taxable year during which a U.S. Holder holds the ADSs, the U.S. Holder may be subject to adverse tax consequences. Generally, gain recognized upon a disposition (including, under certain circumstances, a pledge) of the ADSs by the U.S. Holder would be allocated ratably over the U.S. Holder's holding period for such shares. The amounts allocated to the taxable year of disposition and to years before we became a PFIC would be taxed as ordinary income. The amount allocated to each other taxable year would be subject to tax at the highest rate in effect for that taxable year for individuals or corporations, as appropriate, and the interest charge generally applicable to underpayments of tax will be imposed on the resulting tax attributable to each such year. Further, to the extent that any distribution received by a U.S. Holder on its ADSs exceeds 125% of the average of the annual distributions on such ADSs received during the preceding three years or the U.S. Holder's holding period, whichever is shorter, that distribution would be subject to taxation in the same manner described immediately above with respect to gain on disposition.
Alternatively, if we are a PFIC and if the ADSs are "regularly traded" on a "qualified exchange," a U.S. Holder could make a mark-to-market election that would result in tax treatment different from the general tax treatment described in the preceding paragraph. The ADSs would be treated as "regularly traded" in any calendar year in which more than a de minimis quantity of the ADSs are traded on a qualified exchange on at least 15 days during each calendar quarter. Nasdaq is a qualified exchange for this purpose. If a U.S. Holder makes the mark-to-market election, the U.S. Holder generally will recognize as ordinary income any excess of the fair market value of the ADSs at the end of each taxable year over their adjusted tax basis, and will recognize an ordinary loss in respect of any excess of the adjusted tax basis of the ADSs over their fair market value at the end of the taxable year (but only to the extent of the net amount of income previously included as a result of the mark-to-market election). If a U.S. Holder makes the election, the U.S. Holder's tax basis in the ADSs will be adjusted to reflect these income or loss amounts. Any gain recognized on the sale or other
81
disposition of ADSs in a year when we are a PFIC will be treated as ordinary income and any loss will be treated as an ordinary loss (but only to the extent of the net amount of income previously included as a result of the mark-to-market election).
However, a mark-to-market election generally cannot be made for equity interests in any lower-tier PFICs that we own, unless shares of such lower-tier PFICs are themselves "regularly traded" on a "qualified exchange," as described above. As a result, even if a U.S. Holder validly makes a mark-to-market election with respect to our ADSs, the U.S. Holder may continue to be subject to the PFIC rules (described above) with respect to its indirect interest in any of our investments that are treated as an equity interest in a PFIC for U.S. federal income tax purposes. U.S. Holders should consult their tax advisors as to the availability and desirability of a mark-to-market election, as well as the impact of such election on interests in any lower-tier PFICs.
In addition, if we are a PFIC or, with respect to particular U.S. Holders, are treated as a PFIC for the taxable year in which we paid a dividend or for the prior taxable year, the preferential rates discussed above with respect to dividends paid to certain non-corporate U.S. Holders would not apply.
U.S. Holders should consult their tax advisers regarding whether we are or may become a PFIC and the potential application of the PFIC rules.
Information reporting and backup withholding
Payments of dividends and sales proceeds that are made within the United States or through certain U.S.-related financial intermediaries generally are subject to information reporting, and may be subject to backup withholding, unless (i) the U.S. Holder is a corporation or other exempt recipient or (ii) in the case of backup withholding, the U.S. Holder provides a correct taxpayer identification number and certifies that it is not subject to backup withholding.
Backup withholding is not an additional tax. The amount of any backup withholding from a payment to a U.S. Holder will be allowed as a credit against the holder's U.S. federal income tax liability and may entitle it to a refund, provided that the required information is timely furnished to the IRS.
If a U.S. Holder owns ADS during any year in which we are a PFIC, such U.S. Holder (including, potentially, indirect holders) will generally be required to file an IRS Form 8621 with such holder's federal income tax return for that year.
Certain U.S. Holders who are individuals may be required to report information relating to their ownership of an interest in certain foreign financial assets, including shares of a non-U.S. person, generally on Form 8938, subject to exceptions (including an exception for shares held through a U.S. financial institution). In addition, certain U.S. Holders may be required to file a FinCEN Form 114 (Report of Foreign Bank and Financial Accounts) with the U.S. Treasury Department each year to report their interests in the ADSs. U.S. Holders should consult their tax advisers regarding their reporting obligations with respect to the ADSs.
F. DIVIDENDS AND PAYING AGENTS
Not applicable.
G. STATEMENT BY EXPERTS
Not applicable.
H. DOCUMENTS ON DISPLAY
Documents referred to and filed with the SEC together with this Annual Report on Form 20-F can be read and copied at the SEC's public reference room located at 100 F Street, NE, Washington, DC 20549. Please call the United States Securities and Exchange Commission at 1-800-SEC-0330 for further information on the public reference rooms.
82
Copies of this Annual Report on Form 20-F as well as our Annual Report 2019 can be downloaded from the Investors page at zealandpharma.com. The contents of our website are not incorporated by reference into this Annual Report on Form 20-F. This Annual Report on Form 20-F is also filed electronically and can be viewed via EDGAR on www.sec.gov.
I. SUBSIDIARY INFORMATION
Not applicable.
ITEM 11 QUALITATIVE AND QUANTITATIVE DISCLOSURES ABOUT MARKET RISKS
Financial exposure and financial risk management
For a description and discussion of the Company's foreign exchange rate risk management, interest rate risk management, credit risk management and liquidity risk management, reference is made to Note 26 'Financial risks' to the consolidated financial statements on pages 84-85 in our Annual Report 2019 and 'Risk management and internal control' on pages 38-39 in our Annual Report 2019.
Sensitivity analysis
When conducting a sensitivity analysis, the Group assesses the change in fair value on the market-sensitive instruments following hypothetical changes in interest and exchange rates. The rates used to mark-to-market the instruments are market data as of December 31, 2019.
For information on interest rate and exchange rate sensitivity analysis in 2019, reference is made to Note 26 'Financial risks—Sensitivity analysis' to the consolidated financial statements on page 84 in our Annual Report 2019.
ITEM 12 DESCRIPTION OF SECURITIES OTHER THAN EQUITY SECURITIES
Not applicable.
Not applicable.
Not applicable.
ITEM 12D AMERICAN DEPOSITARY SHARES
Our ADS program is administrated by The Bank of New York Mellon. Each ADS represents one ordinary share (or a right to receive one ordinary share) deposited with the Copenhagen office of Danske Bank A/S, as custodian for the depositary in the United States. Each ADS also represents any other securities, cash or other property which may be held by the depositary. The depositary's office at which the ADSs are administered is located at 101 Barclay Street, New York, New York 10286. The Bank of New York Mellon's principal executive office is located at 225 Liberty Street, New York, New York 10286.
83
Pursuant to the terms of the deposit agreement, the holders of ADSs are required to pay the following fees:
Persons depositing or withdrawing ordinary shares or ADSs must pay:
|
For: | |
|---|---|---|
| $5.00 (or less) per 100 ADSs (or portion of 100 ADSs) | • Issue of ADSs, including issues resulting from a distribution of ordinary shares or rights or other property |
|
|
• Cancellation of ADSs for the purpose of withdrawal, including if the deposit agreement terminates |
|
$0.05 (or less) per ADS |
• Any cash distribution to you |
|
A fee equivalent to the fee that would be payable if securities distributed to you had been ordinary shares and the shares had been deposited for issue of ADSs |
• Distribution of securities distributed to holders of deposited securities which are distributed by the depositary to you |
|
$0.05 (or less) per ADS per calendar year |
• Depositary services |
|
Registration or transfer fees |
• Transfer and registration of ordinary shares on our share register to or from the name of the depositary or its agent when you deposit or withdraw shares |
|
Expenses of the depositary |
• Cable, telex and facsimile transmissions (when expressly provided in the deposit agreement) |
|
|
• Converting foreign currency to U.S. dollars |
|
Taxes and other governmental charges the depositary or the custodian have to pay on any ADS or share underlying an ADS, for example, share transfer taxes, stamp duty or withholding taxes |
• As necessary |
|
Any charges incurred by the depositary or its agents for servicing the deposited securities |
• As necessary |
The depositary collects its fees for delivery and surrender of ADSs directly from investors depositing ordinary shares or surrendering ADSs for the purpose of withdrawal or from intermediaries acting for them. The depositary collects fees for making distributions to investors by deducting those fees from the amounts distributed or by selling a portion of distributable property to pay the fees. The depositary may collect its annual fee for depositary services by deduction from cash distributions or by directly billing investors or by charging the book-entry system accounts of participants acting for them. The depositary may collect any of its fees by deduction from any cash distribution payable to ADS holders that are obligated to pay those fees. The depositary may generally refuse to provide for-fee services until its fees for those services are paid.
From time to time, the depositary may make payments to us to reimburse or share revenue from the fees collected from ADS holders, or waive fees and expenses for services provided, generally relating to costs and expenses arising out of establishment and maintenance of the ADS program. In performing its duties under the deposit agreement, the depositary may use brokers, dealers or other service providers that are affiliates of the depositary and that may earn or share fees or commissions.
84
The depositary may convert currency itself or through any of its affiliates and, in those cases, acts as principal for its own account and not as agent, advisor, broker or fiduciary on behalf of any other person and earns revenue, including, without limitation, transaction spreads, that it will retain for its own account. The revenue is based on, among other things, the difference between the exchange rate assigned to the currency conversion made under the deposit agreement and the rate that the depositary or its affiliate receives when buying or selling foreign currency for its own account. The depositary makes no representation that the exchange rate used or obtained in any currency conversion under the deposit agreement will be the most favorable rate that could be obtained at the time or that the method by which that rate will be determined will be the most favorable to ADS holders, subject to the depositary's obligations under the deposit agreement. The methodology used to determine exchange rates used in currency conversions is available upon request.
See Exhibit 2.1 to this Annual Report on Form 20-F for additional information on the ADSs.
ITEM 13 DEFAULTS, DIVIDEND ARREARAGES AND DELINQUENCIES
None.
ITEM 14 MATERIAL MODIFICATIONS TO THE RIGHTS OF SECURITY HOLDERS AND USE OF PROCEEDS
None.
ITEM 15 CONTROLS AND PROCEDURES
A. Disclosure Controls and Procedures
Our management, with the participation of our Chief Executive Officer and Chief Financial Officer, has evaluated the effectiveness of our disclosure controls and procedures (as defined in Rules 13a- 15(e) and 15d- 15(e) under the Securities Exchange Act of 1934, as amended), as of the end of the period covered by this Annual Report on Form 20-F. Based on such evaluation, our principal executive officer and principal financial officer have concluded that as of such date, our disclosure controls and procedures were not effective as of December 31, 2019 due to the material weaknesses described below.
B. Management's Annual Report on Internal Control over Financial Reporting
Our management is responsible for establishing and maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting.
Our internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and preparation of financial statements for external purposes in accordance with IFRS.
Internal control over financial reporting is defined in rules 13a-15(f) and 15d-15(f) under the Exchange Act as a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles, and includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the Company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the Company are being made
85
only in accordance with authorizations of management and directors of the Company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the Company's assets that could have a material effect on the audited consolidated financial statements.
Management, under the supervision and with the participation of our Chief Executive Officer and Chief Financial Officer, conducted an evaluation of the effectiveness of the internal control over financial reporting as of December 31, 2019, using the criteria set forth by the Committee of Sponsoring Organizations of the Treadway Commission in Internal Control—Integrated Framework (2013). Based on the evaluation performed, management concluded that material weaknesses existed as of December 31, 2019 as described below.
Material weaknesses
In connection with our financial statement preparation process for the years ended December 31, 2019, 2018, 2017, 2016 and 2015, we identified material weaknesses, including lack of sufficient competencies related to IFRS and SEC reporting knowledge for the purposes of timely and reliable financial reporting. Under the standards established by the PCAOB, a material weakness is a deficiency, or a combination of deficiencies, that creates a reasonable possibility that a material misstatement of a Company's annual financial statements will not be prevented or detected on a timely basis.
The material weaknesses we identified related to our existing processes to assess risks and to design and implement effective control activities over financial reporting. In particular, we did not have formalized risk assessment, oversight and compliance processes or formalized control descriptions for all of our key controls. During 2018 and 2019 these areas were developed and implemented, and we are working in 2020 to properly integrate them further into our routines and practices in the organization. Where control descriptions existed, they need to be further developed to ensure that they include all relevant information to enable the operating effectiveness of such controls. We will also work to ensure that adequate controls are performed in all areas. Where control activities were dependent on certain information, which is referred to as our IUC, we did not perform or document controls to assess the completeness and accuracy of such information. Internal capabilities have not been sufficient around the special and unusual transactions where we are relying on external assistance for proper handling of related risks. We have not adequately monitored control activities and identified control deficiencies; thus, we have been unable to evaluate whether other deficiencies, individually or in combination, resulted in a reasonable possibility that a material misstatement of our annual financial statements would not be prevented or detected on a timely basis. Further, restatements resulting from the correction of certain misstatements in our quarterly and annual financial statements have been identified, including in the first quarter of 2019 related to the recognition of warrants. These restatements occurred due to the lack of sufficient overall review of the financial statements and lack of oversight of the application and implementation of accounting policies and accounting standards by the Company.
We initiated several steps to begin to remediate the material weaknesses identified by us. In addition to engaging external subject matter experts in key areas to improve our competencies with respect to IFRS and SEC reporting, in late 2018 and early 2019 we implemented a formalized process for risk assessment, completed the design and documentation of our key controls to respond to the identified risks, and initiated implementation of a formalized process for performing our evaluation of the effectiveness of our internal controls over financial reporting. We hired a new Chief Executive Officer in April 2019 and a new Chief Financial Officer in October 2019. In 2019, we also continued to focus on our accounting and reporting functions by replacing and adding more capable resources and working towards improving competencies and skills. In 2020, we expect to focus on utilizing our information technology infrastructure to drive efficiency and effectiveness in processes and internal
86
controls through automation and standardization, while we continue to engage with external subject matter experts in key areas to improve our competencies with respect to IFRS and SEC reporting.
However, despite efforts taken to remediate the material weaknesses, the new accounting staff we onboarded in mid-2019 has not been operating under designed controls for a sufficient period to allow sufficient testing to demonstrate that such controls were operating effectively. Further, in testing of certain controls that was performed, management identified control deficiencies which in the aggregate constituted material weaknesses.
As such, management has not been able to obtain sufficient evidence that our implemented controls would be effective in remediating the material weaknesses previously identified. Accordingly, a reasonable possibility exists that a material misstatement of the Company's annual or interim financial statements will not be prevented or detected on a timely basis by the Company's internal controls. Management, including Chief Executive Officer and Chief Financial Officer, thus concluded that internal controls over financial reporting were not effective as of December 31, 2019.
Plan for remediation
As we continue to evaluate and work to improve internal control over financial reporting during 2020, management may determine to take additional measures to address control deficiencies. We have hired, and plan to continue to hire, additional finance and accounting personnel with appropriate expertise to perform specific functions and to further assist in the implementation of improved processes and internal controls, build our financial management and reporting infrastructure and further develop and document our accounting policies and financial reporting procedures, including ongoing senior management review and audit committee oversight.
While management believes that the steps that we have taken and plans we continue to take will improve our overall system of internal control over financial reporting and will remediate identified material weaknesses, the material weaknesses cannot be considered remediated until the applicable relevant controls operate for a sufficient period of time and management has concluded, through testing that these controls are operating effectively. No assurance can be provided at this time that the actions and remediation efforts will effectively remediate the material weaknesses described above or prevent the incidence of other material weaknesses in the our internal control over financial reporting in the future. Further, while our goal is to remediate the material weaknesses in 2020, we do not know the specific time frame needed to fully remediate the material weaknesses identified above. See "Item 3—Key Information—D. Risk Factors." Management, including the Chief Executive Officer and Chief Financial Officer, does not expect that disclosure controls and procedures or internal control over financial reporting will prevent all misstatements, even as the remediation measures are implemented and further improved to address the material weaknesses. The design of any system of internal controls is based in part upon certain assumptions about the likelihood of future events, and there can be no assurance that any design will succeed in achieving the stated goals under all potential future conditions.
C. Attestation Report of the Registered Public Accounting Firm
This Annual Report on Form 20-F does not include an attestation report of our independent registered public accounting firm regarding our internal control over financial reporting due to an exemption provided by the JOBS Act for emerging growth companies.
D. Changes in Internal Control over Financial Reporting
As noted above, management is undertaking steps to remediate the material weaknesses identified in 2019, 2018, 2017, 2016 and 2015 and reported in this Annual Report on Form 20-F, albeit such controls are not yet deemed to be operating effectively. There have been no other changes in our
87
internal control over financial reporting (as defined in Rule 13a-15(f) under the Exchange Act) that occurred during the period covered by this Annual Report that has materially affected, or is reasonably likely to materially affect, internal control over financial reporting.
ITEM 16A AUDIT COMMITTEE FINANCIAL EXPERTS
Our Audit Committee consists of Leonard Kruimer, Jeffrey Berkowitz and Martin Nicklasson, and is chaired by Leonard Kruimer. The Audit Committee reviews and considers matters relating to accounting, audit and regulatory control with our auditors and executive management in accordance with the working terms of reference of the Audit Committee. Our Audit Committee oversees our accounting and financial reporting processes and the audits of our consolidated financial statements.
Each member satisfies the independence requirements of the corporate governance standards of Nasdaq, and Leonard Kruimer qualifies as an "Audit Committee financial expert," as defined in Nasdaq Rule 5605(c)(2)(A) and as determined by our Board of Directors.
We have adopted a written code of business conduct, or code of conduct, which outlines the principles of legal and ethical business conduct under which we do business. The code of conduct applies to all of our board members and employees. This document is available under the "Corporate governance" tab in the "About Zealand" section of our website (www.zealandpharma.com). We undertake to provide to any person, without charge, upon request, a copy of the code of business conduct. Requests shall be made on mail info@zealandpharma.com.
ITEM 16C PRINCIPAL ACCOUNTANT FEES AND SERVICES
Reference is made to Note 5 'Fees to auditors appointed at the Annual General Meeting' to the consolidated financial statements on page 65 in our Annual Report 2019 regarding fees paid to our statutory auditors.
Audit fees
Audit fees consist of fees billed for professional services rendered by the principal accountant for the audit of the registrant's annual financial statements or services that are normally provided by the accountant in connection with statutory and regulatory filings or engagements for those fiscal years.
Audit-Related fees
Audit-Related fees consist of assurance and related services by the principal accountant that are reasonably related to the performance of the audit or review of the registrant's financial statements and are not reported under "Audit fees". Fees for audit-related services include consultations concerning financial accounting reporting standards and involvement as independent auditor in capital-related transactions.
Tax fees
Tax fees consist of fees billed for professional services rendered by the principal accountant for tax compliance, tax advice, and tax planning, including tax fees billed for tax consultations.
All other fees
All other fees consist of products and services provided by the principal accountant, other than the services reported in "Audit fees", "Audit-Related fees" and "Tax fees".
88
Fees for other services comprise fees billed for other permitted services such as assistance with cybersecurity awareness training.
Pre-approval policies
The Audit Committee assesses and pre-approves all audit and non-audit services provided by the statutory auditors. The pre-approval includes the type of service and a fee budget.
ITEM 16D EXEMPTIONS FROM THE LISTING STANDARDS FOR AUDIT COMMITTEES
Not applicable.
ITEM 16E PURCHASES OF EQUITY SECURITIES BY THE ISSUER AND AFFILIATED
Not applicable.
ITEM 16F CHANGE IN REGISTRANT'S CERTIFYING ACCOUNTANT
Not applicable.
As a foreign private issuer and as permitted by the listing requirements of Nasdaq, we rely on certain home country corporate governance practices rather than the corporate governance requirements of Nasdaq.
We qualify as a foreign private issuer and our ADSs are listed on Nasdaq. As a result, in accordance with the listing requirements of Nasdaq, we rely on home country governance requirements and certain exemptions thereunder rather than relying on the corporate governance requirements of Nasdaq. For example, we are exempt from certain rules under the Exchange Act that regulate disclosure obligations and procedural requirements related to the solicitation of proxies, consents or authorizations applicable to a security registered under the Exchange Act, including the U.S. proxy rules under Section 14 of the Exchange Act. In addition, our officers and directors are exempt from the reporting and "short-swing" profit recovery provisions of Section 16 of the Exchange Act and related rules with respect to their purchases and sales of our securities. Moreover, while we currently publish annual and quarterly reports on our website pursuant to the rules of Nasdaq Copenhagen and have filed such financial reports on an annual and quarterly basis with the SEC, we are not required to file such reports with the SEC as frequently or as promptly as U.S. public companies and are not required to file quarterly reports on Form 10-Q or current reports on Form 8-K that a domestic company would be required to file under the Exchange Act. Accordingly, there may be less publicly available information concerning our company than there would be if we were not a foreign private issuer.
In addition, the Listing Rules for the Nasdaq Stock Market (the "Nasdaq Listing Rules"), for domestic U.S. issuers require listed companies to have, among other things, a majority of their board members be independent, and to have independent director oversight of executive compensation, nomination of board members and corporate governance matters. While we currently comply, and intend to continue to comply, with these requirements, we are permitted to follow home country practice in lieu of the above requirements. Danish law does not require that a majority of our board consist of independent directors or the implementation of a nominating and corporate governance committee, and our board may thus in the future not include, or include fewer, independent directors than would be required if we were subject to the Nasdaq Listing Rules, or our board may decide that it is in our interest not to have a compensation committee or nominating and corporate governance committee, or have such committees governed by practices that would not comply with the Nasdaq Listing Rules. We follow home country practice with regard to, among other things, quorum
89
requirements generally applicable to general meetings of shareholders. Danish law only has a limited regulatory regime for the solicitation of proxies, thus our practice will vary from the requirement of Nasdaq Listing Rule 5620(b). In addition, our shareholders have authorized our board of directors to issue securities, including in connection with certain events such as the acquisition of shares or assets of another company, the establishment of or amendments to equity-based compensation plans for employees, rights issues at or below market price, certain private placements and directed issues at or above market price. To this extent, our practice varies from the requirements of Nasdaq Listing Rule 5635, which generally requires an issuer to obtain shareholder approval for the issuance of securities in connection with such events.
ITEM 16H MINE SAFETY DISCLOSURE
Not applicable.
90
See response to Item 18.
The financial statements required by this item accompany this Annual Report on Form 20-F in the form of our Annual Report 2019 on pages 48-86 (see Item 19 of this Annual Report on Form 20-F).
Reconciliation of non-IFRS financial measures
In our Annual Report 2019, Zealand discloses non-IFRS financial measures of the Group that reflect adjustments to the most directly comparable measure calculated and presented in accordance with IFRS. The inclusion of non-IFRS measures has been expressly permitted by the Danish Business Authorities and thereby exempted from the prohibition in Item 10(e)(1)(ii)(C) of Regulation S-K. However, the non-IFRS financial measures may not be defined and calculated by other companies in the same manner and may not be comparable with such measure.
Reference is made to the section 'Alternative performance measures for the Group (non-audited)' on page 100 in our Annual Report 2019.
a. Annual Report
The following pages from our Annual Report 2019, furnished to the SEC on Form 6-K, dated March 13, 2020, are incorporated by reference into this Annual Report on Form 20-F. The content of websites and other sources referenced on these pages are not incorporated by reference into this Annual Report on Form 20-F.
Page(s) incorporated by reference from our Annual Report 2019
Management review |
||||
2019 Achievements |
8 | |||
2020 Outlook and objectives |
11 | |||
Transforming peptides |
12 - 25 | |||
Corporate governance |
32 - 34 | |||
Risk management and internal control |
38 - 39 | |||
Financial review |
40 - 42 | |||
Shareholder information |
43 - 44 | |||
Board of directors and corporate management |
45 - 47 | |||
Consolidated Financial Statements |
||||
Consolidated Income statements |
50 | |||
Consolidated statements of comprehensive income |
50 | |||
Consolidated statements of financial position |
51 | |||
Consolidated statements of cash flows |
52 | |||
Consolidated statements of changes in equity |
52 | |||
Notes to the Consolidated financial statements |
54 - 86 | |||
Statement of the Board of Directors and Executive Management |
101 |
91
b. Exhibits
List of exhibits:
| Exhibit No. | Description | Method of filing | |||
|---|---|---|---|---|---|
| 1.1 | Articles of Association | Incorporated by reference to the Registrant's Report furnished to the SEC on Form 6-K on April 26, 2019. | |||
2.1 |
Description of Securities registered under Section 12 of the Securities Act of 1933, as amended |
Filed together with this Form 20-F 2019. |
|||
2.2 |
Deposit Agreement |
Incorporated by reference to Exhibit 4.1 to the Registrant's Registration Statement on Form F-1/A filed with the SEC August 3, 2017. |
|||
4.1 |
Collaborative Research and License Agreement, dated as of March 20, 2019, by and between the Registrant and Alexion Pharma Holding Unlimited Company† |
Filed together with this Form 20-F 2019. |
|||
4.2 |
Purchase Agreement, dated as of October 22, 2019, by and between the Registrant, Encycle Therapeutics Inc. and certain other parties thereto† |
Filed together with this Form 20-F 2019. |
|||
8.1 |
List of subsidiaries |
Filed together with this Form 20-F 2019. |
|||
12.1 |
Certification of the Principal Executive Officer. |
Filed together with this Form 20-F 2019. |
|||
12.2 |
Certification of the Financial Officer |
Filed together with this Form 20-F 2019. |
|||
13.1 |
Principal Executive Officer certification pursuant to 18 U.S.C. section 1350 |
Furnished together with this Form 20-F 2019. |
|||
13.2 |
Principal Financial Officer certification pursuant to 18 U.S.C. section 1350 |
Furnished together with this Form 20-F 2019. |
|||
15.1 |
Extracts from the Registrant's Annual Report 2019 |
Incorporated by reference to our Annual Report 2019, furnished to the SEC on Form 6-K dated March 13, 2020, as exhibit 99.1(a) identified in Item 19.a of this Form 20-F. |
|||
15.2 |
Consent of independent registered public accounting firm |
Filed together with this Form 20-F 2019. |
|||
EX-101.INS |
XBRL Instance Document |
Incorporated by reference to the Registrant's report furnished to the SEC on Form 6-K on March 13, 2020 as exhibit 99.1(a). |
|||
92
| Exhibit No. | Description | Method of filing | |||
|---|---|---|---|---|---|
| EX-101.SCH | XBRL Taxonomy Extension Schema Document | Incorporated by reference to the Registrant's report furnished to the SEC on Form 6-K on March 13, 2020 as exhibit 99.1(a). | |||
EX-101.CAL |
XBRL Taxonomy Extension Calculation Linkbase Document |
Incorporated by reference to the Registrant's report furnished to the SEC on Form 6-K on March 13, 2020 as exhibit 99.1(a). |
|||
EX-101.DEF |
XBRL Taxonomy Extension Definition Linkbase Document |
Incorporated by reference to the Registrant's report furnished to the SEC on Form 6-K on March 13, 2020 as exhibit 99.1(a). |
|||
EX-101.IAB |
XBRL Taxonomy Extension Labels Linkbase Document |
Incorporated by reference to the Registrant's report furnished to the SEC on Form 6-K on March 13, 2020 as exhibit 99.1(a). |
|||
EX-101.PRE |
XBRL Taxonomy Extension Presentation Linkbase Document |
Incorporated by reference to the Registrant's report furnished to the SEC on Form 6-K on March 13, 2020 as exhibit 99.1(a). |
|||
- †
- Portions of this exhibit have been omitted as permitted by Form 20-F.
93
The Registrant hereby certifies that it meets all of the requirements for filing on Form 20-F and that it has duly caused and authorized the undersigned to sign this Annual Report on its behalf.
| Zealand Pharma A/S | ||||||
/s/ EMMANUEL DULAC |
/s/ MATTHEW DALLAS |
|||||
| Name: | Emmanuel Dulac | Name: | Matthew Dallas | |||
| Title: | President and Chief Executive Officer | Title: | Senior Vice President and Chief Financial Officer | |||
Denmark
Dated: March 13, 2020
94
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the shareholders and the Board of Directors of Zealand Pharma A/S
Opinion on the Financial Statements
We have audited the consolidated statements of financial position of Zealand Pharma A/S and subsidiaries (the "Company") as of December 31, 2019 and 2018, the related consolidated income statements and consolidated statements of comprehensive income (loss), changes in equity and cash flow for each of the three years in the period ended December 31, 2019, and the related notes (collectively referred to as the "financial statements") included in Exhibit 15.1 on pages 50-86. In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2019 and 2018, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2019, in conformity with International Financial Reporting Standards as issued by the International Accounting Standards Board.
Restatement of 2018 and 2017 financial statements
As discussed in Note 1 to the financial statements, the 2018 and 2017 financial statements have been restated to correct a misstatement in relation to warrant expenses.
Change in Accounting Principle
As discussed in Note 1 to the financial statements, effective January 1, 2019, the Company adopted IFRS 16 Leases, using the modified retrospective approach.
Basis for Opinion
These financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on the Company's financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company's internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles
95
used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
Deloitte
Statsautoriseret Revisonspartnerselskab
CVR no: 33963556
| /s/ Sumit Sudan State Authorized Public Accountant |
/s/ Kåre Valtersdorf State Authorized Public Accountant |
Copenhagen, Denmark
March 13, 2020
We have served as the Company's auditor since 2014.
96
Exhibit 2.1
DESCRIPTION OF THE REGISTRANT’S SECURITIES
REGISTERED PURSUANT TO SECTION 12 OF THE
SECURITIES EXCHANGE ACT OF 1934
Set forth below is a summary of certain information concerning our share capital, as well as a description of certain provisions of our articles of association and relevant provisions of the Danish Companies Act. The summary includes certain references to, and descriptions of, material provisions of our articles of association and Danish law. The summary below contains only material information concerning our share capital and corporate status and does not purport to be complete and is qualified in its entirety by reference to our articles of association and applicable Danish law. Further, please note that if you are a holder of ordinary shares represented by American Depositary Shares, or ADSs, then you are not treated as one of our shareholders and do not have any shareholder rights.
GENERAL
Zealand Pharma A/S is incorporated as a public limited liability company under Danish law and is registered with the Danish Business Authority in Copenhagen, Denmark under registration number (CVR) no. 20045078. Our ordinary shares have been listed on Nasdaq Copenhagen since November 2010 under the symbol “ZEAL” and the ADSs have been listed on the Nasdaq Global Select Market in the United States since August 2017 under the symbol “ZEAL.”
DESCRIPTION OF OUR SHARES
As of December 31, 2019, our registered, issued and outstanding share capital was DKK 36,054,661, distributed into 36,054,661 ordinary shares of nominal value DKK 1 each, and excludes up to 1,690,147 shares that may be issued upon the exercise of outstanding warrants.
Authorizations to Our Board of Directors
Our board of directors is authorized to increase our share capital as follows:
· Until April 19, 2023, our board of directors is authorized to increase our share capital by issuing new shares by up to nominally DKK 6,150,265 of new shares (equal to 6,150,265 new shares of a nominal amount of DKK 1 per share). As of December 31, 2019 the nominal share capital increase available under this authorization is 1,372,406 (equal to 1,372,406 new shares of a nominal amount of DKK 1 per share). The capital increase shall be effected at market price and must be implemented without pre-emption rights for the existing shareholders. Our board of directors may decide to implement a capital increase by way of a cash contribution, contribution in kind or debt conversion. Further, during this period, our board of directors is authorized to increase our share capital by issuing new shares by up to a nominal amount of DKK 6,150,265(equal to 6,150,265 new shares of a nominal amount of DKK 1 per share). Such capital increase shall be implemented with pre-emption rights for the existing shareholders and the subscription price may be a favorable price fixed by our board of directors. Our board of directors may decide to implement the capital increase by way of a cash contribution, contribution in kind or debt conversion. Any capital increases pursuant to such authorizations cannot exceed a total aggregate nominal amount of DKK 6,150,265.
· Until April 21, 2020, our board of directors is authorized to grant warrants with a right to subscribe for 2,750,000 shares. As of December 31, 2019, as a result of issuances and lapses of warrants, the number of warrants available for issuance under this authorization is 780,737. The shareholders will not have pre-emptive subscription rights when our board of directors exercises this authorization and the specific terms and conditions in this respect are to be determined by our board of directors. Our board of directors shall determine, in its discretion, the exercise price, as well as other terms and conditions for the warrants, provided that the exercise price corresponds to a minimum of the market price at the time of issuance of the warrants, unless such warrants are issued to employees. Pursuant to relevant provisions of the Danish Companies Act in force from time to time, our board of directors may reapply or reissue any lapsed non-exercised warrants,
provided that such reapplication or reissue is made under terms and conditions and with the time limits specified under the authority.
New shares issued according to the authorizations listed above will rank pari passu with our existing shares in accordance with our articles of association.
Further, our board of directors is, in the period until the next annual general meeting, authorized on behalf of the company to acquire its own shares and/or ADSs for a total nominal value of up to 10% of our share capital. The price paid for such shares or ADSs may not deviate by more than 10% from the price quoted by Nasdaq Copenhagen or Nasdaq Global Select Market, respectively, at the time of acquisition.
Pre-emptive Rights
If our shareholders at a general meeting resolve to increase our share capital by a cash contribution, section 162 of the Danish Companies Act will apply. Under that section, shareholders have a pre-emptive right to subscribe for new shares in proportion to their existing shareholdings. However, the pre-emptive right may be derogated from by a majority comprising at least two-thirds of the votes cast, as well as at least two-thirds of the share capital represented at the general meeting, provided the share capital increase takes place at market price or nine-tenths of the votes cast, as well as at least nine-tenths of the share capital represented at the general meeting if the share capital increase takes place below market price, unless (i) such capital increase is directed at certain but not all shareholders (in which case all shareholders must consent); or (ii) such capital increase is directed at our employees whereby a majority comprising at least two-thirds of the votes cast, as well as at least two-thirds of the share capital represented at the general meeting is required. Further, the pre-emptive rights may be derogated from by an exercise of the board of directors of a valid authorization in our articles of association, provided that the share capital increase takes place at or above market price, unless adopted in accordance with the procedures as set out above.
Shareholders’ Register
We are obliged pursuant to Danish law to maintain a shareholders’ register (Ejerbog) for our ordinary shares. The shareholders’ register is maintained by Computershare A/S, Lottenborgvej 26, DK-2800 Kgs. Lyngby, Denmark, our Danish share registrar and transfer agent. It is mandatory that the shareholders’ register is maintained within the European Union and that it is available to public authorities.
Pursuant to the Danish Companies Act, public and private limited liability companies are required to register with the Danish Business Authority information regarding shareholders who own at least 5% of the share capital or the voting rights. Pursuant to this provision, we file registrations with the Danish Public Shareholders’ Register of the Danish Business Authority. Shareholders that exceed or fall below the ownership threshold must notify us, and we will subsequently file the information with the Danish Business Authority. Reporting is further required upon passing or falling below thresholds of 10%, 15%, 20%, 25%, 50%, 90%, and 100% as well as one-third and two-thirds of the votes or the share capital. This also applies to beneficial holders of our shares, such as holders of the ADSs.
Articles of Association and Danish Corporate Law
With respect to our articles of association, the following should be emphasized:
Objectives
Our company has been established with the objectives of engaging in research, trade, manufacture and related activities, primarily within the pharmaceutical industry.
Rights and Restrictions in Relation to Existing Shares
· No share carries any special rights.
· Each share with a nominal value of DKK 1 carries one vote at general meetings.
· The shares are negotiable instruments, and no restrictions apply to the transferability of the shares.
· No shareholder shall be obliged to let his shares be redeemed in full or in part by us or by any other party, except as provided in the Danish Companies Act.
· All shares shall be registered in the names of the holders and shall be entered in our shareholders’ register.
Adoptions of decisions on our general meetings
All resolutions put to the vote of shareholders at general meetings are subject to adoption by a simple majority of votes, unless the Danish Companies Act or our articles of association prescribes other requirements.
Notice Convening Annual and Extraordinary General Meetings
General meetings shall be held in Greater Copenhagen (Storkøbenhavn). General meetings shall be convened by the board of directors giving not less than three weeks’ and not more than five weeks’ notice. General meetings shall be announced by publication in the IT information system of the Danish Business Authority and on our website. Furthermore, all shareholders registered in our shareholders’ register who have so requested shall be notified by letter or email. The notice shall set out the time and place for the general meeting and the issues to be considered at the general meeting. If the general meeting is to consider a proposal to amend our articles of association, then the notice shall specify the material content of the proposal.
A shareholder’s right to attend general meetings and to vote is determined on the basis of the shares that the shareholder owns on the registration date which date is one week before the general meeting is held.
Any shareholder shall be entitled to attend general meetings, provided he or she has requested an admission card from our offices not later than three days prior to the relevant meeting. The admission card will be issued to the shareholders registered in our shareholders’ register. The shareholder may attend in person or be represented by proxy, and a shareholder shall be entitled to attend together with an advisor. A shareholder may vote by proxy or by postal, and a form for this use shall be made available on our website no later than three weeks prior to the general meeting. A vote by mail must be received by us not later than three days prior to the general meeting in order to be counted at the general meeting.
Extraordinary general meetings shall be held as directed by the shareholders at the general meeting, the board of directors or an auditor, or upon a written request to the board of directors by shareholders holding not less than 5% of the share capital for consideration of a specific issue. The general meeting shall be convened with three to five weeks notice within 14 days after the proper request has been received by our board of directors.
Provisions as to the Level of Equity Investments to be Notified to Us and the Danish Authorities
Shareholders in Danish companies with shares admitted to trading and official listing on Nasdaq Copenhagen are, pursuant to Section 38 of the Danish Capital Markets Act, required to give simultaneous notice to the company and the Danish Financial Supervisory Authority, or the FSA, of the shareholding in the company, when the shareholding reaches, exceeds or falls below thresholds of 5%, 10%, 15%, 20%, 25%, 50% or 90% and limits of one-third or two-thirds of the voting rights or nominal value of the total share capital.
A shareholder in a company means a natural or legal person who, directly or indirectly, holds: (i) shares in the company on behalf of himself and for his own account; (ii) shares in the company on behalf of himself, but for the account of another natural or legal person; or (iii) depository receipts, where such holder is considered a shareholder in relation to the underlying shares represented by the depository receipts.
The duty to notify set forth above further applies to natural and legal persons who are entitled to acquire, sell or exercise voting rights which are:
(i) held by a third party with whom that natural or legal person has concluded an agreement, which obliges them to adopt, by concerted exercise of the voting rights they hold, a lasting common policy towards the management of the issuer in question (common duty to inform for all parties to the agreement);
(ii) held by a third party under an agreement concluded with that natural or legal person providing for the temporary transfer of the voting rights in question in return for consideration;
(iii) attached to shares which are lodged as collateral for that natural or legal person, provided the person controls the voting rights and declares an intention of exercising them;
(iv) attached to shares in which that natural or legal person has a lifelong right of disposal;
(v) held, or may be exercised within the meaning of (i) to (iv), by an undertaking controlled by that person or entity;
(vi) attached to shares deposited with that natural or legal person and which the person can exercise at his own discretion in the absence of specific instructions from the shareholders;
(vii) held by a third party in its own name on behalf of that person; or
(viii) exercisable by that person through a proxy where that person may exercise the voting rights at his discretion in the absence of specific instructions of the shareholder.
The duty to notify set forth above also applies to anyone, who directly or indirectly holds (a) financial instruments that afford the holder either an unconditional right to acquire or the discretion as to his right to acquire existing shares (e.g., share options); and/or (b) financial instruments based on existing shares and with an economic effect equal to that of the financial instruments mentioned in (a), regardless of them not affording the right to purchase existing shares (e.g., the ADSs or, under the circumstances, cash-settled derivatives linked to the value of our shares or ADSs). Holding these kinds of financial instruments counts towards the thresholds mentioned above and may thus trigger a duty to notify by themselves or when accumulated with a holding of shares or ADSs. The FSA will in certain cases publish information concerning sanctions imposed, including, as a general rule, the name of the shareholder in question, as a consequence of non-compliance with the above rules.
The notification shall be made promptly but not later than four weekdays after the shareholder was aware or should have become aware of the completion of the transaction, and in accordance with the provisions of Danish Executive Order on Major Shareholders. The shareholder is deemed to have become aware of the completion of the transaction no later than two weekdays after the completion of the transaction. The shareholder shall disclose the change in voting rights and shares, including the number of voting rights (and the division of voting rights between share classes, if applicable) and shares held directly or indirectly by the shareholder following the transaction. The notification shall further state the transaction date on which the threshold was reached or no longer reached and the identity of the shareholder as well as the identity of any natural or legal person with the right to vote on behalf of the shareholder and in the case of a group structure, the chain of controlled undertakings through which voting rights are effectively held. The information shall be notified to the company and simultaneously submitted electronically to the FSA. Failure to comply with the notification requirements is punishable by fine or suspension of voting rights in instances of gross or repeated non-compliance.
When an obligation to notify rests on more than one natural or legal person, the notification may be made through a joint notification. However, use of a joint notification does not exempt the individual shareholders or natural or legal persons from their responsibilities in connection with the obligation to notify or the contents of the notification.
After receipt of the notification, but not later than three weekdays thereafter, the company shall publish the contents of the notification.
Furthermore, the general duty of notification under Section 55 of the Danish Companies Act in respect of notification of significant holdings (similar to the thresholds set out in the Danish Capital Markets Act Section 38) applies,
including when the limit of 100% of the share capital’s voting rights or nominal value of the company is reached or are no longer reached.
The EU Short Selling Regulation (EU Regulation 236/2012) Includes Certain Notification Requirements in connection with Short Selling of Shares Admitted to Trading on a Trading Venue (including Nasdaq Copenhagen) and Securities or Derivatives that Relate to Such Shares (including the ADSs).
When a natural or legal person reaches or falls below a net, short position of 0.2% of the issued share capital of a company that has shares admitted to trading on a trading venue, such person shall make a private notification (i.e. such notification will not be made public) to the relevant competent authority, which in Denmark is the FSA. The obligation to notify the FSA, moreover, applies in each case where the short position reaches or no longer reaches 0.1% above the 0.2% threshold. In addition, when a natural or legal person reaches or falls below a net short position of 0.5% of the issued share capital of a company that has shares admitted to trading on a trading venue and each 0.1% above that, such person shall make a public notification of its net short position via the FSA. The notification requirements apply to both physical and synthetic short positions. In addition uncovered short selling (naked short selling) of shares admitted to trading on a trading venue is prohibited.
Mandatory Tender Offers
The Danish Capital Markets Act (Part 8) and the Danish Executive Order on Takeover include rules concerning public offers for the acquisition of shares admitted to trading on a regulated market (including Nasdaq Copenhagen).
If a shareholding is transferred, directly or indirectly, in a company with one or more share classes admitted to trading on a regulated market, to an acquirer or to persons acting in concert with such acquirer, the acquirer and the persons acting in concert with such acquirer, if applicable, shall give all shareholders of the company the option to dispose of their shares on identical terms, if the acquirer or the persons acting in concert with such acquirer gains control over the company as a result of the transfer.
Control as mentioned above exists if the acquirer or persons acting in concert with such acquirer, directly or indirectly, holds at least one-third of the voting rights in the company, unless it can be clearly proven in special cases that such ownership does not constitute control. An acquirer or persons acting in concert with such acquirer who does not hold at least one-third of the voting rights in a company, nevertheless has control when the acquirer has or persons acting in concert with such acquirer have:
· the right to control at least one-third of the voting rights in the company according to an agreement with other investors; or
· the right to appoint or dismiss a majority of the members of the central governing body.
Voting rights attached to treasury shares shall be included in the calculation of voting rights.
The Danish Capital Markets Act contains specific exemptions from the obligation to submit a mandatory takeover offer, including transfers of shares by inheritance or transfer within the same group and as a result of a creditor’s debt enforcement proceedings. Exemptions from the mandatory tender offer rules may be granted under special circumstances by the FSA.
Comparison of Danish Corporate Law and Our Articles of Association and Delaware Corporate Law
The following comparison between Danish corporate law, which applies to us, and Delaware corporate law, the law under which many publicly listed companies in the United States are incorporated, discusses shareholder rights and obligations and certain additional matters. This summary is subject to Danish law, including the Danish Companies Act, and Delaware corporate law, including the Delaware General Corporation Law. Further, please note that if you are a holder of the ADSs, then you are not treated as one of our shareholders and do not have any shareholder rights in Zealand Pharma A/S.
Shareholder Rights
Notice of Meeting
Denmark. According to the Danish Companies Act and as implemented in our articles of association, general meetings in listed limited liability companies shall be convened by the board of directors with a minimum of three weeks’ notice and a maximum of five weeks’ notice. A convening notice shall also be forwarded to shareholders recorded in our shareholders’ register who have requested such notification. There are specific requirements as to the information and documentation required to be disclosed in connection with the convening notice.
Delaware. Under Delaware law, unless otherwise provided in the certificate of incorporation or bylaws, written notice of any meeting of the stockholders must be given to each stockholder entitled to vote at the meeting not less than 10 nor more than 60 days before the date of the meeting and shall specify the place, date, hour and purpose or purposes of the meeting.
Voting Rights
Denmark. Each share confers the right to cast one vote at the general meeting of shareholders, unless the articles of association provide otherwise. Each holder of shares may cast as many votes as it holds shares. Voting instructions may be given only in respect of a number of ADSs representing an integral number of shares or other deposited securities. Shares that are held by us or our direct or indirect subsidiaries do not confer the right to vote.
ADS holders may only exercise voting rights with respect to the shares underlying their respective ADSs in accordance with the provisions of the deposit agreement, which provides that a holder may vote the shares underlying any ADSs for any particular matter to be voted on by our shareholders either by withdrawing the shares underlying the ADSs or, to the extent permitted by applicable law and as permitted by the depositary, by requesting temporary registration as shareholder and authorizing the depositary to act as proxy. The depositary will try, as far as practical, to vote the shares underlying the ADSs as instructed by the ADS holders.
Delaware. Under the Delaware General Corporation Law, each stockholder is entitled to one vote per share of stock, unless the certificate of incorporation provides otherwise. In addition, the certificate of incorporation may provide for cumulative voting at all elections of directors of the corporation, or at elections held under specified circumstances. Either the certificate of incorporation or the bylaws may specify the number of shares and/or the amount of other securities that must be represented at a meeting in order to constitute a quorum, but in no event can a quorum consist of less than one-third of the shares entitled to vote at a meeting.
Stockholders as of the record date for the meeting are entitled to vote at the meeting, and the board of directors may fix a record date that is no more than 60 nor less than 10 days before the date of the meeting, and if no record date is set then the record date is the close of business on the day next preceding the day on which notice is given, or if notice is waived then the record date is the close of business on the day next preceding the day on which the meeting is held. The determination of the stockholders of record entitled to notice or to vote at a meeting of stockholders shall apply to any adjournment of the meeting, but the board of directors may fix a new record date for the adjourned meeting.
Shareholder Proposals
Denmark. According to the Danish Companies Act, extraordinary general meetings of shareholders will be held whenever our board of directors or our appointed auditor requires. In addition, one or more shareholders each representing at least 5% of the registered share capital of the company may, in writing, require that a general meeting be convened. If such a demand is made, the board of directors shall convene the general meeting with three to five weeks notice within 14 days thereafter.
All shareholders have the right to present proposals for adoption at the annual general meeting, provided that the proposals are submitted at least six weeks prior to the meeting. In the event that the request is made at a later date, the board of directors will determine whether the proposals were made in due time to be included on the agenda.
Delaware. Delaware law does not specifically grant stockholders the right to bring business before an annual or special meeting of stockholders. However, if a Delaware corporation is subject to the SEC’s proxy rules, a stockholder who owns at least $2,000 in market value, or 1% of the corporation’s securities entitled to vote, may propose a matter for a vote at an annual or special meeting in accordance with those rules.
Action by Written Consent
Denmark. Under Danish law, shareholders may take action and pass resolutions by written consent if such consent is unanimous. However, for a listed company, this method of adopting resolutions is generally not feasible.
Delaware. Although permitted by Delaware law, publicly listed companies do not typically permit stockholders of a corporation to take action by written consent.
Appraisal Rights
Denmark. The concept of appraisal rights does not exist under Danish law, except in connection with statutory redemption rights according to the Danish Companies Act.
According to Section 73 of the Danish Companies Act, a minority shareholder may require a majority shareholder that holds more than 90% of the company’s registered share capital to redeem his or her shares. Similarly, a majority shareholder holding more than 90% of the company’s share capital may, according to Section 70 of the Danish Companies Act, squeeze out the minority shareholders. In the event that the parties cannot agree to the redemption squeeze out price, this shall be determined by an independent evaluator appointed by the court. Additionally, there are specific regulations in Sections 249, 267, 285 and 305 of the Danish Companies Act that require compensation in the event of national or cross-border mergers and demergers. Moreover, shareholders who vote against a cross-border merger or demerger are, according to Sections 286 and 306 of the Danish Companies Act, entitled to have their shares redeemed.
Delaware. The Delaware General Corporation Law provides for stockholder appraisal rights, or the right to demand payment in cash of the judicially determined fair value of the stockholder’s shares, in connection with certain mergers and consolidations.
Shareholder Suits
Denmark. Under Danish law, only a company itself can bring a civil action against a third party; an individual shareholder does not have the right to bring an action on behalf of a company. However, if shareholders representing at least 10% of the share capital have opposed at a general meeting a decision to grant discharge to a member of our board of directors or our executive management or refrain from bringing law suits against, among other persons, a member of our board of directors or executive management, a shareholder may bring a derivative action on behalf of our company against, among other persons, a member of our board of directors or executive management. An individual shareholder may, in its own name, have an individual right to take action against such third party in the event that the cause for the liability of that third party also constitutes a negligent act directly against such individual shareholder.
Delaware. Under the Delaware General Corporation Law, a stockholder may bring a derivative action on behalf of the corporation to enforce the rights of the corporation. An individual also may commence a class action suit on behalf of himself and other similarly situated stockholders where the requirements for maintaining a class action under Delaware law have been met. A person may institute and maintain such a suit only if that person was a stockholder at the time of the transaction which is the subject of the suit. In addition, under Delaware case law, the plaintiff normally must be a stockholder at the time of the transaction that is the subject of the suit and throughout the duration of the derivative suit. Delaware law also requires that the derivative plaintiff make a demand on the directors of the corporation to assert the corporate claim before the suit may be prosecuted by the derivative plaintiff in court, unless such a demand would be futile.
Repurchase of Shares
Denmark. Danish limited liability companies may not subscribe for newly issued shares in their own capital. Such companies may, however, according to the Danish Companies Act Sections 196-201, acquire fully paid shares of themselves, provided that the board of directors has been authorized to do so by the shareholders at a general meeting. Such authorization can only be given for a maximum period of five years and the authorization shall fix (i) the maximum value of the shares and (ii) the minimum and the highest amount that the company may pay for the shares. Such purchase of shares may generally only be acquired using distributable reserves. In addition, the board of directors may, on behalf of the company, acquire the company’s own shares, without authorization, in case it is necessary to avoid a considerable and imminent detrimental effect on the company and provided certain conditions are met. In case the company has acquired its own shares under such circumstances the board of directors is obligated to inform the shareholders of such acquisition at the next general meeting.
Delaware. Under the Delaware General Corporation Law, a corporation may purchase or redeem its own shares unless the capital of the corporation is impaired or the purchase or redemption would cause an impairment of the capital of the corporation. A Delaware corporation may, however, purchase or redeem out of capital any of its preferred shares or, if no preferred shares are outstanding, any of its own shares if such shares will be retired upon acquisition and the capital of the corporation will be reduced in accordance with specified limitations.
Anti-Takeover Provisions
Denmark. Under Danish law, it is possible to implement limited protective anti-takeover measures. Such provisions may include, among other things, (i) different share classes with different voting rights and (ii) notification requirements concerning participation in general meetings. We have currently not adopted any such provisions, except for the obligation to request an admission card. See description above under the caption “Articles of Association and Danish Corporate Law—Notice Convening Annual and Extraordinary General Meetings.”
Delaware. In addition to other aspects of Delaware law governing fiduciary duties of directors during a potential takeover, the Delaware General Corporation Law also contains a business combination statute that protects Delaware companies from hostile takeovers and from actions following the takeover by prohibiting some transactions once an acquirer has gained a significant holding in the corporation.
Section 203 of the Delaware General Corporation Law prohibits “business combinations,” including mergers, sales and leases of assets, issuances of securities and similar transactions by a corporation or a subsidiary with an interested stockholder that beneficially owns 15% or more of a corporation’s voting stock, within three years after the person becomes an interested stockholder, unless:
· the transaction that will cause the person to become an interested stockholder is approved by the board of directors of the target prior to the transaction;
· after the completion of the transaction in which the person becomes an interested stockholder, the interested stockholder holds at least 85% of the voting stock of the corporation not including shares owned by persons who are directors and officers of interested stockholders and shares owned by specified employee benefit plans; or
· after the person becomes an interested stockholder, the business combination is approved by the board of directors of the corporation and holders of at least 66.67% of the outstanding voting stock, excluding shares held by the interested stockholder.
A Delaware corporation may elect not to be governed by Section 203 by a provision contained in the original certificate of incorporation of the corporation or an amendment to the original certificate of incorporation or to the bylaws of the company, which amendment must be approved by a majority of the shares entitled to vote and may not be further amended by the board of directors of the corporation. Such an amendment is not effective until 12 months following its adoption.
Inspection of Books and Records
Denmark. According to Section 150 of the Danish Companies Act, a shareholder may, at the annual general meeting or at a general meeting whose agenda includes such item, request an inspection of the company’s books regarding specific issues concerning the management of the company or specific annual reports. If approved by shareholders with a simple majority, one or more investigators are elected. If the proposal is not approved by a simple majority but 25% of the share capital votes in favor of the proposal, then the shareholder can request the court to appoint an investigator.
Delaware. Under the Delaware General Corporation Law, any stockholder may inspect certain of the corporation’s books and records, for any proper purpose, during the corporation’s usual hours of business.
Pre-Emptive Rights
Denmark. As a general rule, shareholders of the company are entitled to subscribe for new shares in proportion to their existing shareholdings in the event of a cash increase of the share capital. Such a cash increase of the share capital can be resolved by the general meeting by at least two-thirds of the votes cast as well as at least two-thirds of the share capital represented at the general meeting.
However, in the below-mentioned scenarios, the general meeting may resolve to depart from the shareholders’ right to proportionate subscription if the following voting requirements are met:
· two-thirds majority requirement: if the new shares issued in connection with the capital increase are subscribed for at market price for the benefit of some of the existing shareholders, the above-mentioned two-thirds majority requirement applies;
· consent requirement: if the new shares issued in connection with the capital increase are subscribed for at a discount for the benefit of some of the existing shareholders, consent from the shareholders who do not get an opportunity to participate in the capital increase must be obtained;
· two-thirds majority requirement: if the new shares issued in connection with the capital increase are subscribed for at market price for the benefit of parties other than the existing shareholders (i.e., a third party or employees of the company), the above-mentioned two-thirds majority requirement applies; and
· nine-tenths majority requirement: if the new shares issued in connection with the capital increase are subscribed for at discount for the benefit of parties other than the existing shareholders or the employees of the company, the voting requirement is at least nine-tenths of the votes cast as well as at least nine-tenths of the share capital represented at the general meeting.
The board of directors may resolve to increase our share capital without pre-emptive subscription rights for existing shareholders pursuant to the authorizations described above under the caption “Authorizations to our Board of Directors.”
Unless future issuances of new shares are registered under the Securities Act or with any authority outside Denmark, U.S. shareholders and shareholders in jurisdictions outside Denmark may be unable to exercise their pre-emptive subscription rights under U.S. securities law.
Delaware. Under the Delaware General Corporation Law, stockholders have no pre-emptive rights to subscribe for additional issues of stock or to any security convertible into such stock unless, and to the extent that, such rights are expressly provided for in the certificate of incorporation.
Dividends
Denmark. Under Danish law, the distribution of ordinary and extraordinary dividends requires the approval of a company’s shareholders at a company’s general meeting. In addition the shareholders may authorize the board of directors to distribute extraordinary dividends. We may only pay out dividends from our distributable reserves, which are defined as results from operations carried forward and reserves that are not bound by law after deduction of loss
carried forward. It is possible under Danish law to pay out extraordinary dividends. The decision to pay out extraordinary dividends shall be accompanied by a balance sheet, and the board of directors determines whether it will be sufficient to use the statement of financial position from the annual report or if an interim statement of financial position for the period from the annual report period until the extraordinary dividend payment shall be prepared. If the decision to distribute extraordinary dividends is passed more than six months after the date of the statement of financial position as set out in our latest adopted annual report, an interim statement of financial position must be prepared and reviewed by our auditor. The statement of financial position or the interim statement of financial position, as applicable, must show that sufficient funds are available for distribution. Our general meeting of shareholders cannot resolve to distribute dividends at an amount exceeding the amount recommended or approved by our Board of Directors. Moreover, ordinary dividends and extraordinary dividends may only be made out of distributable reserves and may not exceed what is considered sound and adequate with regard to our financial condition or be to the detriment of our creditors.
Delaware. Under the Delaware General Corporation Law, a Delaware corporation may pay dividends out of its surplus (the excess of net assets over capital), or in case there is no surplus, out of its net profits for the fiscal year in which the dividend is declared and/or the preceding fiscal year (provided that the amount of the capital of the corporation is not less than the aggregate amount of the capital represented by the issued and outstanding stock of all classes having a preference upon the distribution of assets). In determining the amount of surplus of a Delaware corporation, the assets of the corporation, including stock of subsidiaries owned by the corporation, must be valued at their fair market value as determined by the board of directors, without regard to their historical book value. Dividends may be paid in the form of shares, property or cash.
Shareholder Vote on Certain Reorganizations
Denmark. Under Danish law, all amendments to the articles of association shall be approved by the general meeting of shareholders with a minimum of two-thirds of the votes cast and two-thirds of the share capital represented at the general meeting. The same applies to solvent liquidations, mergers with the company as the discontinuing entity, mergers with the company as the continuing entity if shares are issued in connection therewith and demergers. Under Danish law, it is debatable whether the shareholders must approve a decision to sell all or virtually all of the company’s business/assets.
Delaware. Under the Delaware General Corporation Law, the vote of a majority of the outstanding shares of capital stock entitled to vote thereon generally is necessary to approve a merger or consolidation or the sale of all or substantially all of the assets of a corporation. The Delaware General Corporation Law permits a corporation to include in its certificate of incorporation a provision requiring for any corporate action the vote of a larger portion of the stock or of any class or series of stock than would otherwise be required. However, under the Delaware General Corporation Law, no vote of the stockholders of a surviving corporation to a merger is needed, unless required by the certificate of incorporation, if (1) the agreement of merger does not amend in any respect the certificate of incorporation of the surviving corporation, (2) the shares of stock of the surviving corporation are not changed in the merger and (3) the number of shares of common stock of the surviving corporation into which any other shares, securities or obligations to be issued in the merger may be converted does not exceed 20% of the surviving corporation’s common stock outstanding immediately prior to the effective date of the merger. In addition, stockholders may not be entitled to vote in certain mergers with other corporations that own 90% or more of the outstanding shares of each class of stock of such corporation, but the stockholders will be entitled to appraisal rights.
Amendments to Governing Documents
Denmark. All resolutions made by the general meeting may be adopted by a simple majority of the votes, subject only to the mandatory provisions of the Danish Companies Act and the articles of association. Resolutions concerning all amendments to the articles of association must be passed by two-thirds of the votes cast as well as two-thirds of the share capital represented at the general meeting. Certain resolutions, which limit a shareholder’s ownership or voting rights, are subject to approval by a nine-tenth majority of the votes cast and the share capital represented at the general meeting. Decisions to impose any or increase any obligations of the shareholders towards the company require unanimity.
Delaware. Under the Delaware General Corporation Law, a corporation’s certificate of incorporation may be amended only if adopted and declared advisable by the board of directors and approved by a majority of the outstanding shares entitled to vote, and the bylaws may be amended with the approval of a majority of the outstanding shares entitled to vote and may, if so provided in the certificate of incorporation, also be amended by the board of directors.
Transfer Agent and Registrar
The transfer agent and registrar for our shares is Computershare A/S, Kongevejen 418, Øverød, DK-2840 Holte, Denmark.
DESCRIPTION OF OUR AMERICAN DEPOSITARY SHARES
American Depositary Shares
The Bank of New York Mellon, as depositary, registers and delivers ADSs. Each ADS represents one ordinary share (or a right to receive one ordinary share) deposited with the Copenhagen office of Danske Bank A/S, as custodian for the depositary in Denmark. Each ADS also represents any other securities, cash or other property which may be held by the depositary in respect of the depositary facility. A copy of our Deposit Agreement among us, the depositary, owners and holders of ADSs was filed with the SEC as an exhibit to our registration statement on Form F-1/A filed on August 3, 2017 (File No. 333-219184).
Any ordinary shares that we may issue, whether directly or upon exercise of warrants, can be deposited for delivery of ADSs. The ADSs may be uncertificated securities or certificated securities evidenced by American Depositary Receipts, or ADRs. The depositary’s office at which the ADSs are administered and its principal executive office are located at 240 Greenwich Street, New York, New York 10286.
You may hold ADSs either (a) directly (i) by having an American Depositary Receipt, also referred to as an ADR, which is a certificate evidencing a specific number of ADSs, registered in your name, or (ii) by having uncertificated ADSs registered in your name, or (b) indirectly by holding a security entitlement in ADSs through your broker or other financial institution that is a direct or indirect participant in The Depository Trust Company, also called DTC. If you hold ADSs directly, you are a registered ADS holder, also referred to as an ADS holder. This description assumes you are an ADS holder. If you hold the ADSs indirectly, you must rely on the procedures of your broker or other financial institution to assert the rights of ADS holders described in this section. You should consult with your broker or financial institution to find out what those procedures are.
Registered holders of uncertificated ADSs receive statements from the depositary confirming their holdings.
We do not treat ADS holders as shareholders and ADS holders do not have shareholder rights. Danish law governs shareholder rights. The depositary is the holder of the shares underlying ADSs. Registered holders of ADSs have ADS holder rights. A deposit agreement among us, the depositary, ADS holders and all other persons indirectly or beneficially holding ADSs sets out ADS holder rights as well as the rights and obligations of the depositary. New York law governs the deposit agreement and the ADSs.
The following is a summary of the material provisions of the deposit agreement. For more complete information, you should read the entire deposit agreement and the form of ADR.
Dividends and Other Distributions
How will you receive dividends and other distributions on the ordinary shares?
The depositary has agreed to pay or distribute to ADS holders the cash dividends or other distributions it or the custodian receives on shares or other deposited securities, upon payment or deduction of its fees and expenses. You will receive these distributions in proportion to the number of shares your ADSs represent.
Cash. The depositary will convert any cash dividend or other cash distribution we pay on the shares into U.S. dollars, if it can do so on a reasonable basis and can transfer the U.S. dollars to the United States. If that is not possible or if any government approval is needed and cannot be obtained, the deposit agreement allows the depositary to distribute the foreign currency only to those ADS holders to whom it is possible to do so. It will hold the foreign currency it cannot convert for the account of the ADS holders who have not been paid. It will not invest the foreign currency and it will not be liable for any interest. Before making a distribution, any withholding taxes, or other governmental charges that must be paid will be deducted. See the section entitled “Taxation.” It will distribute only whole U.S. dollars and cents and will round fractional cents to the nearest whole cent. If the exchange rates fluctuate during a time when the depositary cannot convert the foreign currency, you may lose some of the value of the distribution
Shares. The depositary may distribute additional ADSs representing any shares we distribute as a dividend or free distribution. The depositary will only distribute whole ADSs. It will sell shares which would require it to deliver a fraction of an ADS (or ADSs representing those shares) and distribute the net proceeds in the same way as it does with cash. If the depositary does not distribute additional ADSs, the outstanding ADSs will also represent the new shares. The depositary may sell a portion of the distributed shares (or ADSs representing those shares) sufficient to pay its fees and expenses in connection with that distribution.
Rights to Purchase Additional Ordinary Shares. If we offer holders of our securities any rights to subscribe for additional shares or any other rights, the depositary may (i) exercise those rights on behalf of ADS holders, (ii) distribute those rights to ADS holders or (iii) sell those rights and distribute the net proceeds to ADS holders, in each case after deduction or upon payment of its fees and expenses. To the extent the depositary does not do any of those things, it will allow the rights to lapse. In that case, you will receive no value for them. The depositary will exercise or distribute rights only if we ask it to and provide satisfactory assurances to the depositary that it is legal to do so. If the depositary will exercise rights, it will purchase the securities to which the rights relate and distribute those securities or, in the case of shares, new ADSs representing the new shares, to subscribing ADS holders, but only if ADS holders have paid the exercise price to the depositary. U.S. securities laws may restrict the ability of the depositary to distribute rights or ADSs or other securities issued on exercise of rights to all or certain ADS holders, and the securities distributed may be subject to restrictions on transfer.
Other Distributions. The depositary will send to ADS holders anything else we distribute on deposited securities by any means it thinks is legal, fair and practical. If it cannot make the distribution in that way, the depositary has a choice. It may decide to sell what we distributed and distribute the net proceeds, in the same way as it does with cash. Or, it may decide to hold what we distributed, in which case ADSs will also represent the newly distributed property. However, the depositary is not required to distribute any securities (other than ADSs) to ADS holders unless it receives satisfactory evidence from us that it is legal to make that distribution. The depositary may sell a portion of the distributed securities or property sufficient to pay its fees and expenses in connection with that distribution. U.S. securities laws may restrict the ability of the depositary to distribute securities to all or certain ADS holders, and the securities distributed may be subject to restrictions on transfer.
The depositary is not responsible if it decides that it is unlawful or impractical to make a distribution available to any ADS holders. We have no obligation to register ADSs, shares, rights or other securities under the Securities Act. We also have no obligation to take any other action to permit the distribution of ADSs, shares, rights or anything else to ADS holders. This means that you may not receive the distributions we make on our shares or any value for them if it is illegal or impractical for us to make them available to you.
Deposit, Withdrawal and Cancellation
How are ADSs issued?
The depositary will deliver ADSs if you or your broker deposits shares or evidence of rights to receive shares with the custodian. Upon payment of its fees and expenses and of any taxes or charges, such as stamp taxes or stock transfer taxes or fees, the depositary will register the appropriate number of ADSs in the names you request and will deliver the ADSs to or upon the order of the person or persons that made the deposit.
How can ADS holders withdraw the deposited securities?
You may surrender your ADSs for the purpose of withdrawal at the depositary’s office. Upon payment of its fees and expenses and of any taxes or charges, such as stamp taxes or stock transfer taxes or fees, the depositary will deliver the shares and any other deposited securities underlying the ADSs to the ADS holder or a person the ADS holder designates at the office of the custodian. Or, at your request, risk and expense, the depositary will deliver the deposited securities at its office, if feasible. The depositary may charge you a fee and its expenses for instructing the custodian regarding delivery of deposited securities.
How can ADS holders interchange between certificated ADSs and uncertificated ADSs?
You may surrender your ADRs to the depositary for the purpose of exchanging your ADRs for uncertificated ADSs. The depositary will cancel the ADRs and will send you a statement confirming that you are the owner of uncertificated ADSs. Alternatively, upon receipt by the depositary of a proper instruction from a registered holder of uncertificated ADSs requesting the exchange of uncertificated ADSs for certificated ADSs, the depositary will execute and deliver to you an ADR evidencing those ADSs.
Voting Rights of Deposited Securities
How do you vote?
ADS holders eligible to vote may instruct the depositary to vote the number of deposited shares their ADSs represent. The depositary will notify ADS holders of shareholders’ meetings and arrange to deliver our voting materials to them if we ask it to. Those materials will describe the matters to be voted on and explain how ADS holders may instruct the depositary how to vote. For instructions to be valid, they must reach the depositary by a date set by the depositary.
If we requested the depositary to act, the depositary will endeavor, as far as practical, subject to the laws of Demark and the provisions of our articles of association or similar documents, to vote or to have its agents vote the shares or other deposited securities as instructed by ADS holders. If we did not ask the depositary to act, you can still send voting instructions and the depositary will endeavor to carry out those instructions, but it is not required to do so.
We cannot assure you that you will receive the voting materials in time to ensure that you can instruct the depositary to vote your shares. In addition, the depositary and its agents are not responsible for failing to carry out voting instructions or for the manner of carrying out voting instructions. This means that you may not be able to exercise your right to vote and there may be nothing you can do if your shares are not voted as you requested.
In order to give you a reasonable opportunity to instruct the depositary as to the exercise of voting rights relating to deposited securities, if we request the depositary to act, we agree to give the depositary notice of any such meeting and details concerning the matters to be voted upon and copies of materials to be made available to ADS holders as far in advance of the meeting date as practicable.
Fees and Expenses of Depositary
What fees and expenses are holders of ADSs responsible for paying?
Pursuant to the terms of the deposit agreement, the holders of ADSs are required to pay the following fees:
|
Persons depositing or withdrawing ordinary shares or |
|
For: |
|
|
|
|
|
$5.00 (or less) per 100 ADSs (or portion of 100 ADSs) |
|
· Issue of ADSs, including issues resulting from a distribution of ordinary shares or rights or other property |
|
|
|
|
|
|
|
· Cancellation of ADSs for the purpose of withdrawal, including if the deposit agreement terminates |
|
Persons depositing or withdrawing ordinary shares or |
|
For: |
|
|
|
|
|
$0.05 (or less) per ADS |
|
· Any cash distribution to you |
|
|
|
|
|
A fee equivalent to the fee that would be payable if securities distributed to you had been ordinary shares and the shares had been deposited for issue of ADSs |
|
· Distribution of securities distributed to holders of deposited securities which are distributed by the depositary to you |
|
|
|
|
|
$0.05 (or less) per ADS per calendar year |
|
· Depositary services |
|
|
|
|
|
Registration or transfer fees |
|
· Transfer and registration of ordinary shares on our share register to or from the name of the depositary or its agent when you deposit or withdraw shares |
|
|
|
|
|
Expenses of the depositary |
|
· Cable, telex and facsimile transmissions (when expressly provided in the deposit agreement)
· Converting foreign currency to U.S. dollars |
|
|
|
|
|
Taxes and other governmental charges the depositary or the custodian have to pay on any ADS or share underlying an ADS, for example, share transfer taxes, stamp duty or withholding taxes |
|
· As necessary |
|
|
|
|
|
Any charges incurred by the depositary or its agents for servicing the deposited securities |
|
· As necessary |
The depositary collects its fees for delivery and surrender of ADSs directly from investors depositing ordinary shares or surrendering ADSs for the purpose of withdrawal or from intermediaries acting for them. The depositary collects fees for making distributions to investors by deducting those fees from the amounts distributed or by selling a portion of distributable property to pay the fees. The depositary may collect its annual fee for depositary services by deduction from cash distributions or by directly billing investors or by charging the book-entry system accounts of participants acting for them. The depositary may collect any of its fees by deduction from any cash distribution payable to ADS holders that are obligated to pay those fees. The depositary may generally refuse to provide for-fee services until its fees for those services are paid.
From time to time, the depositary may make payments to us to reimburse or share revenue from the fees collected from ADS holders, or waive fees and expenses for services provided, generally relating to costs and expenses arising out of establishment and maintenance of the ADS program. In performing its duties under the deposit agreement, the depositary may use brokers, dealers or other service providers that are affiliates of the depositary and that may earn or share fees or commissions.
The depositary may convert currency itself or through any of its affiliates and, in those cases, acts as principal for its own account and not as agent, advisor, broker or fiduciary on behalf of any other person and earns revenue, including, without limitation, transaction spreads, that it will retain for its own account. The revenue is based on, among other things, the difference between the exchange rate assigned to the currency conversion made under the deposit agreement and the rate that the depositary or its affiliate receives when buying or selling foreign currency for its own account. The depositary makes no representation that the exchange rate used or obtained in any currency conversion under the deposit agreement will be the most favorable rate that could be obtained at the time or that the method by which that rate will be determined will be the most favorable to ADS holders, subject to the depositary’s obligations under the deposit agreement. The methodology used to determine exchange rates used in currency conversions is available upon request.
Liability of Owner for Payment of Taxes
You are responsible for any taxes or other governmental charges payable on your ADSs or on the deposited securities represented by any of your ADSs. The depositary may refuse to register any transfer of your ADSs or allow you to
withdraw the deposited securities represented by your ADSs until those taxes or other charges are paid. It may apply payments owed to you or sell deposited securities represented by your American Depositary Shares to pay any taxes owed and you will remain liable for any deficiency. If the depositary sells deposited securities, it will, if appropriate, reduce the number of ADSs to reflect the sale and pay to ADS holders any proceeds, or send to ADS holders any property, remaining after it has paid the taxes.
Tender and Exchange Offers; Redemption, Replacement or Cancellation of Deposited Securities
The depositary will not tender deposited securities in any voluntary tender or exchange offer unless instructed to do by an ADS holder surrendering ADSs and subject to any conditions or procedures the depositary may establish.
If deposited securities are redeemed for cash in a transaction that is mandatory for the depositary as a holder of deposited securities, the depositary will call for surrender of a corresponding number of ADSs and distribute the net redemption money to the holders of called ADSs upon surrender of those ADSs.
If there is any change in the deposited securities such as a sub-division, combination or other reclassification, or any merger, consolidation, recapitalization or reorganization affecting the issuer of deposited securities in which the depositary receives new securities in exchange for or in lieu of the old deposited securities, the depositary will hold those replacement securities as deposited securities under the deposit agreement. However, if the depositary decides it would not be lawful to hold the replacement securities because those securities could not be distributed to ADS holders or for any other reason, the depositary may instead sell the replacement securities and distribute the net proceeds upon surrender of the ADSs.
If there is a replacement of the deposited securities and the depositary will continue to hold the replacement securities, the depositary may distribute new ADSs representing the new deposited securities or ask you to surrender your outstanding ADRs in exchange for new ADRs identifying the new deposited securities.
If there are no deposited securities underlying ADSs, including if the deposited securities are cancelled, or if the deposited securities underlying ADSs have become apparently worthless, the depositary may call for the surrender of those ADSs or cancel those ADSs upon notice to the ADS holders.
Amendment and Termination of the Deposit Agreement
How may the deposit agreement be amended?
We may agree with the depositary to amend the deposit agreement and the ADRs without your consent for any reason. If an amendment adds or increases fees or charges, except for taxes and other governmental charges or expenses of the depositary for registration fees, facsimile costs, delivery charges or similar items, or prejudices a substantial right of ADS holders, it will not become effective for outstanding ADSs until 30 days after the depositary notifies ADS holders of the amendment. At the time an amendment becomes effective, you are considered, by continuing to hold your ADSs, to agree to the amendment and to be bound by the ADRs and the deposit agreement as amended.
How may the deposit agreement be terminated?
The depositary will initiate termination of the deposit agreement if we instruct it to do so. The depositary may initiate termination of the deposit agreement if:
· 60 days have passed since the depositary told us it wants to resign but a successor depositary has not been appointed and accepted its appointment;
· we delist our shares from an exchange on which they were listed and do not list the shares on another exchange;
· we appear to be insolvent or enter insolvency proceedings;
· all or substantially all the value of the deposited securities has been distributed either in cash or in the form of securities;
· there are no deposited securities underlying the ADSs or the underlying deposited securities have become apparently worthless; or
· there has been a replacement of deposited securities.
If the deposit agreement will terminate, the depositary will notify ADS holders at least 90 days before the termination date. At any time after the termination date, the depositary may sell the deposited securities. After that, the depositary will hold the money it received on the sale, as well as any other cash it is holding under the deposit agreement, unsegregated and without liability for interest, for the pro rata benefit of the ADS holders that have not surrendered their ADSs. Normally, the depositary will sell as soon as practicable after the termination date.
After the termination date and before the depositary sells, ADS holders can still surrender their ADSs and receive delivery of deposited securities, except that the depositary may refuse to accept a surrender for the purpose of withdrawing deposited securities if it would interfere with the selling process. The depositary may refuse to accept a surrender for the purpose of withdrawing sale proceeds until all the deposited securities have been sold. The depositary will continue to collect distributions on deposited securities, but, after the termination date, the depositary is not required to register any transfer of ADSs or distribute any dividends or other distributions on deposited securities to the ADS holder (until they surrender their ADSs) or give any notices or perform any other duties under the deposit agreement except as described in this paragraph.
Limitations on Obligations and Liability
Limits on our Obligations and the Obligations of the Depositary; Limits on Liability to Holders of ADSs
The deposit agreement expressly limits our obligations and the obligations of the depositary. It also limits our liability and the liability of the depositary. We and the depositary:
· are only obligated to take the actions specifically set forth in the deposit agreement without negligence or bad faith;
· are not liable if either of us is prevented or delayed by law or circumstances beyond our control from performing our ligations under the deposit agreement;
· are not liable if either of us exercises, or fails to exercise, discretion permitted under the deposit agreement;
· are not liable for the inability of any holder of ADSs to benefit from any distribution on deposited securities that is not made available to holders of ADSs under the terms of the deposit agreement, or for any special, consequential or punitive damages for any breach of the terms of the deposit agreement;
· are not liable for any tax consequences to any holders of ADSs on account of their ownership of ADSs;
· have no obligation to become involved in a lawsuit or other proceeding related to the ADSs or the deposit agreement on your behalf or on behalf of any other person; and
· may rely upon any documents we believe in good faith to be genuine and to have been signed or presented by the proper person.
In the deposit agreement, we and the depositary agree to indemnify each other under certain circumstances. Additionally, we, the depositary and each owner and holder of ADSs, to the fullest extent permitted by applicable law, waive the right to a jury trial in an action against us or the depositary arising out of or relating to the deposit agreement.
Requirements for Depositary Actions
Before the depositary will deliver or register a transfer of an ADS, make a distribution on an ADS, or permit withdrawal of ordinary shares, the depositary may require:
· payment of share transfer or other taxes or other governmental charges and transfer or registration fees charged by third parties for the transfer of any ordinary shares or other deposited securities;
· satisfactory proof of the identity and genuineness of any signature or other information it deems necessary; and
· compliance with regulations it may establish, from time to time, consistent with the deposit agreement, including presentation of transfer documents.
The depositary may refuse to deliver ADSs or register transfers of ADSs generally when the transfer books of the depositary or our transfer books are closed or at any time if the depositary or we think it advisable to do so.
Your Right to Receive the Ordinary Shares Underlying Your ADSs
ADS holders have the right to cancel their ADSs and withdraw the underlying ordinary shares at any time except:
· when temporary delays arise because: (1) the depositary has closed its transfer books or we have closed our transfer books; (2) the transfer of ordinary shares is blocked to permit voting at a shareholders’ meeting; or (3) we are paying a dividend on our ordinary shares;
· when you owe money to pay fees, taxes and similar charges; and
· when it is necessary to prohibit withdrawals in order to comply with any laws or governmental regulations that apply to ADSs or to the withdrawal of ordinary shares or other deposited securities.
This right of withdrawal is not limited by any other provision of the deposit agreement.
Pre-release of ADSs
The deposit agreement permits the depositary to deliver ADSs before deposit of the underlying ordinary shares. This is called a pre-release of the ADSs. The depositary may also deliver ordinary shares upon cancellation of pre-released ADSs (even if the ADSs are canceled before the pre-release transaction has been closed out). A pre-release is closed out as soon as the underlying ordinary shares are delivered to the depositary.
The depositary may receive ADSs instead of ordinary shares to close out a pre-release. The depositary may pre-release ADSs only under the following conditions: (1) before or at the time of the pre-release, the person to whom the pre-release is being made represents to the depositary in writing that it or its customer owns the ordinary shares or ADSs to be deposited; (2) the pre-release is fully collateralized with cash or other collateral that the depositary considers appropriate; and (3) the depositary must be able to close out the pre-release on not more than five business days’ notice. In addition, the depositary will limit the number of ADSs that may be outstanding at any time as a result of prerelease, although the depositary may disregard this limit from time to time if it determines it is appropriate to do so.
Direct Registration System
In the deposit agreement, all parties to the deposit agreement acknowledge that the Direct Registration System, also referred to as DRS, and Profile Modification System, or Profile, will apply to uncertificated ADSs upon acceptance thereof to DRS by DTC. DRS is the system administered by DTC under which the depositary may register the ownership of uncertificated ADSs and such ownership will be evidenced by periodic statements sent by the depositary to the registered holders of uncertificated ADSs. Profile is a required feature of DRS that allows a DTC participant, claiming to act on behalf of a registered holder of ADSs, to direct the depositary to register a transfer of those ADSs
to DTC or its nominee and to deliver those ADSs to the DTC account of that DTC participant without receipt by the depositary of prior authorization from the ADS holder to register that transfer.
In connection with and in accordance with the arrangements and procedures relating to DRS/Profile, the parties to the deposit agreement understand that the depositary will not determine whether the DTC participant that is claiming to be acting on behalf of an ADS holder in requesting registration of transfer and delivery as described in the paragraph above has the actual authority to act on behalf of the ADS holder (notwithstanding any requirements under the Uniform Commercial Code). In the deposit agreement, the parties agree that the depositary’s reliance on and compliance with instructions received by the depositary through the DRS/Profile System and in accordance with the deposit agreement will not constitute negligence or bad faith on the part of the depositary.
Shareholder Communications; Inspection of Register of Holders of ADSs
The depositary will make available for your inspection at its office all communications that it receives from us as a holder of deposited securities that we make generally available to holders of deposited securities. The depositary will send you copies of those communications or otherwise make those communications available to you if we ask it to. You have a right to inspect the register of holders of ADSs, but not for the purpose of contacting those holders about a matter unrelated to our business or the ADSs.
TAXATION
Danish Tax Considerations
The following discussion describes the material Danish tax consequences under present law of an investment in the ADSs (representing our ordinary shares). The summary is for general information only and does not purport to constitute exhaustive tax or legal advice. It is specifically noted that the summary does not address all possible tax consequences relating to an investment in the ADSs. The summary is based solely on the tax laws of Denmark in effect as of December 31, 2019. Danish tax laws may be subject to change, possibly with retroactive effect.
The summary does not cover investors to whom special tax rules apply, and, therefore, may not be relevant, for example, to investors subject to the Danish Tax on Pension Yields Act (i.e., pension savings), professional investors, certain institutional investors, insurance companies, pension companies, banks, stockbrokers, estates and investors with tax liability on return on pension investments. The summary does not cover taxation of individuals and companies who carry on a business of purchasing and selling shares. The summary only sets out the tax position of the direct owners of the ADSs and further assumes that the direct investors are the beneficial owners (as understood in accordance with Danish law) of the ADSs and any dividends thereon. Sales are assumed to be sales to a third party.
Potential investors in the ADSs are advised to consult their tax advisors regarding the applicable tax consequences of acquiring, holding and disposing of the ADSs based on their particular circumstances.
Investors who may be affected by the tax laws of other jurisdictions should consult their tax advisors with respect to the tax consequences applicable to their particular circumstances as such consequences may differ significantly from those described herein.
It is currently subject to uncertainty under Danish tax legislation and case law how listed ADSs are to be treated for Danish tax purposes. For the purpose of the below, it is assumed that a holder of an ADS will be treated as the direct owner of the shares underlying the ADSs and accordingly as the direct shareholder for Danish tax purposes. Thus, the ADSs listed in the U.S. should, for Danish tax purposes, be treated as listed shares since the company’s ordinary shares are admitted to trading on a regulated market.
Taxation of Danish Tax Resident Holders of the ADSs
Sale of the ADSs (Individuals)
Gains from the sale of shares are taxed as share income at a rate of 27% on the first DKK 54,000 (for cohabiting spouses, a total of DKK 108,000) and at a rate of 42% on share income exceeding DKK 54,000 (for cohabiting spouses over DKK 108,000). Such amounts are subject to annual adjustments and include all share income (i.e., all capital gains and dividends derived by the individual or cohabiting spouses, respectively).
Gains and losses on the sale of shares are calculated as the difference between the purchase price and the sales price. The purchase price is generally determined using the average method as a proportionate part of the aggregate purchase price for all the shareholder’s shares in the company.
Losses on listed shares may only be offset against other share income deriving from listed shares, (i.e., received dividends and capital gains on the sale of listed shares) and subject to the Danish Tax Agency having received certain information concerning the ownership of the shares in due time. Unused losses will automatically be offset against a cohabiting spouse’s share income deriving from listed shares and any additional losses can be carried forward and offset against future share income deriving from listed shares.
Sale of the ADSs (Companies)
For the purpose of taxation of sales of shares made by shareholders (companies), a distinction is made between Subsidiary Shares, Group Shares, Tax-Exempt Portfolio Shares and Taxable Portfolio Shares (note that the ownership threshold described below is applied on the basis of the number of all shares issued by the company, and not on the basis of the number of the ADSs issued):
“Subsidiary Shares” is generally defined as shares owned by a shareholder holding at least 10% of the nominal share capital of the issuing company.
“Group Shares” is generally defined as shares in a company in which the shareholder of the company and the issuing company are subject to Danish joint taxation or fulfill the requirements for international joint taxation under Danish law (i.e., the company is controlled by the shareholder).
“Tax-Exempt Portfolio Shares” is defined as shares not admitted to trading on a regulated market owned by a shareholder holding less than 10% of the nominal share capital of the issuing company.
“Taxable Portfolio Shares” is defined as shares that do not qualify as Subsidiary Shares, Group Shares or Tax-Exempt Portfolio Shares, e.g. shares admitted to trading on a regulated market (such as the ADSs) owned by a shareholder holding less than 10% of the nominal share capital of the issuing company.
Gains or losses on disposal of Subsidiary Shares and Group Shares and Tax-Exempt Portfolio Shares are not included in the taxable income of the shareholder.
Special rules apply with respect to Subsidiary Shares and Group Shares to prevent exemption through certain holding company structures just as other anti-avoidance rules may apply. These rules will not be described in further detail.
Capital gains from the sale of Taxable Portfolio Shares admitted to trading on a regulated market are taxable at a rate of 22% irrespective of ownership period. Losses on such shares are generally deductible. Gains and losses on Taxable Portfolio Shares admitted to trading on a regulated market are taxable according to the mark-to-market principle (in Danish “lagerprincippet”).
According to the mark-to-market principle, each year’s taxable gain or loss on Taxable Portfolio Shares is calculated as the difference between the market value of the shares at the beginning of the tax year and the market value of the shares at the end of the tax year. Thus, taxation will take place on an accrual basis even if no shares have been disposed of and no gains or losses have been realized.
If the Taxable Portfolio Shares are sold or otherwise disposed of before the end of the income year, the taxable income of that income year equals the difference between the value of the Taxable Portfolio Shares at the beginning of the income year and the value of the Taxable Portfolio Shares at realization. If the Taxable Portfolio Shares are acquired
and realized in the same income year, the taxable income equals the difference between the acquisition sum and the realization sum. If the Taxable Portfolio Shares are acquired in the income year and not realized in the same income year, the taxable income equals the difference between the acquisition sum and the value of the shares at the end of the income years.
A change of status from Subsidiary Shares/Group Shares/Tax-Exempt Portfolio Shares to Taxable Portfolio Shares (or vice versa) is for tax purposes deemed to be a disposal of the shares and a reacquisition of the shares at market value at the time of change of status.
Special transitional rules apply with respect to the right to offset capital losses realized by the end of the 2009 income year against taxable gains on shares in the 2010 income year or later.
Dividends (Individuals)
Dividends paid to individuals who are tax residents of Denmark are taxed as share income, as described above. All share income must be included when calculating whether the amounts mentioned above are exceeded. Dividends paid to individuals are generally subject to 27% withholding tax.
Dividends (Companies)
Dividends paid on Taxable Portfolio Shares are subject to the standard corporation tax rate of 22% irrespective of ownership period.
The distributing company must withhold tax at a rate of 22%. The withholding tax is considered a prepayment of corporate income tax.
Dividends received on Subsidiary Shares and Group Shares are tax-exempt irrespective of ownership period.
Taxation of Shareholders Residing Outside Denmark
Sale of the ADSs (Individuals and Companies)
Holders of the ADSs not resident in Denmark are normally not subject to Danish taxation on any gains realized on the sale of shares, irrespective of the ownership period, subject to certain anti-avoidance rules seeking to prevent that taxable dividend payments are converted to tax exempt capital gains. If an investor holds the ADSs in connection with a trade or business conducted from a permanent establishment in Denmark, gains on shares may be included in the taxable income of such activities pursuant to the rules applying to Danish tax residents as described above.
Dividends (Individuals)
Under Danish law, dividends paid in respect of shares are generally subject to Danish withholding tax at a rate of 27%. Non-residents of Denmark are not subject to additional Danish income tax in respect to dividends received on shares.
If the withholding tax rate applied is higher than the applicable final tax rate for the shareholder, a request for a refund (clawback) of Danish tax in excess hereof can be made by the shareholder. A refund claim must meet certain requirements, and the Danish Tax Agency has recently published new guidance on the documentation necessary for processing refund claims. The guidance is available in English from the Danish Tax Agency’s website, https://skat.dk/skat.aspx?oId=2244931&vId=0&lang=US. The information on, or information that can be accessed through, such website is not part of and should not be incorporated by reference into this Annual Report on Form 20-F. We have included such website address as an inactive textual reference.
The shareholder can make a request for refund in the following situations:
Double Taxation Treaty
In the event that the shareholder is a resident of a state with which Denmark has entered into a double taxation treaty, the shareholder may generally, through certain certification procedures, seek a refund from the Danish tax authorities of the tax withheld in excess of the applicable treaty rate, which is typically 15%. Denmark has entered into tax treaties with approximately 80 countries, including the United States and almost all members of the European Union. The treaty between Denmark and the United States generally provides for a 15% tax rate.
Lower tax rate applicable to certain investors
If the shareholder holds less than 10% of the nominal share capital (in the form of ordinary shares in the company and not on the basis of the number of the ADSs issued) of the company and the shareholder is tax resident in a state which has a double tax treaty or an international agreement, convention or other administrative agreement on assistance in tax matters according to which the competent authority in the state of the shareholder is obligated to exchange information with Denmark, dividends are subject to tax at a rate of 15%. If the shareholder is tax resident outside the European Union, it is an additional requirement for eligibility for the 15% tax rate that the shareholder together with related shareholders holds less than 10% of the nominal share capital of the company. Note that the reduced tax rate does not affect the withholding rate, why the shareholder must also in this situation claim a refund (clawback) as described above to benefit from the reduced rate.
Where a non-resident of Denmark holds shares, which can be attributed to a permanent establishment in Denmark, dividends are taxable pursuant to the rules applying to Danish tax residents described above.
Dividends (Companies)
Dividends from Subsidiary Shares are exempt from Danish withholding tax provided the taxation of the dividends is to be waived or reduced in accordance with the Parent-Subsidiary Directive (2011/96/EU) or in accordance with a tax treaty with the jurisdiction in which the company investor is tax resident. If Denmark is to reduce taxation of dividends to a foreign company under a tax treaty, Denmark will not—as a matter of domestic law—exercise such right and will in general not impose any withholding tax. Further, dividends from Group Shares—which does not qualify as Subsidiary Shares—are exempt from Danish withholding tax provided the company investor is a resident of the European Union or the EEA and provided the taxation of dividends should have been waived or reduced in accordance with the Parent- Subsidiary Directive (2011/96/EU) or in accordance with a tax treaty with the country in which the company investor is tax resident had the shares been Subsidiary Shares.
Dividend payments on Taxable Portfolio Shares will generally be subject to tax at a rate of 22%.
The Danish withholding tax rate is, however, 27%. Consequently, the foreign shareholder will have a right to request a refund (clawback) of Danish tax in the amount of at least 5% of the total dividend in order to achieve the applicable effective tax rate (22% or lower).
A refund claim must meet certain requirements, and the Danish Tax Agency has recently published new guidance on the documentation necessary for processing refund claims. The guidance is available in English from the Danish Tax Agency’s website, https://skat.dk/skat.aspx?oId=2244931&vId=0&lang=US. The information on, or information that can be accessed through, such website is not part of and should not be incorporated by reference into this Annual Report on Form 20-F. We have included such website address as an inactive textual reference.
Refunds of withheld taxes in excess of the applicable effective tax rate can be requested by the shareholder in the following situations:
Double Taxation Treaty
In the event that the shareholder is a resident of a state with which Denmark has entered into a double taxation treaty, the shareholder may generally, through certain certification procedures, seek a refund from the Danish tax authorities of the tax withheld in excess of the applicable treaty rate, which is typically 15%. Denmark has entered into tax treaties with approximately 80 countries, including the United States and almost all members of the European Union. The treaty between Denmark and the United States generally provides for a 15% rate.
Lower tax rate applicable to certain investors
If the shareholder holds less than 10% of the nominal share capital (in the form of ordinary shares in the company and not on the basis of the number of the ADSs issued) in the company and the shareholder is resident in a jurisdiction which has a double taxation treaty or an international agreement, convention or other administrative agreement on assistance in tax according to which the competent authority in the state of the shareholder is obligated to exchange information with Denmark, dividends are generally subject to a tax rate of 15%. If the shareholder is tax resident outside the European Union, it is an additional requirement for eligibility for the 15% tax rate that the shareholder together with related shareholders holds less than 10% of the nominal share capital of the company. Note that the reduced tax rate does not affect the withholding rate of 27%. Hence, the shareholder must also in this situation claim a refund as described above to benefit from the reduced rate.
Where a non-resident company of Denmark holds shares that can be attributed to a permanent establishment in Denmark, dividends are generally taxable pursuant to the rules applying to Danish tax residents described above.
Share Transfer Tax and Stamp Duties
No Danish share transfer tax or stamp duties are payable on transfer of the shares.
Tax abuse
On January 1, 2019, a new general anti avoidance rule was introduced in Danish tax legislation, which implements the anti-tax avoidance directive’s (EU/2016/1164) Article 6 into the Danish Tax Assessment Act, Section 3. Section 3 provides that an arrangement or series of arrangements (i) not entered into for commercial reasons reflecting the underlying economic reality and (ii) which are implemented for the primary purpose of obtaining, or one of the primary purposes of which is to obtain, a tax benefit which is against the purpose and intent of the Danish tax laws; should be ignored for purposes of calculating the Danish tax liability.
Material U.S. federal income tax consequences to U.S. Holders
The following discussion describes the material U.S. federal income tax consequences to U.S. Holders (as defined below) under present law of an investment in the ADSs. The effects of any applicable state or local laws, or other U.S. federal tax laws such as estate and gift tax laws, the alternative minimum tax or the Medicare contribution tax on net investment income (except to the extent specifically set forth below), are not discussed. This summary applies only to investors who hold the ADSs as capital assets (generally, property held for investment). This discussion is based on the Code, U.S. Treasury regulations promulgated thereunder, judicial decisions, published rulings and administrative pronouncements of the U.S. Internal Revenue Service, or the IRS, and the income tax treaty between the United States and Denmark, or the Treaty, all as in effect as of December 31, 2019. All of the foregoing authorities are subject to change, which change could apply retroactively and could affect the tax consequences described below. We have not requested a ruling from the U.S. Internal Revenue Service, or the IRS with respect to the statements made and the conclusions reached in the following summary, and there can be no assurance that the IRS or a court will agree with such statements and conclusions. Accordingly, investors should consult their own tax advisors concerning the U.S. federal, state, local and non-U.S. tax consequences of acquiring, owning and disposing of our ordinary shares or ADSs in their particular circumstances.
The following discussion does not address all U.S. federal income tax consequences relevant to a beneficial owner’s particular circumstances or to beneficial owners subject to particular rules, including:
· U.S. expatriates and certain former citizens or long-term residents of the United States;
· persons whose functional currency for U.S. federal income tax purposes is not the U.S. dollar;
· persons holding the ADSs as part of a hedge, straddle or other risk reduction strategy or as part of a conversion transaction or other integrated investment;
· banks, insurance companies and other financial institutions;
· real estate investment trusts or regulated investment companies;
· brokers, dealers or traders in securities, commodities or currencies who use a mark-to-market method of tax accounting;
· S corporations, partnerships, or other entities or arrangements treated as partnerships for U.S. federal income tax purposes;
· tax-exempt organizations or governmental organizations;
· persons who acquired the ADSs pursuant to the exercise of any employee share option or otherwise as compensation;
· persons subject to special tax accounting rules as a result of any item of gross income with respect to the ADSs being taken into account in an “applicable financial statement” (as defined in the Code);
· persons that own or are deemed to own 10% or more of our equity by vote or value;
· persons that hold their ADSs through a permanent establishment or fixed base outside the United States; and
· persons deemed to sell the ADSs under the constructive sale provisions of the Code.
U.S. HOLDERS ARE URGED TO CONSULT THEIR TAX ADVISORS REGARDING THE APPLICATION OF THE U.S. FEDERAL TAX RULES TO THEIR PARTICULAR CIRCUMSTANCES AS WELL AS THE U.S. STATE AND LOCAL AND NON-U.S. TAX CONSEQUENCES TO THEM OF THE PURCHASE, OWNERSHIP AND DISPOSITION OF THE ADSs.
For purposes of this discussion, a “U.S. Holder” is a beneficial owner of the ADSs that, for U.S. federal income tax purposes, is or is treated as any of the following:
· an individual who is a citizen or resident of the United States;
· a corporation, or other entity taxable as a corporation, created or organized in or under the laws of the United States, any state therein, or the District of Columbia;
· an estate, the income of which is subject to U.S. federal income tax regardless of its source; or
· a trust that (1) is subject to the primary supervision of a U.S. court and all substantial decisions of which are under the control of one or more “United States persons” (within the meaning of Section 7701(a)(30) of the Code), or (2) has a valid election in effect to be treated as a United States person for U.S. federal income tax purposes.
If you are a partner in a partnership (or other entity taxable as a partnership for U.S. federal income tax purposes) that holds the ADSs, your tax treatment generally will depend on your status and the activities of the partnership. Partnerships holding the ADSs and the partners in such partnerships should consult their tax advisors regarding the U.S. federal income tax consequences applicable to them.
The discussion below assumes that the representations contained in the deposit agreement are true and that the obligations in the deposit agreement and any related agreement will be complied with in accordance with their terms. Generally, a U.S. Holder of an ADS should be treated for U.S. federal income tax purposes as holding the ordinary shares represented by the ADS. Accordingly, no gain or loss will be recognized upon an exchange of ADSs for ordinary shares. The U.S. Treasury has expressed concerns that intermediaries in the chain of ownership between the holder of an ADS and the issuer of the security underlying the ADS may be taking actions that are inconsistent with
the beneficial ownership of the underlying security. Accordingly the creditability of foreign taxes, if any, as described below, could be affected by actions taken by intermediaries in the chain of ownership between a U.S. Holder and our company if as a result of such actions the U.S. Holder is not properly treated as the beneficial owner of underlying ordinary shares.
Taxation of dividends and other distributions on the ADSs
Subject to the PFIC rules discussed below, the gross amount of any distribution to you with respect to the ADSs will be included in your gross income as dividend income when actually or constructively received to the extent of your pro rata share of our current or accumulated earnings and profits (as determined under U.S. federal income tax principles). To the extent the amount of the distribution exceeds your pro rata share of our current and accumulated earnings and profits, it will be treated first as a return of your tax basis in the ADSs, and to the extent the amount of the distribution exceeds your tax basis, the excess will be taxed as capital gain. However, we do not intend to calculate our earnings and profits under U.S. federal income tax principles. Therefore, you should expect a distribution will generally be reported as ordinary dividend income for such purposes. Any dividends will not be eligible for the dividends-received deduction allowed to corporations in respect of dividends received from other U.S. corporations.
If we are eligible for benefits under the Treaty, dividends a U.S. Holder receives from us generally will be “qualified dividend income.” If certain holding period and other requirements are met, including a requirement that we are not a PFIC in the year of the dividend or the immediately preceding year, qualified dividend income of an individual or other non-corporate U.S. Holder generally will be subject to preferential tax rates. You should consult your tax advisor regarding the availability of these preferential tax rates under your particular circumstances.
As discussed in “Taxation—Danish Tax Considerations,” payments of dividends by us may be subject to Danish withholding tax. The rate of withholding tax applicable to U.S. Holders that are eligible for benefits under the Treaty is reduced to a maximum of 15%. For U.S. federal income tax purposes, U.S. Holders will be treated as having received the amount of Danish taxes withheld by us, and as then having paid over the withheld taxes to the Danish taxing authorities. As a result of this rule, the amount of dividend income included in gross income for U.S. federal income tax purposes by a U.S. Holder with respect to a payment of dividends may be greater than the amount of cash actually received (or receivable) by the U.S. Holder from us with respect to the payment.
Dividends will generally constitute foreign source income for foreign tax credit limitation purposes. Subject to the discussion of the PFIC rules below, any tax withheld with respect to distributions on the ADSs at the rate applicable to a U.S. Holder may, subject to a number of complex limitations, be claimed as a foreign tax credit against such U.S. Holder’s U.S. federal income tax liability or may be claimed as a deduction for U.S. federal income tax purposes. The limitation on foreign taxes eligible for credit is calculated separately with respect to specific classes of income. For this purpose, dividends distributed by us with respect to the ADSs generally will constitute “passive category income.” The rules with respect to the foreign tax credit are complex and involve the application of rules that depend upon a U.S. Holder’s particular circumstances. You are urged to consult your tax advisor regarding the availability of the foreign tax credit under your particular circumstances.
In general, the amount of a distribution paid to a U.S. Holder in a foreign currency will be the U.S. dollar value of the foreign currency calculated by reference to the spot exchange rate on the day the depositary receives the distribution, in the case of the ADSs, or on the day the distribution is received by the U.S. Holder, in the case of ordinary shares, regardless of whether the foreign currency is converted into U.S. dollars at that time. Any foreign currency gain or loss a U.S. Holder realizes on a subsequent conversion of foreign currency into U.S. dollars will be U.S. source ordinary income or loss. If dividends received in a foreign currency are converted into U.S. dollars on the day they are received, a U.S. Holder should not be required to recognize foreign currency gain or loss in respect of the dividend.
Taxation of disposition of the ADSs
Subject to the PFIC rules discussed below, you will recognize gain or loss on any sale, exchange or other taxable disposition of an ADS equal to the difference between the amount realized (in U.S. dollars) on the disposition of the ADS and your adjusted tax basis (in U.S. dollars) in the ADS. The adjusted tax basis in your ordinary shares or ADSs generally will be equal to the cost of such ordinary shares or ADSs. Any such gain or loss generally will be capital gain or loss, and generally will be long-term capital gain or loss if you have held the ADS for more than one year at
the time of sale, exchange or other taxable disposition. Otherwise, such gain or loss generally will be short-term capital gain or loss. Long-term capital gains recognized by certain non-corporate U.S. Holders, including individuals, generally will be taxable at a reduced rate. The deductibility of capital losses is subject to limitations. Any such gain or loss you recognize generally will be treated as U.S. source income or loss for foreign tax credit limitation purposes. You should consult your tax advisor regarding the proper treatment of gain or loss in your particular circumstances.
Passive foreign investment company
The application of the PFIC rules is subject to uncertainty in several respects. Whether we will be a PFIC in any year depends on the composition of our income and assets, our market capitalization, and the relative fair market value of our assets from time to time, which we expect may vary substantially over time. Among other things, because (i) we currently own a significant amount of passive assets, including cash, and (ii) the value of our assets (including our intangible assets) that generate non-passive income for PFIC purposes is uncertain and may vary substantially over time, we cannot assure you we will not be a PFIC for our current taxable year or for any future taxable year. Based on our analysis of our income, assets, activities and market capitalization for our taxable year ended December 31, 2019, we believe that we were not classified as a PFIC for the taxable year ended December 31, 2019. However, because our PFIC status is subject to a number of uncertainties, our financials for the year ending December 31, 2019 have not yet been finalized or audited, it is very early in the year and we have very limited financial information for our current taxable year, neither we nor our tax advisors can provide any assurances that we will not be considered a PFIC in the prior, current, or any future taxable year and our PFIC status may change from year to year. In particular, we have not yet made any determination as to our expected PFIC status for the current year, and any such expectation would be subject to change based on, among other factors, our use of cash, the source and nature of our income, and the price of our ordinary shares or ADSs. Accordingly, we may be a PFIC for the taxable year ending December 31, 2020, or any future taxable year.
A non-U.S. corporation is considered a PFIC for any taxable year if either:
· at least 75% of its gross income for such taxable year is passive income, or
· at least 50% of the value of its assets (based on an average of the quarterly values of the assets during a taxable year) is attributable to assets that produce or are held for the production of passive income.
For purposes of the above calculations, if a non-U.S. corporation owns, directly or indirectly, 25% or more of the total value of the outstanding shares of another corporation, it will be treated as if it (a) held a proportionate share of the assets of such other corporation and (b) received directly a proportionate share of the income of such other corporation. Passive income generally includes dividends, interest, rents, royalties and capital gains, but generally excludes rents and royalties which are derived in the active conduct of a trade or business and which are received from a person other than a related person.
A separate determination must be made each taxable year as to whether we are a PFIC (after the close of each such taxable year). The determination of whether we are a PFIC is a fact-intensive determination made on an annual basis and the applicable law is subject to varying interpretation. Because the value of our assets for purposes of the asset test will generally be determined by reference to the market price of the ADSs, our PFIC status will depend in large part on the market price of the ADSs, which may fluctuate significantly. In addition, changes in the composition of our income or assets may cause us to become a PFIC.
If we are a PFIC for any year during which you hold the ADSs, we generally will continue to be treated as a PFIC with respect to you for all succeeding years during which you hold the ADSs (regardless of whether we continue to meet the tests described above), unless we cease to be a PFIC and you make a “deemed sale” election with respect to the ADSs you hold. If such election is made, you will be deemed to have sold the ADSs you hold at their fair market value on the last day of the last taxable year in which we qualified as a PFIC, and any gain from such deemed sale would be subject to the consequences described below. After the deemed sale election, the ADSs with respect to which the deemed sale election was made will not be treated as shares in a PFIC unless we subsequently again become a PFIC.
For each taxable year we are treated as a PFIC with respect to you, you will be subject to special tax rules with respect to any “excess distribution” (as defined below) you receive and any gain you realize from a sale or other disposition (including a pledge) of the ADSs, unless you make a “mark-to-market” election as discussed below. Distributions you receive in a taxable year that are greater than 125% of the average annual distributions you received during the shorter of the three preceding taxable years or your holding period for the ADSs will be treated as an “excess distribution.” Under these special tax rules, if you receive any “excess distribution” or realize any gain from a sale or other disposition of the ADSs:
· the “excess distribution” or gain will be allocated ratably over your holding period for the ADSs,
· the amount allocated to the current taxable year, and any taxable year before the first taxable year in which we were a PFIC, will be treated as ordinary income, and
· the amount allocated to each other year will be subject to the highest tax rate in effect for that year and the interest charge generally applicable to underpayments of tax will be imposed on the resulting tax attributable to each such year.
Gains (but not losses) realized on the sale of the ADSs cannot be treated as capital, even if you hold the ADSs as capital assets.
If we are treated as a PFIC with respect to you for any taxable year, to the extent any of our subsidiaries are also PFICs, you will be deemed to own your proportionate share of any such lower-tier PFIC, and you may be subject to the rules described in the preceding two paragraphs with respect to the shares of such lower-tier PFICs you would be deemed to own. As a result, you may incur liability for any “excess distribution” described above if we receive a distribution from such lower-tier PFICs or if any shares in such lower-tier PFICs are disposed of (or deemed disposed of). You should consult your tax advisor regarding the application of the PFIC rules to any of our subsidiaries.
Alternatively, a U.S. Holder of “marketable stock” (as defined below) in a PFIC may make a “mark-to-market” election for such stock to elect out of the general tax treatment for PFICs discussed above. If you make a “mark-to-market” election for the ADSs, you will include in income for each year we are a PFIC an amount equal to the excess, if any, of the fair market value of the ADSs as of the close of your taxable year over your adjusted basis in such ADSs. You are allowed a deduction for the excess, if any, of the adjusted basis of the ADSs over their fair market value as of the close of the taxable year. However, deductions are allowable only to the extent of any net “mark-to-market” gains on the ADSs included in your income for prior taxable years. Amounts included in your income under a “mark-to-market” election, as well as gain on the actual sale or other disposition of the ADSs, are treated as ordinary income. Ordinary loss treatment also applies to the deductible portion of any “mark-to-market” loss on the ADSs, as well as to any loss realized on the actual sale or disposition of the ADSs to the extent the amount of such loss does not exceed the net “mark-to-market” gains previously included for the ADSs. Your basis in the ADSs will be adjusted to reflect any such income or loss amounts. If you make a valid “mark-to-market” election, the tax rules that apply to distributions by corporations that are not PFICs would apply to distributions by us, except the lower applicable tax rate for qualified dividend income would not apply. If we cease to be a PFIC when you have a “mark-to-market” election in effect, gain or loss realized by you on the sale of the ADSs will be a capital gain or loss and taxed in the manner described above under “Taxation of disposition of the ADSs.”
The “mark-to-market” election is available only for “marketable stock,” which is stock that is traded in other than de minimis quantities on at least 15 days during each calendar quarter, or regularly traded, on a qualified exchange or other market, as defined in applicable U.S. Treasury regulations. Any trades that have as their principal purpose meeting this requirement will be disregarded. The ADSs are listed on the Nasdaq Global Select Market and, accordingly, provided the ADSs are regularly traded, the “mark-to-market” election would be available to you if we are a PFIC and you are a holder of ADSs. Once made, the election cannot be revoked without the consent of the IRS unless the ADSs cease to be “marketable stock.” If we are a PFIC for any year in which the U.S. Holder owns ADSs but before a “mark-to-market” election is made, the interest charge rules described above will apply to any “mark-to-market” gain recognized in the year the election is made. If any of our subsidiaries are or become PFICs, the “mark-to-market” election will not be available with respect to the shares of such subsidiaries that are treated as owned by you. Consequently, you could be subject to the PFIC rules with respect to income of the lower-tier PFICs the value of which already had been taken into account indirectly via “mark-to-market” adjustments. A U.S. Holder should consult
its tax advisors as to the availability and desirability of a “mark-to-market” election, as well as the impact of such election on interests in any lower-tier PFICs.
In certain circumstances, a U.S. Holder of stock in a PFIC can make a “qualified electing fund election” to mitigate some of the adverse tax consequences of holding stock in a PFIC by including in income its share of the corporation’s income on a current basis. However, we do not currently intend to prepare or provide the information that would enable you to make a “qualified electing fund election.”
Unless otherwise provided by the U.S. Treasury, each U.S. shareholder of a PFIC is required to file an annual report containing such information as the U.S. Treasury may require. A U.S. Holder’s failure to file the annual report will cause the statute of limitations for such U.S. Holder’s U.S. federal income tax return to remain open with regard to the items required to be included in such report until three years after the U.S. Holder files the annual report, and, unless such failure is due to reasonable cause and not willful neglect, the statute of limitations for the U.S. Holder’s entire U.S. federal income tax return will remain open during such period. U.S. Holders should consult their tax advisors regarding the requirements of filing such information returns under these rules, taking into account the uncertainty as to whether we are currently treated as or may become a PFIC.
THE U.S. FEDERAL INCOME TAX RULES RELATING TO PFICS ARE COMPLEX. PROSPECTIVE U.S. INVESTORS ARE URGED TO CONSULT THEIR OWN TAX ADVISORS WITH RESPECT TO THE ACQUISITION, OWNERSHIP AND DISPOSITION OF OUR ORDINARY SHARES OR ADSS, THE CONSEQUENCES TO THEM OF AN INVESTMENT IN A PFIC, ANY ELECTIONS AVAILABLE WITH RESPECT TO OUR ORDINARY SHARES OR ADSS AND THE IRS INFORMATION REPORTING OBLIGATIONS WITH RESPECT TO THE ACQUISITION, OWNERSHIP AND DISPOSITION OF OUR ORDINARY SHARES OR ADSS.
Medicare contribution tax on net investment income
Certain U.S. Holders that are individuals, estates or trusts and whose income exceeds certain thresholds generally are subject to a 3.8% tax on all or a portion of their net investment income, which may include their gross dividend income and net gains from the disposition of ADSs. If you are a “United States person” that is an individual, estate or trust, you are encouraged to consult your tax advisors regarding the applicability of this Medicare tax to your income and gains in respect of your investment in ADSs.
Information reporting and backup withholding
Dividend payments with respect to the ADSs and proceeds from the sale, exchange or other disposition of the ADSs may be subject to information reporting to the IRS and U.S. backup withholding. Certain U.S. Holders are exempt from backup withholding, including corporations and certain tax-exempt organizations. A U.S. Holder will be subject to backup withholding if such holder is not otherwise exempt and such holder:
· fails to furnish the holder’s taxpayer identification number, which for an individual is ordinarily his or her social security number;
· furnishes an incorrect taxpayer identification number;
· is notified by the IRS that the holder previously failed to properly report payments of interest or dividends; or
· fails to certify under penalties of perjury that the holder has furnished a correct taxpayer identification number and that the IRS has not notified the holder that the holder is subject to backup withholding.
Backup withholding is not an additional tax. Any amounts withheld under the backup withholding rules may be allowed as a refund or a credit against the U.S. Holder’s U.S. federal income tax liability, provided the required information is timely furnished to the IRS. U.S. Holders should consult their tax advisors regarding their qualification for an exemption from backup withholding and the procedures for obtaining such an exemption.
Certain Reporting Requirements
U.S. Holders paying more than $100,000 for our ordinary shares or ADSs generally may be required to file IRS Form 926 reporting the payment of the offer price for our ordinary shares or ADSs to us. Substantial penalties may be imposed upon a U.S. Holder that fails to comply. Each U.S. Holder should consult its own tax advisor as to the possible obligation to file IRS Form 926.
Foreign Asset Reporting
Certain individual U.S. Holders are required to report information relating to an interest in our ordinary shares or ADSs, subject to certain exceptions (including an exception for shares held in accounts maintained by U.S. financial institutions) by filing IRS Form 8938 (Statement of Specified Foreign Financial Assets) with their federal income tax return. U.S. Holders are urged to consult their tax advisors regarding their information reporting obligations, if any, with respect to their ownership and disposition of our ordinary shares or ADSs.
Exhibit 4.1
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
EXECUTION DRAFT
COLLABORATIVE RESEARCH AND LICENSE AGREEMENT
between
Alexion Pharma Holding Unlimited Company
and
Zealand Pharma A/S.
Dated March 20, 2019
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
COLLABORATIVE RESEARCH AND LICENSE AGREEMENT
THIS COLLABORATIVE RESEARCH AND LICENSE AGREEMENT (the “Agreement”), effective as of March 20, 2019 (the “Effective Date”), is entered into by and between Zealand Pharma A/S, a public limited liability company organized under the laws of Denmark having a place of business at Smedland 36, 2600 Glostrup, Copenhagen, Denmark (“ZEALAND”) and Alexion Pharma Holding Unlimited Company, an unlimited liability company incorporated under the laws of Ireland with a principal place of business at 22 Victoria Street, Hamilton HM 12 Bermuda (“ALEXION”, collectively with ZEALAND, the “Parties” and each, a “Party”)
RECITALS
WHEREAS, ZEALAND has developed and owns or has rights to certain patents and technology relating to the discovery, design, and development of peptide-based drug candidates and methods of making and using the same;
WHEREAS, ALEXION is engaged in, among other things, discovery, clinical Development and Commercialization of pharmaceutical products and has expertise in diseases involving the Complement Pathway (as defined below);
WHEREAS, ALEXION and ZEALAND are interested in collaborating to perform research and development activities to identify Candidate Products for targets in the Complement Pathway (each as defined below), which Candidate Products, once selected, would be further developed by the Parties and commercialized by ALEXION, in accordance with the terms and conditions set forth in this Agreement; and
WHEREAS, ALEXION desires to obtain from ZEALAND the rights and licenses set forth herein, and ZEALAND desires to grant such rights and licenses to ALEXION, including exclusive worldwide licenses, all on the terms and conditions set forth in this Agreement.
NOW, THEREFORE, the Parties hereto agree as follows:
1. DEFINITIONS
1.1 “Acceptance” means: (a) with respect to an NDA, receipt of a written communication from the applicable Regulatory Authority acknowledging that it has received such Regulatory Approval Application and that such Regulatory Approval Application is sufficiently complete to permit a substantive review for approval purposes; or (b) with respect to an IND, receipt of a written notice or other confirmation in accordance with Applicable Law that the IND is valid and effective and a Phase 1 clinical study may be commenced. For clarity, a clinical trial authorisation issued by a competent authority in a member state in the European Union (such expression shall include the United Kingdom) would constitute a confirmation that the Phase 1 clinical study may be commenced.
1.2 “Acquirer” has the meaning set forth in Section 13.1.
1.3 “Additional Target” has the meaning set forth in Section 2.5.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
1.4 “Additional Target Notice” has the meaning set forth in Section 2.5.
1.5 “Additional Target Trigger Date” has the meaning set forth in Section 2.5.
1.6 “Affiliates” means, with respect to a Party or Third Party, any company or business or entity controlled by, controlling, or under common control with such Party or Third Party. For the purpose of this definition, “control” means direct or indirect beneficial ownership of at least fifty percent (50%) interest in the voting stock (or the equivalent) of such person or entity or having the right to direct, appoint or remove a majority or more of the members of its board of directors (or their equivalent), or having the power to control the general management of such person or entity, by contract, law or otherwise.
1.7 “Agreement Patent Rights” has the meaning set forth in Section 9.3.1.
1.8 “ALEXION Background Intellectual Property” means (a) any [***] that (i) is [***] under this Agreement and (ii) is [***] and (b) any [***] in (a). For the avoidance of doubt, the list of Complement Pathway Targets set forth on APPENDIX 2 hereto shall, except to the extent it falls within the exemptions set out in Section 8.4, be deemed to be ALEXION Background Intellectual Property, and shall be subject to the confidentiality and non-use obligations in Article 8.
1.9 “ALEXION Indemnitees” has the meaning set forth in Section 11.1.
1.10 “Applicable Law” means all applicable laws, rules and regulations (including any rules, regulations, guidelines or other requirements of the Regulatory Authorities or other governmental agency) that may be in effect from time to time.
1.11 “Auditor” has the meaning set forth in Section 6.13.1.
1.12 “Board of Directors” has the meaning set forth in Section 1.21(i).
1.13 “Business Day” means any day other than (i) Saturday, (ii) Sunday or (iii) any day on which commercial banks in Boston, MA USA or Copenhagen, Denmark are authorized or required by law to close.
1.14 “Calendar Quarter” means a period of three calendar months ending on March 31st, June 30th, September 30th or December 31st in any Calendar Year; provided that the first Calendar Quarter of the Term shall begin on the Effective Date and end on the last day of the then current Calendar Quarter and the last Calendar Quarter of the Term shall begin on the first day of such Calendar Quarter and end on the last day of the Term.
1.15 “Calendar Year” means a one-year period beginning on January 1st and ending on December 31st; provided that the first Calendar Year of the Term shall begin on the Effective Date and end on December 31 of the then current Calendar Year and the last Calendar Year of the Term shall begin on the first day of such Calendar Year and end on the last day of the Term.
1.16 “Candidate Product(s)” means, on a Target-by-Target basis, the specific molecule(s) existing as of the Effective Date (including any Existing Lead Molecules) or identified by ZEALAND (including through any permitted subcontractor or sublicensee) through the conduct of the
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Discovery/Development Program that (i) meet the Candidate Product Criteria set forth in the relevant Development Plan or (ii) [***] that are otherwise designated by ALEXION in writing to be a “Candidate Product”.
1.17 “Candidate Product Criteria” means, with respect to a Target, the success criteria agreed upon by the Parties for the molecules directed to such Target as set forth in the applicable Discovery Plan.
1.18 “C.F.R.” means the U.S. Code of Federal Regulations.
1.19 “cGCP” means all applicable good clinical practice laws, regulations, and standards related to the design, conduct, performance, monitoring, auditing, recording, analyses and reporting of Clinical Trials, including, as applicable (a) as set forth in the International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use Harmonized Tripartite Guideline for Good Clinical Practice (ICH E6 R2) and any other guidelines related to good clinical practice for trials on medicinal products in the Territory, (b) the Declaration of Helsinki (2004) as last amended at the 52nd World Medical Association in October 2000 and any further amendments or clarifications thereto, (c) 21 C.F.R. Parts 50 (Protection of Human Subjects), 56 (Institutional Review Boards), and 312 (Investigational New Drug Application), and 42 C.F.R. Part 45 (Protection of Human Subjects or the Common Rule), and (d) the equivalent Applicable Law in the region in the Territory, each as applicable from time to time and in each case, that provide for, among other things, assurance that the clinical data and reported results are credible and accurate and protect the rights, integrity, and confidentiality of trial subjects.
1.20 “cGMP” means all applicable current good manufacturing practice requirements, standards and guidelines, including, as applicable, (a) the U.S. regulations related to current good manufacturing practices for drugs, biologics, devices and combination products, including 21 C.F.R. Parts 4, 210, 211, 312, 600-680, and 820 and related guidance documents, (b) European Directive 2003/94/EC and Eudralex 4, (c) the principles detailed in the International Conference on Harmonization’s Q7 guidelines, and (d) the equivalent Applicable Law in any relevant country or region, each as may be applicable from time to time.
1.21 “Change of Control” means, with respect to a Party, the occurrence of any of the following after the Effective Date:
i. any “person” or “group” (as such terms are defined below): (a) is or becomes the “beneficial owner” (as defined below), directly or indirectly, of shares of capital stock or other interests (including partnership interests) of such Party then outstanding and normally entitled (without regard to the occurrence of any contingency) to vote in the election of the directors, managers or similar supervisory positions (“Voting Stock”) of such Party representing fifty percent (50%) or more of the total voting power of all outstanding classes of Voting Stock of such Party; or (b) has the power, directly or indirectly, to elect a majority of the members of the Party’s board of directors, or similar governing body (“Board of Directors”); or
ii. such Party enters into a merger, consolidation or similar transaction with another Person (whether or not such Party is the surviving entity) and as a result of such
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
merger, consolidation or similar transaction (a) the members of the Board of Directors of such Party immediately prior to such transaction constitute less than a majority of the members of the Board of Directors of such Party or such surviving Person immediately following such transaction; or (b) the Persons that beneficially owned, directly or indirectly, the shares of Voting Stock of such Party immediately prior to such transaction cease to beneficially own, directly or indirectly, shares of Voting Stock of such Party representing at least a majority of the total voting power of all outstanding classes of Voting Stock of the surviving Person; or
iii. such Party sells or transfers to any Third Party, in one or more related transactions, properties or assets representing all or substantially all of such Party’s consolidated total assets to which this Agreement relates; or
iv. the holders of capital stock of such Party approve a plan or proposal for the liquidation or dissolution of such Party.
For the purpose of this definition of “Change of Control,” (a) “person” and “group” have the meanings given such terms under Section 13(d) and 14(d) of the United States Securities Exchange Act of 1934 and the term “group” includes any group acting for the purpose of acquiring, holding or disposing of securities within the meaning of Rule 13d-5(b)(1) under the said Act; (b) a “beneficial owner” will be determined in accordance with Rule 13d-3 under the aforesaid Act; and (c) the terms “beneficially owned” and “beneficially own” will have meanings correlative to that of “beneficial owner.”
1.22 “Clinical Trial” means a clinical trial in humans of a product, including a Phase I Clinical Trial, a Phase II Clinical Trial or a Pivotal Clinical Trial.
1.23 “CMO” means a Third Party contract manufacturer.
1.24 “CMO Supply Agreement” has the meaning set forth in Section 5.1.
1.25 “Collaboration IP” means any Intellectual Property that is [***] (a) [***], or (b) [***], in each case under clauses (a) and (b) [***] under this Agreement; provided that any such Intellectual Property that is [***].
1.26 “Combination Product” means (a) any single product in finished form containing as active ingredients both a Product and one or more other pharmaceutically active compounds or substances, whether co-formulated or co-packaged (i.e., within a single box or sales unit); or (b) any Product sold in combination with one or more other products (such as devices) or services for a single invoice price; or (c) any Product sold where the sale of the Product is only available with the purchase of other products or services (such other pharmaceutically active compounds or substances, or such other products (such as devices) or services referred to in clauses (a) through (c) hereof, the “Other Components”).
1.27 “Commercialization”, “Commercializing” or “Commercialize” means any and all activities related to the pre-marketing, launching, marketing, promotion (including advertising and detailing), labeling, pricing, distribution, storage, handling, offering for sale, selling, having sold, importing and exporting for sale, having imported and exported for sale, distribution, having
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
distributed, customer service and support, and post-marketing safety surveillance and reporting of a product (including a Product), but not including Development or Manufacturing.
1.28 “Commercially Reasonable Efforts” means, with respect to [***], that level of efforts and resources [***], taking into account, as applicable, [***], and all other relevant factors. The [***] set forth in the immediately preceding sentence shall in no event require [***]. Commercially Reasonable Efforts shall be determined [***].
1.29 “Competitor” means any [***] company with [***].
1.30 “Complement Pathway” means any [***], including the [***] pathways.
1.31 “Complement Pathway Target(s)” means the Lead Target and such other Complement Pathway targets listed on APPENDIX 2.
1.32 “Confidential Information” has the meaning as defined in Section 8.1.
1.33 “Control” or “Controlled” means, with respect to any Intellectual Property, the possession by a Party or any of its Affiliates, whether by ownership or license (other than by a license granted under this Agreement), of the ability to grant to the other Party access to or ownership of such Intellectual Property, or, a license or a sublicense in, to or under such Intellectual Property, as provided herein, without requiring the consent of a Third Party or violating the terms of any agreement or other arrangement with any Third Party. A Party shall not be deemed to Control any Intellectual Property that is owned or controlled by an Acquirer, except to the extent that any such Patent Rights or Know-How were developed in the course of such Party’s or such Acquirer’s performance of activities under this Agreement or through the use or application of the other Party’s Intellectual Property or Confidential Information.
1.34 “Cover”, “Covering” or “Covered” means, with respect to a product, technology, process, method or mode of administration that, in the absence of ownership of or a license granted under a particular Patent Right, the Manufacture, use, offer for sale, sale or importation of such product or composition of matter or the practice of such technology, process, method or mode of administration would infringe such Patent Right.
1.35 “CPI” means the Consumer Price Index as published by Statistics Denmark from time to time.
1.36 “Created Lead Molecule” means any peptide or other molecule that is created and identified by or on behalf of ZEALAND or its Affiliates (including through any permitted subcontractor or sublicensee) through conduct of the Lead Discovery/Development Program to have or that is intended to have pharmaceutically useful binding to or modulation of the Lead Target.
1.37 “Created Lead Product” means any pharmaceutical composition in any formulation or for any route of administration, and in any dosage strength or presentation that is, contains or consists of one or more Candidate Products that are Created Lead Molecules whether alone or with other active ingredients but excluding any Existing Lead Molecules.
1.38 “Damages” has the meaning set forth in Section 11.1.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
1.39 “Debarred” means, with respect to an individual or entity, that such individual or entity has been debarred or suspended under 21 U.S.C. §335(a) or (b), the subject of a conviction described in 21 U.S.C. §335a, disqualified as a clinical investigator under 21 C.F.R. §§312.70 or 812.119, disqualified as a testing facility under 21 C.F.R. §58.202, excluded from a federal or governmental health care program, debarred from federal contracting, convicted of or pled nolo contendere to any felony, or to any federal or state legal violation (including misdemeanors) relating to prescription drug products or fraud, the subject to OFAC sanctions or on the OFAC list of specially designated nationals, or the subject of any similar sanction of any governmental authority in the Territory.
1.40 “Defaulting Party” has the meaning set forth in Section 12.3.
1.41 “Deliverables” means any and all deliverables to be generated or provided by ZEALAND in connection with (i) the Discovery/Development Activities conducted by ZEALAND under the Discovery Plan and any Development Plans of a Discovery/Development Program, as specified therein or (ii) the [***] Work Plan as specified therein.
1.42 “Develop” or “Development” means non-clinical and clinical drug research and development activities, whether before or after Regulatory Approval, including drug metabolism and pharmacokinetics, screening, discovery, synthesis, translational research, toxicology, pharmacology, test method development and stability testing, process and packaging development and improvement, process validation, process scale-up, formulation development, delivery system development, quality assurance and quality control development, statistical analysis, conduct of Clinical Trials, regulatory affairs, the preparation and submission of regulatory filings, Clinical Trial regulatory activities, and any other activities directed towards obtaining or maintaining Regulatory Approval of any product (including a Product). Development includes use and importation of the relevant product to conduct such Discovery/Development Activities. Development will not include Commercialization activities.
1.43 “Development Decision Notice” means, with respect to one or more peptides or molecules, a written notice provided by ALEXION to ZEALAND notifying ZEALAND as to whether ALEXION has designated such peptide(s) or molecule(s) as a Candidate Product for further Development under a Development Plan of the applicable Discovery/Development Program as set forth in Section 2.2.4.
1.44 “Development Milestone Event” has the meaning set forth in Section 6.3.1.
1.45 “Development Milestone Payment” has the meaning set forth in Section 6.3.1.
1.46 “Development Plan” has the meaning set forth in Section 2.1.2(b).
1.47 “Development Plan Term” has the meaning set forth in Section 2.1.7.
1.48 “Diligent Efforts” means, with respect to [***], that level of efforts and resources [***] in order to [***], except to the extent of any [***].
1.49 “Disclosing Party” has the meaning set forth in Section 8.1.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
1.50 “Discontinued Product” means any Candidate Product, Existing Lead Product, Created Lead Product, or Optioned Product (in each case, other than one directed to a Target that is the subject of any Discovery/Development Program or is being Developed, Commercialized, or Manufactured by or on behalf of ALEXION, its Affiliates or its Sublicensees): (i) for which ALEXION decides to permanently discontinue all Development, Commercialization, and Manufacturing, (ii) which ALEXION decides to terminate pursuant to Section 12.6, or (iii) which is the subject of termination by ZEALAND pursuant to Section 12.2.1 or to the extent [***] that is the subject of a Patent Challenge by ALEXION and termination by ZEALAND pursuant to Section 12.7, in each case except to the extent that [***].
1.51 “Discovery/Development Activities” means the activities set forth in each Discovery Plan or Development Plan to be conducted by ZEALAND with respect to a Target or any Candidate Product pursuant to a Discovery/Development Program during the Discovery/Development Program Term for such Target or Candidate Product.
1.52 “Discovery/Development Program” means, on a Target-by-Target basis, the program of Discovery/Development Activities undertaken for each Target under the applicable Discovery Plan and any Development Plans as set forth in Article 2 including the Lead Development Program attached as APPENDIX 1 hereto, and any Discovery/Development Programs directed to a Selected Target, a Replacement Target or an Additional Target attached to this Agreement as APPENDIX 3 and incorporated herein after the Effective Date.
1.53 “Discovery/Development Program Term” has the meaning set forth in Section 2.1.5.
1.54 “Discovery Plan” has the meaning set forth in Section 2.1.2(a).
1.55 “Discovery Plan Term” has the meaning set forth in Section 2.1.6.
1.56 “DMF” has the meaning given in Section 4.3.
1.57 “Dollar” means the U.S. dollar, and “$” will be interpreted accordingly.
1.58 “Effective Date” has the meaning set forth in the preamble.
1.59 “EMA” has the meaning set forth in Section 1.129.
1.60 “Evaluation Period” means, with respect to one or more peptides or molecules included in the Results or Deliverables under a Discovery Plan, the period of time commencing upon the [***] for such peptides or molecules and ending on [***] (unless [***]).
1.61 “Executive Meeting” has the meaning set forth in Section 13.8.
1.62 “Executive Officers” has the meaning set forth in Section 13.8.
1.63 “Existing Lead Molecule” means any cyclic peptide or other molecule (i) created by or on behalf of ZEALAND or its Affiliates (including through any subcontractor or sublicensee) and (ii) that has been identified and demonstrated to be directed to or to have pharmaceutically useful binding to or modulation of the Lead Target, all of which are identified on Schedule 1.63.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
1.64 “Existing Lead Product” means any pharmaceutical composition in any formulation or for any route of administration, and in any dosage strength or presentation that is, contains or consists of one or more Existing Lead Molecules whether alone or with other active ingredients.
1.65 “Existing Product Sales Milestone Event” has the meaning set forth in Section 6.3.2
1.66 “Existing Product Sales Milestone Payment” has the meaning set forth in Section 6.3.2.
1.67 “Expended Discovery Costs” has the meaning set forth in Section 2.2.3.
1.68 “FD&C Act” means the Federal Food, Drug and Cosmetic Act.
1.69 “FDA” has the meaning set forth in Section 1.129.
1.70 “Field” means any and all prophylactic, diagnostic and therapeutic uses in humans, including the use of Products for the diagnosis, treatment, palliation or prevention of any disease or medical or aesthetic condition in humans.
1.71 “First Commercial Sale” means, on a country-by-country and Product-by-Product basis, the [***]. For the avoidance of doubt, [***] shall not be considered a First Commercial Sale.
1.72 “FTE” means a qualified full time person, or more than one person working the equivalent of a full-time person, where “full time” is based upon a total of [***] working hours per Calendar Year of scientific or technical work carried out by a duly qualified employee of ZEALAND. Overtime and work on weekends, holidays and the like shall not be counted with any multiplier (e.g. time-and-a-half or double time) toward the number of hours that are used to calculate the FTE contribution.
1.73 “FTE Rate” means [***] dollars ($[***]) per FTE for the Calendar Years [***], subject to annual adjustment beginning on [***] to reflect any year to year percentage change in the average CPI for the immediately preceding Calendar Year, provided that any annual increase shall not exceed [***].
1.74 “GAAP” means the generally accepted accounting principles in the United States of America (US GAAP), which principles are currently used at the relevant time and consistently applied by the applicable Party.
1.75 “Generic Competition” means, with respect to a given Product or Combination Product in a given country in the Territory, that the sales of one (1) or more Generic Products in such country (other than a Generic Product sold by ALEXION or its Affiliates or by a Sublicensee under a license granted by ALEXION or its Affiliates) achieve, [***]. If no data is commercially available, then [***].
1.76 “Generic Product” means, with respect to a Product, any product that is sold by a Third Party under a Regulatory Approval granted by a Regulatory Authority to a Third Party which Third Party has not obtained the right to market or sell such product from ALEXION (including as a Sublicensee, subcontractor, or Third Party distributor of ALEXION or any of its Affiliates) and
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
either (a) [***] or (b) [***], including but not limited to any product [***]. A Product [***] will not constitute a Generic Product.
1.77 “GLP” means all applicable good laboratory practice laws, regulations, and standards, including, as applicable, as set forth in the then-current regulation for Good Laboratory Practice for Nonclinical Laboratory Studies in 21 C.F.R. Part 58, and the equivalent Applicable Law in the region in the Territory, each as may be applicable from time to time.
1.78 “IND” means an Investigational New Drug application described in §505(i) of the FD&C Act and subject to 21 C.F.R. Part 312 or any comparable filings outside of the United States (such as a Clinical Trial Application in the European Union) required to commence human clinical trials in such country or region, and all supplements or amendments that may be filed with respect to the foregoing.
1.79 “Indemnification Claim Notice” has the meaning set forth in Section 11.3.
1.80 “Indemnified Party” has the meaning set forth in Section 11.3.
1.81 “Indemnifying Party” has the meaning set forth in Section 11.3.
1.82 “Infringed Patent” has the meaning set forth in Section 9.4.1.
1.83 “Insolvency Event” has the meaning as defined in Section 12.3.
1.84 “Intellectual Property” or “IP” means all (a) Patent Rights and (b) all rights, including Patent Rights, in, under and to all Inventions and Know-How.
1.85 “Invention” means any process, method, utility, formulation, composition of matter, article of manufacture, material, creation, discovery or finding, or any improvement thereof, that is conceived and/or reduced to practice, whether patentable or not.
1.86 “Invoice” means an original invoice sent by ZEALAND to ALEXION with respect to any payment due hereunder meeting the reasonable requirements provided by ALEXION to ZEALAND within thirty (30) days of the Effective Date and as may be updated from time to time.
1.87 “Joint Patent Rights” has the meaning set forth in Section 9.1.2.
1.88 “Joint Steering Committee” or “JSC” has the meaning set forth in Section 3.2.1.
1.89 “Know-How” means all scientific and technical information, know-how and data, in any tangible or intangible form and whether or not patentable, including trade secrets, specifications, instructions, processes, formulae, methods, practices, techniques, results, formulations, software, algorithms, technology, test data (including pharmacological, biological, chemical, biochemical, toxicological and clinical test data, analytical and quality control data and stability data), chemistry, manufacturing and controls information, studies and procedures, and other technology applicable to compounds, formulations, compositions, products or to their Manufacture, research and Development, registration, use or Commercialization or methods of assaying or testing them and also including any and all presentations, summaries and analyses thereof.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
1.90 “Lead Development Program” means, with respect to the Lead Target, the written summary attached hereto as APPENDIX 1 of the specific Discovery/Development Activities to be conducted by ZEALAND under the Discovery Plan and the initial Development Plan of the Lead Discovery/Development Program, the timelines and budget for completion of such activities with respect to the Lead Target.
1.91 “Lead Target” means [***] (also referred to as “[***]”) as further identified in Appendix 2.
1.92 “Licensed Intellectual Property” or “Licensed IP” means, on a Candidate Product-by-Candidate Product, Product-by-Product, Existing Lead Molecule-by-Existing Lead Molecule, Created Lead Molecule-by-Created Lead Molecule, or Target-by-Target basis as applicable, any and all Intellectual Property that is: (1) (i) owned or Controlled by ZEALAND or any of its Affiliates as of the Effective Date or at any time during the Term and/or (ii) conceived, reduced to practice or otherwise discovered (a) solely by or on behalf of ZEALAND or its Affiliates or subcontractors, or (b) jointly by, on the one hand, by or on behalf of ZEALAND or its Affiliates or subcontractors and, on the other hand, by or on behalf of Alexion or its Affiliates or subcontractors in each case under clause (ii) in the course of conducting the Discovery/Development Activities, Manufacturing, or otherwise arising out of ZEALAND’s performance of its obligations under this Agreement and (2) necessary or useful to Develop, have Developed, Manufacture, have Manufactured, use, have used, offer for sale, have offered for sale, sell, have sold, export, have exported, import, have imported, Commercialize, have Commercialized or otherwise exploit Candidate Products, Existing Lead Molecules, Created Lead Molecules, or Products.
1.93 “Licensed Patent Rights” means any Patent Rights in and to the Licensed IP, including those listed on APPENDIX 5.
1.94 “[***] Product” means [***] or [***] or [***] pursuant to Section 2.2.4.
1.95 “MAA” means an application for the authorization to market a product in any country or group of countries outside the U.S., as defined in the Applicable Laws and filed with the Regulatory Authority of a given country or group of countries.
1.96 “Major European Market” means any of [***].
1.97 “Manufacture” or “Manufacturing” means any and all activities directed to manufacturing, processing, packaging, labeling, filling, finishing, assembly, shipping, storage, or freight of any pharmaceutical product, drug substance, drug product, finished product, placebo, or comparator agent (or any components or process steps involving any such product or any companion diagnostic), as the case may be, including quality assurance and stability testing, characterization testing, quality control release testing of any such product including any drug substance, drug product or finished product quality assurance batch record review and release of any such product, process development, qualification, and validation, scale-up, pre-clinical, clinical, and commercial manufacture and analytic development, and product characterization.
1.98 “Mediation Period” has the meaning set forth in Section 13.8.2.
1.99 “NDA” means a New Drug Application, as described in §505(b) of the FD&C Act and subject to 21 C.F.R. Part 314, including any amendments submitted pursuant to 21 C.F.R. §314.60, or any
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
analogous application or submission with any Regulatory Authority to obtain authorization to market a pharmaceutical product outside of the United States.
1.100 “Net Sales” means with respect to a Product, in a particular period the sum of (i) and (ii):
(i) net sales amounts [***]; and
(ii) net sales amounts [***].
The calculations described in clauses (i) and (ii) above shall [***]. For the avoidance of doubt, [***] shall not be included in Net Sales.
In the event a Product is [***], Net Sales shall be calculated by [***]. In the event that [***], the [***] in the above described equation shall be replaced with [***]; provided, however, that [***].
1.101 “Non-Defaulting Party” has the meaning set forth in Section 12.3.
1.102 “Non-Sublicensing Party” has the meaning set forth in Section 9.2.6.
1.103 “[***] Product” means any [***] (a) that [***] and (b) for which [***], that [***].
1.104 “[***]” means [***].
1.105 “[***] Period” has the meaning set forth in Section 9.2.7.
1.106 “[***] Period” means, for [***], the period (1) beginning on the later of: (a) the [***] year anniversary of [***] or (b) [***] year anniversary of [***] and (2) ending [***] months thereafter.
1.107 “[***]” has the meaning set forth in Section 9.2.7.
1.108 “Optioned Molecule” means any peptide or other molecule that is created and identified by or on behalf of ZEALAND or its Affiliates (including through any permitted subcontractor or sublicensee) through conduct of the Discovery/Development Program that binds to or modulates a Selected Target, a Replacement Target or Additional Target to a pharmacologically relevant extent.
1.109 “Optioned Product” means any pharmaceutical composition in any formulation or for any route of administration, and in any dosage strength or presentation that is, contains or consists of one or more Candidate Products that are Optioned Molecules whether alone or with other active ingredients.
1.110 “Optioned Target” means any Selected Target designated pursuant to Section 2.1.3, any Replacement Target designated pursuant to Section 2.2.3 and any Additional Target designated pursuant to Section 2.5.
1.111 “Orange Book” has the meaning set forth in Section 9.4.4.
1.112 “Other Component(s)” has the meaning set forth in Section 1.26.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
1.113 “Paragraph IV Certification” has the meaning set forth in Section 9.4.5.
1.114 “Paragraph IV Proceeding” has the meaning set forth in Section 9.4.5(b).
1.115 “Patent Rights” means any and all (i) patents, (ii) patent applications, including all provisional and non-provisional applications, Patent Cooperation Treaty (PCT) applications, substitutions, continuations, continuations-in-part, divisions and renewals, and all patent rights granted thereon, (iii) all patents-of-addition, reissues, re-examinations and extensions or restorations by existing or future extension or restoration mechanisms, including supplementary protection certificates and equivalents thereof, (iv) inventor’s certificates, letters patent, or (v) any other substantially equivalent form of government issued right substantially similar to any of the foregoing described in subsections (i) through (v) above, anywhere in the world.
1.116 “Patent Term Extension” has the meaning set forth in Section 9.6.
1.117 “Per Created Lead Product Sales” has the meaning set forth in Section 6.5.2.
1.118 “Per Existing Lead Product Sales” has the meaning set forth in Section 6.5.1.
1.119 “Per Optioned Lead Product Sales” has the meaning set forth in Section 6.5.2.
1.120 “Per Target Development Cap” have the meaning set forth in Section 2.3.
1.121 “Phase I Clinical Trial” means a human clinical trial of a product, the principal purpose of which is a preliminary determination of safety in healthy individuals or patients, and that meets the requirements of 21 CFR § 312.21(a) or, if conducted in a country other than the United States, the foreign equivalent thereof.
1.122 “Phase II Clinical Trial” means a human clinical trial of a product, the principal purpose of which is a determination of safety and efficacy in the target patient population, and that meets the requirements of 21 CFR § 312.21(b) or, if conducted in a country other than the United States, the foreign equivalent thereof.
1.123 “Pivotal Clinical Trial” means a human clinical trial of a product (whether or not specifically referred to as a pivotal trial), the design of which is acknowledged by the FDA to be sufficient for such clinical trial to satisfy the requirements of 21 CFR § 312.21(c) or, if conducted in a country outside the United States, a similar clinical trial prescribed by the applicable Regulatory Authority in such country which is acknowledged by such Regulatory Authority to be sufficient for such clinical trial to satisfy the requirements of a pivotal efficacy and safety clinical trial.
1.124 “PMDA” has the meaning set forth in Section 1.129.
1.125 “Product(s)” means any product incorporating a Candidate Product (or derivative thereof) for which ALEXION has delivered a Development Decision Notice stating that ALEXION elects to continue Development of such Candidate Product in accordance with Section 2.2.4.
1.126 “Program Leaders” has the meaning set forth in Section 3.1.1.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
1.127 “Receiving Party” has the meaning set forth in Section 8.1.
1.128 “Regulatory Approval” means, with respect to a Product in a country in the Territory, (a) any and all licenses, registrations, authorizations and approvals of the applicable Regulatory Authority, including NDAs or any foreign equivalent thereof, as applicable, and (b) if applicable, any and all pricing or reimbursement authorizations and approvals, in each case ((a) and (b)) that are necessary to Manufacture, distribute, sell, obtain reimbursement for, market, use, store, and import such Product in such country.
1.129 “Regulatory Authority” means (i) any governmental authority, notified body or other organization in a country or region that regulates the Manufacture or sale of pharmaceutical or medicinal products or medical devices, including the United States Food and Drug Administration (the “FDA”), Japan’s Pharmaceuticals and Medical Devices Agency (“PMDA”) and the European Medicines Agency (“EMA”), and any successors thereto, and (ii) any other relevant bodies authorized by Applicable Law to review or otherwise exercise oversight over MAAs, other regulatory filings or Regulatory Approvals.
1.130 “Regulatory Documentation” has the meaning given in Section 4.3.
1.131 “Replacement Development Cap” has the meaning set forth in Section 2.2.3.
1.132 “Replacement Target” has the meaning given in Section 2.2.3.
1.133 “Results” means, on a Target-by-Target, Candidate Product-by-Candidate Product or Product-by-Product basis, as applicable, [***] related to [***], in each case, with respect to [***].
1.134 “Royalty Term” means with respect to each Product and each country in the Territory, the period beginning on the date of the First Commercial Sale of such Product in such country, and ending upon [***].
1.135 “Sales Milestone Event” has the meaning set forth in Section 6.3.3.
1.136 “Sales Milestone Payment” has the meaning set forth in Section 6.3.3.
1.137 “Selected Target” has the meaning given in Section 2.1.3.
1.138 “[***] Product” means [***] or [***] or [***] pursuant to Section 2.2.4.
1.139 “sNDA” means a supplement to an NDA as set forth in 21 C.F.R. §§314.70 and 314.71, including any amendments submitted pursuant to 21 C.F.R. §314.60, or any analogous submission with any Regulatory Authority outside of the United States.
1.140 “[***]” has the meaning set forth in Section 12.5.
1.141 “Sublicensees” means any Third Party, including a co-development, co-promotion or co-marketing partner, to whom ALEXION or any of its Affiliates grants a sublicense of its rights hereunder to further Develop or Commercialize Products, but excluding service providers, clinical research organizations, manufacturers, wholesalers and other distributors of finished Product.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
1.142 “Sublicensing Party” has the meaning set forth in Section 9.2.6.
1.143 “[***] Model” has the meaning set forth in Section 2.7.1.
1.144 “[***] Model Work Plan” has the meaning set forth in Section 2.7.1.
1.145 “Target” means the Lead Target, any other Selected Target (other than any Selected Target which has been replaced pursuant to Section 2.2.3), Additional Target or Replacement Target.
1.146 “Target Exercise Notice” has the meaning set forth in Section 2.1.3.
1.147 “Target Option” has the meaning set forth in Section 2.1.3.
1.148 “Target Option Exercise Fee” has the meaning set forth in Section 6.2.
1.149 “Target Option Period” means the period beginning on [***] and ending on the date that is [***] year anniversary of [***].
1.150 “[***]” means, with respect to [***], all [***] to the extent that such [***] or [***], or [***] that is [***].
1.151 “[***] Patent Rights” has the meaning set forth in Section 9.3.1.
1.152 “Taxes” has the meaning set forth in Section 6.11.
1.153 “Term” has the meaning set forth in Section 12.1.
1.154 “Territory” means all of the countries of the world, and their territories and possessions.
1.155 “[***] Activities” shall mean the activities set forth in Schedule 1.155, as may be amended by the JSC from time to time.
1.156 “Third Party” means any person or entity other than ALEXION or ZEALAND or their respective Affiliates.
1.157 “Third Party Claim” shall have the meaning as set forth in Section 11.1.
1.158 “U.S. Prime Rate” has the meaning set forth in Section 6.10.
1.159 “Valid Claim” means, with respect to a particular country, a claim of an issued patent Covering the composition of matter or use of a given Product that has not expired or been abandoned, or been revoked, held invalid or unenforceable by a patent office, court or other governmental agency of competent jurisdiction in a final and non-appealable judgment (or judgment from which no appeal has been taken within the allowable time period).
1.160 “Voting Stock” has the meaning set forth in Section 1.21(i).
1.161 “ZEALAND Costs” means (i) [***] and (ii) ZEALAND’S [***], in each case ((i) and (ii)), in the [***] of (a) Discovery/Development Activities performed under the applicable
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Discovery/Development Program or activities under the [***] Work Plan and in accordance with budget set forth under such Discovery/Development Program or [***] Work Plan or (b) activities for which ZEALAND is to be reimbursed under Section 2.7.4 ([***] Technology Transfer).
1.162 “ZEALAND Indemnitees” has the meaning set forth in Section 11.2.
1.163 “ZEALAND Patent Rights” has the meaning set forth in Section 9.3.2.
1.164 “ZEALAND Target Notice” has the meaning set forth in Section 2.5.
2. RESEARCH AND DEVELOPMENT PROGRAM
2.1 Generally.
2.1.1 Goal. With respect to any Target, the Parties agree to engage in a Discovery/Development Program to identify and Develop one or more Candidate Products directed to the Lead Target, any other Selected Targets, Replacement Targets and Additional Targets as further described in a Discovery Plan and one or more Development Plans under each Discovery/Development Program.
2.1.2 Discovery/Development Program Plans. Each Discovery/Development Program shall include a Discovery Plan and one or more Development Plans in respect to a Target. The Parties may update and amend the Discovery Plans or Development Plans from time to time through the JSC, subject to Sections 3.2.4 and 3.2.5.
(a) Discovery Plan. The Discovery Plan under a Discovery/Development Program shall set forth, at a minimum: (i) Candidate Product Criteria with respect to the applicable Target; (ii) the specific activities to be conducted by ZEALAND in order to identify Candidate Products from any previously identified, existing or new molecules meeting the Candidate Product Criteria and provide ALEXION with sufficient data and information with respect to such Candidate Product(s) for ALEXION to determine whether to designate such Candidate Product as an Existing Lead Product, Created Lead Product, or Optioned Product, as applicable; and (iii) a proposed timeline and budget for the performance of the Discovery Plan (“Discovery Plan”).
(b) Development Plan. Each Development Plan under a Discovery/Development Program shall set forth, at a minimum, the activities to be conducted by ZEALAND in order to carry out further Development of any Candidate Products thereunder including (i) the generation of sufficient data and information to [***] in connection with obtaining an Acceptance of an IND for a Product containing or consisting of such Candidate Product directed to the applicable Target and (ii) a proposed timeline and budget for the performance of the Development Plan (“Development Plan”). The Lead Discovery/Development Program including a Discovery Plan and a Development Plan setting forth the specific activities to be conducted by ZEALAND to identify and Develop Candidate Products directed to the Lead Target as agreed to by the Parties is attached hereto as APPENDIX 1.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
2.1.3 Target Option. At any time during the Target Option Period, ALEXION shall have the option (“Target Option”) to select any Complement Pathway Target in addition to the Lead Target (each a “Selected Target”) as the subject of Discovery/Development Activities under a Discovery/Development Program with respect to such Selected Target, provided that ALEXION may have no more than three (3) Complement Pathway Targets as Selected Targets unless the Parties otherwise agree in writing. ALEXION shall exercise each Target Option by written notice that specifies the Selected Target and is delivered to ZEALAND prior to the expiration of the Target Option Period (each, such notice a “Target Exercise Notice”). The Selected Targets are in addition to the Additional Targets.
2.1.4 Discovery/Development Programs For Selected Targets and Additional Targets. Upon receipt by ZEALAND of a Target Exercise Notice specifying a Selected Target or an Additional Target Notice specifying an Additional Target, ZEALAND shall promptly prepare a Discovery Plan and Development Plan under a Discovery/Development Program for such Selected Target or Additional Target to be agreed by ALEXION in writing. Upon ALEXION’s written approval of such Discovery Plan and Development Plan pursuant to this Section 2.1.4 and payment of the Target Option Exercise Fee: (i) such Selected Target or Additional Target shall become a Target for all purposes hereunder, (ii) such Discovery Plan and Development Plan shall be attached hereto in APPENDIX 3, and (iii) ZEALAND shall commence Discovery/Development Activities under such Discovery Plan.
2.1.5 Discovery/Development Program Term. Each Discovery/Development Program shall have a term: (1) commencing: (i) for the Lead Discovery/Development Program, on the Effective Date and (ii) for any other Discovery/Development Program, upon approval by ALEXION of the Discovery Plan and first Development Plan under such Discovery/Development Program and (2) expiring upon the earlier of: (a) the date of termination of the last of any remaining Discovery Plan and Development Plan under such Discovery/Development Program as determined by the JSC; (b) in the case of a Selected Target, the date of receipt by ZEALAND of a notice from ALEXION nominating a replacement pursuant to Section 2.2.3 of the Target that is the subject of such Discovery/Development Program; (c) the date of termination by ALEXION in its sole discretion of such Discovery/Development Program; or (d) when ZEALAND has expended an amount under such Discovery/Development Program equal to the relevant Per Target Development Cap and ALEXION declines to pay ZEALAND Costs for further Discovery/Development Activities pursuant to Section 2.3.3 (the “Discovery/Development Program Term”).
2.1.6 Discovery Plan Term. Each Discovery Plan shall be performed, on a Target-by-Target basis by ZEALAND, under a corresponding Discovery/Development Program during the period (1) commencing (i) for the Discovery Plan of the Lead Discovery/Development Program, on the Effective Date or (ii) for any other Discovery Plan, upon ALEXION’s approval of such Discovery Plan and (2) expiring upon the earlier of (a) the date of completion of all activities contemplated by the Parties under such Discovery Plan as determined by the JSC; (b) the date of termination by ALEXION in its sole discretion of
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
such Discovery Plan; or (c) the date of termination of the corresponding Discovery/Development Program (the “Discovery Plan Term”).
2.1.7 Development Plan Term. Each Development Plan shall be performed, on a Candidate Product-by-Candidate Product basis by ZEALAND, during the period (1) commencing, for any Development Plan under a Discovery/Development Program directed to any Target, upon ALEXION’s confirmation of approval of such Development Plan, as may be updated or modified, following the delivery of a Development Decision Notice pursuant to Section 2.2.5 and (2) expiring upon the earlier of (a) the date of Acceptance by a Regulatory Authority, in [***] of an IND filed in connection with the Candidate Product applicable to such Development Plan; (b) the date of completion of all activities contemplated by the Parties under such Development Plan as determined by the JSC; (c) the date of termination by ALEXION in its sole discretion of such Development Plan; or (d) the date of termination of the applicable Discovery/Development Program (the “Development Plan Term”).
2.2 Performance of Discovery/Development Program.
2.2.1 Diligent Efforts. On a Target-by-Target and Candidate Product-by-Candidate Product basis, ZEALAND shall use its Diligent Efforts to conduct each Discovery/Development Program in accordance with the timeframes set forth in the relevant Discovery Plan and any Development Plans thereunder, in accordance with all Applicable Laws (including GLP) and the provisions of this Agreement. On a Discovery/Development Program-by-Discovery/Development Program Basis, ZEALAND shall dedicate the efforts of a number of FTEs capable of carrying out the Discovery/Development Activities set forth in the corresponding Discovery Plan and any Development Plans to a professional workmanlike standard and shall provide all necessary materials and facilities therefor, as provided in each such Discovery/Development Program.
2.2.2 Discovery Plan. During each Discovery Plan Term, ZEALAND will deliver to ALEXION samples of all peptide(s) or other molecule(s) that meet the Candidate Product Criteria set forth in the applicable Discovery Plan, together with a report as set forth in Section 2.4, summarizing the Results and Deliverables of the Discovery Plan within [***] days of submission to ALEXION of each such peptide(s) or other molecule(s). With respect to peptides or other molecules evaluated by ZEALAND which did not meet the Candidate Product Criteria but [***], the Results and Deliverables shall also include the identities of such other peptides or molecules and the results of each such evaluation. ZEALAND shall deliver to ALEXION, upon its written request, samples of each peptide or other molecule that did not meet the Candidate Product Criteria but [***] and sufficient data and information with respect to such peptide or molecule for ALEXION to determine whether to designate such peptide or molecule as an Existing Lead Product, Created Lead Product, or Optioned Product as applicable. Any and all costs incurred by ALEXION in the evaluation of such Candidate Products or such other molecules by ALEXION during the applicable Evaluation Period shall be borne by ALEXION.
2.2.3 Failure to Deliver Candidate Products. On a Selected Target-by-Selected Target basis, in the event that ZEALAND fails to deliver to ALEXION any peptides or other molecules
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
meeting the Candidate Product Criteria, within [***] months of [***] under a given Discovery/Development Program, or as otherwise agreed by the JSC, ALEXION shall have the right to nominate in writing any [***] Target as a replacement for the Selected Target that is the subject of such Discovery/Development Program (a “Replacement Target”). On the receipt by ZEALAND of such nomination, the peptides or other molecules included in the Results and Deliverables under the Discovery/Development Program for the Selected Target that has been replaced shall be treated to the extent the minimum information required in Section 2.5 has been provided, as though they were peptides or molecules for which ALEXION has not elected to add to this Agreement pursuant to Section 2.5. Effective as of such nomination (i) the JSC shall prepare a Discovery Plan and a Development Plan under a Discovery/Development Program corresponding to such Replacement Target pursuant to Section 2.1.4 and (ii) upon ALEXION’s approval of such Discovery Plan and Development Plan the Replacement Target shall automatically be deemed a Target for all purposes under this Agreement and ZEALAND shall commence Discovery/Development Activities under such Discovery Plan. Within [***] days of the nomination of a Replacement Target by ALEXION, ZEALAND shall provide ALEXION with a summary of ZEALAND Costs expended under the Discovery Plan of the Discovery/Development Program directed to the replaced Selected Target (“Expended Discovery Costs”). The Per Target Development Cap for the Discovery/Development Program directed to the Replacement Target shall be equal to [***] (“Replacement Development Cap”). Each Selected Target may [***] pursuant to this Section 2.2.3 (and [***]) and this Section 2.2.3 shall [***].
2.2.4 Development Decision Notice. For each peptide or molecule, ALEXION shall determine whether ALEXION wishes to designate one or more peptide(s) or molecule(s) to be Candidate Product(s) for progression to Discovery/Development Activities under a Development Plan of the applicable Discovery/Development Program. In the case of any Development Decision Notice delivered under the Lead Discovery/Development Program, such notice shall, for an Existing Lead Molecule or Created Lead Molecule, whether such molecule is [***] or [***]. ALEXION shall consider ZEALAND’S comments regarding such designation(s) but shall make any designation(s) in its sole discretion. For clarity, a Development Decision Notice with respect to a peptide or other molecule that did not meet all of the applicable Candidate Product Criteria but which [***] shall be considered a designation of such peptide or other molecule as a “Candidate Product”. In the event that ALEXION does not provide a Development Decision Notice in respect of any Candidate Products with respect to a Selected Target, Replacement Target or Additional Target prior to the end of the Evaluation Period applicable to such Target, such Candidate Products shall be treated as though they are peptides or molecules ALEXION has not elected to add to this Agreement pursuant to Section 2.5.
2.2.5 Development Plan. Upon the receipt by ZEALAND of a Development Decision Notice designating, in accordance with this Agreement, a peptide or molecule as a Candidate Product for further Development, the JSC will promptly review the Development Plan under the applicable Discovery/Development Program approved by ALEXION with respect to the applicable Target and determine whether any updates or other modifications are warranted based on the identity of the Candidate Product and other information including the Results and Deliverables generated during the applicable Discovery Plan.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Upon ALEXION’s confirmation of approval of such Development Plan, or approval with respect to any updates or modifications, such Development Plan (if updated or modified) shall become part of the applicable Discovery/Development Program and, in the case of a Development Plan directed to the Lead Target, be attached hereto in APPENDIX 1 or, in the case of an Optioned Target, be attached hereto in APPENDIX 3, and, only then, shall ZEALAND commence Discovery/Development Activities thereunder.
2.2.6 Subcontracting. ZEALAND may not subcontract any of its Discovery/Development Activities except with the prior written consent, not to be unreasonably withheld, conditioned or delayed, of ALEXION and in compliance with the provisions of Section 9.2.6; provided that, ALEXION’s consent shall not be required with respect to any subcontractor listed on APPENDIX 6, which list shall be provided for approval by ALEXION, such approval not to be unreasonably withheld, conditioned or delayed, within [***] days after the Effective Date.
2.2.7 Research and Evaluation Material and Technology Transfer. The Results and Deliverables under any Discovery/Development Program shall include materials and samples of any previously identified, existing or new peptides or molecules that do not meet the Candidate Product Criteria but [***], that were screened, analyzed, identified, discovered, or generated during any Discovery/Development Activities under the Discovery/Development Program, including of any specific activities to be conducted by ZEALAND to identify Candidate Products, as reasonably necessary or useful for ALEXION to exercise its rights to evaluate Candidate Products as set forth herein and otherwise practice the licenses granted under this Agreement.
2.3 Discovery/Development Program Costs.
2.3.1 ZEALAND’s Responsibilities. ZEALAND shall be responsible for all costs and expenses of conducting all Discovery/Development Activities under the Discovery Plan and any Development Plans, including as may be updated by the JSC, of each Discovery/Development Program, provided that ZEALAND Costs in total for the Discovery/Development Activities under a given Discovery/Development Program for a Target shall not exceed the following amounts (each the “Per Target Development Cap” for an applicable Target):
(a) the Per Target Development Cap for the Lead Target shall be [***] Dollars ($[***]);
(b) the Per Target Development Cap for a Selected Target that has not been replaced pursuant to Section 2.2.3 shall be [***] Dollars ($[***]);
(c) the Per Target Development Cap for a Replacement Target shall be: (i) for the Discovery Plan, [***]; and (ii) for the Development Plan, [***].
2.3.2 Effect of a Development Decision Notice in relation to a Replacement Target. Where ALEXION has replaced a Selected Target pursuant to Section 2.2.3, and it subsequently delivers a Development Decision Notice in respect of one or more Candidate Products in
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
respect of such Replacement Target, it shall, in accordance with Section 2.3.3(a) pay to ZEALAND an amount equivalent to [***].
2.3.3 ALEXION’S Payment Obligations. ALEXION’s payment obligations with respect to the performance of a Discovery/Development Program shall be limited to amounts payable pursuant to Sections 2.3.2, 6.1.1 and 6.2; provided that [***]. The Parties shall [***].
(a) Reimbursement Procedure. For all Discovery/Development Activities for which ALEXION is responsible under this Section 2.3.3, ALEXION shall pay [***]. ZEALAND shall invoice ALEXION for [***], with an itemized documentation reasonably supporting the Invoice, and ALEXION shall pay ZEALAND all undisputed amounts due under this Section 2.3.3 within [***] days following ALEXION’s receipt of the applicable Invoice for [***].
(b) No Obligation. Except as expressly set forth in this Section 2.3.3 ALEXION shall have no obligation to [***]. ZEALAND shall not be required to [***].
2.4 Reports. During the Discovery/Development Program Term of a Discovery/Development Program for a particular Target, ZEALAND shall furnish ALEXION with written reports to update ALEXION on the status of the Discovery/Development Activities being performed thereunder. During each Discovery Plan Term, ZEALAND shall provide such reports [***] and each such report shall include at a minimum [***]. During the Development Plan Term of each Development Plan, ZEALAND shall provide reports on a [***] basis and each such report shall include at a minimum [***]. Each report (whether for the Discovery Plan or Development Plan) must be sufficient in content and detail to allow ALEXION to evaluate the progress of the Discovery/Development Activities in relation to the requirements, goals, and budget of the applicable Discovery/Development Program and to exercise its rights hereunder. In addition, ZEALAND will provide to ALEXION such other information as may be related to or required under any Discovery Plan or Development Plan or otherwise for the performance of the Discovery/Development Program, and which is reasonably requested by ALEXION.
2.5 Additional Targets. From [***] until [***], ZEALAND shall not and shall cause its Affiliates not to (a) Develop, Commercialize or Manufacture any molecule or other product that is directed to, binds to, or otherwise modulates any [***] Target (other than activities performed on behalf of ALEXION with respect to a Target) or (b) enable, assist or grant any rights to a Third Party to conduct any activities described in clause (a) without first notifying ALEXION in writing (“ZEALAND Target Notice”). The ZEALAND Target Notice shall specify the [***] Target and include such information as has been generated by ZEALAND on such [***] Target (including, at a minimum [***]). Upon the receipt of a ZEALAND Target Notice, ALEXION shall have [***] days in which to request reasonable additional information and access with respect to such [***] Target and ZEALAND shall provide ALEXION with such information and access. The date on which ALEXION has received such additional information and access shall be the “Additional Target Trigger Date”. ALEXION shall have the right, in its sole discretion, to add such [***] Target as an “Additional Target” under the terms and conditions of this Agreement applicable to an Optioned Target in addition to the three Optioned Targets provided in this Agreement by written notice delivered within [***] months of the Additional Target Trigger Date (“Additional Target
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Notice”). If ALEXION does not so elect to add the Additional Target within [***] months of the Additional Target Trigger Date, ZEALAND may pursue the manufacture, development and commercialization of the peptides or molecules with respect to the [***] Target (other than any Target) in each case solely if and as set forth in the ZEALAND Target Notice.
2.6 Exclusivity. Prior to [***], other than: (i) [***] performance under this Agreement; (ii) the [***]; and (iii) the [***] as permitted under Section 2.5; [***] shall not, and shall cause its Affiliates not to, (a) Develop, Commercialize or Manufacture any [***] or (b) enable, assist or grant any rights to a Third Party to conduct any activities described in clause (a) of this sentence. During ***], [***] shall not, and shall cause its Affiliates not to (x) commence or conduct the Development, Commercialization or Manufacture of any [***] or (y) assist, enable, or grant any rights to a Third Party to conduct any activities described in clause (x) of this sentence.
2.7 [***] Model.
2.7.1 [***] Model Work Plan. Within [***] days of the Effective Date, the JSC will prepare a work plan describing specific activities to be conducted by ZEALAND for Developing a [***] model (“[***] Model”)] for use in Developing Candidate Products or Products. Such work plan (“[***] Model Work Plan”) will include, at a minimum, a detailed timeline for Developing the [***] Model and a budget for all activities described therein. The JSC may, from time to time, update the work plan.
2.7.2 Performance of [***] Work Plan.
(a) ZEALAND will use its Diligent Efforts (in accordance with Section 2.7.2(b)) to carry out the activities within the times agreed upon and set forth in the [***] Work Plan, accordance with all Applicable Laws and the provisions of this Agreement.
(b) ZEALAND shall devote the efforts of suitably qualified and trained employees and research assistants capable of carrying out the activities set forth in the [***] Work Plan to a professional workmanlike standard and shall provide all necessary materials and facilities therefor.
(c) ZEALAND may not subcontract any of its activities under the [***] Work Plan except with the prior written consent of ALEXION (such consent not to be unreasonably withheld, conditioned or delayed), and in compliance with the provisions of Section 9.2.6; provided that, ALEXION’s consent shall not be required with respect to any subcontractor listed on APPENDIX 6.
(d) The Results and Deliverables under the [***] Work Plan shall include materials and samples including of any previously identified, existing or new molecules used in, screened, analyzed, identified, discovered, or generated in the [***] Work Plan.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
2.7.3 [***] Work Plan Costs; Payment. For all activities under the [***] Work Plan, ALEXION shall pay [***]. ZEALAND shall invoice ALEXION, and ALEXION shall pay ZEALAND all undisputed amounts due under this Section 2.7.3 for each [***], with an itemized documentation reasonably supporting the Invoice within [***] days following ALEXION’s receipt of the applicable Invoice for [***].
2.7.4 [***] Technology Transfer. In addition to the information, data and materials exchanged between the Parties through the JSC with respect to the [***] Work Plan, within [***] days after the conclusion of the [***] Work Plan, ZEALAND shall transfer, at [***], to ALEXION all the data, information, materials and samples set out as Results and Deliverables under the [***] Work Plan. In addition to the foregoing technology transfer, at ALEXION’s reasonable request, ZEALAND shall provide ALEXION at [***] up to [***] FTE hours of support, including technical and quality support, to enable ALEXION to use such Results and Deliverables in the Development, Commercialization, Manufacture and other exploitation of any Product, Target, or Candidate Product.
2.8 Discovery/Development Quality. ZEALAND will conduct all Discovery/Development Activities and any activities under the [***] Work Plan in accordance with Applicable Law (including GLP) and the provisions of this Agreement.
2.9 Inspection and Auditing Rights. ALEXION shall have the right, at [***], to audit, inspect and observe the facilities utilized by ZEALAND for the Discovery/Development Activities, and any activities under the [***] Work Plan, including ZEALAND’s compliance with Applicable Laws and the terms and conditions of this Agreement, during normal business hours and upon at least [***] days’ prior notice. ZEALAND shall make available to ALEXION all relevant records and reports. ZEALAND agrees to respond to ALEXION’s audit findings within [***] days of receipt of ALEXION’s audit report, to take prompt corrective action to remedy any observed violations of the terms of this Agreement or of Applicable Law. Such audits may be conducted no more than [***] per [***], provided that ALEXION may also conduct follow-up audits or inspections at [***] at any time or times during a Calendar Year that are directed at significant or critical quality issues observed during the regular audit or brought to ALEXION’s attention by Regulatory Authorities.
3. GOVERNANCE
3.1 Collaboration Management.
3.1.1 Discovery/Development Program Leaders. Each Party will appoint a single individual to act as a point of contact between the Parties to support the Discovery/Development Programs and the Discovery/Development Programs (the “Program Leaders”). The initial Program Leaders shall be set forth in APPENDIX 4 — Title: “Contact List” hereto. Either Party may change the person designated as Program Leader upon written notice (including, in this case, via email notification) to the other Party, provided that such new Program Leader is, in the reasonable opinion of the appointing Party, suitably qualified.
3.1.2 Roles and Responsibilities. The Program Leaders will be responsible for (i) facilitating the flow of information and otherwise promoting communication of the day-to-day work
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
for each Discovery/Development Program and Discovery/Development Program, (ii) coordinating all work to be conducted under each Discovery/Development Program and Discovery/Development Program, and (iii) all scientific and technical questions addressed by one Party to the other Party. The Program Leaders shall conduct regular telephone conferences every [***] weeks, or more frequently as deemed necessary or appropriate by the Parties, to exchange information regarding the progress of each research project under and according to the applicable Discovery Plan or any Development Plans under a Discovery/Development Program and [***] Work Plan.
3.2 Governance.
3.2.1 Joint Steering Committee. Within thirty (30) days after the Effective Date of this Agreement, ZEALAND and ALEXION will form a joint steering committee (the “Joint Steering Committee” or “JSC”), which will be comprised of an equal number of representatives from each Party, as further provided below. Subject to (i) the prior written consent of the other Party, (ii) a written non-conflict of interest statement of any individual invited by a Party but not employed by such Party, and (iii) execution of a confidentiality agreement, either Party may invite non-voting employees and, as approved in advance by the other Party, which approval shall not be unreasonably withheld, conditioned or delayed, consultants or scientific advisors to attend meetings of the JSC and to review and discuss the Discovery/Development Program and the Discovery/Development Program.
3.2.2 Meetings. The JSC will meet in person (alternating between a site designated by each of ZEALAND and ALEXION) or by teleconference at least once every [***], or with such other frequency as the Parties may mutually agree provided always that there shall be at least one face-to-face meeting [***]. Specific meeting dates will be defined by mutual agreement of the Parties.
3.2.3 Responsibilities. The JSC shall oversee the Discovery/Development Programs and activities under the [***] Work Plan. Within such scope the JSC shall, subject to Section 3.2.4 and Section 3.2.5:
(a) review the efforts and the progress of the Parties in the conduct of the Discovery/Development Programs and activities under the [***] Work Plan;
(b) review ZEALAND Costs (both for the past and expected) under each Discovery/Development Program with respect to the applicable Per Target Development Cap or Replacement Development Cap during the Discovery Plan Term of each Discovery Plan, review the [***] reports under Section 2.4 and any Results and Deliverables with respect to each peptide or molecule (i) meeting the Candidate Product Criteria (if any) or (ii) evaluated with respect to the Candidate Product Criteria (if any). The JSC shall consider and advise regarding the progress towards the
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Candidate Product Criteria and any technical and budgetary issues that arise;
(c) during each Discovery Plan Term, review ZEALAND’s [***] update reports to ALEXION on the status of the Discovery/Development Activities being performed for each Target, including with respect to the status of the [***] Activities and any peptides or other molecules in relation to the Candidate Product Criteria;
(d) during each Development Plan Term, review ZEALAND’s [***] update reports to ALEXION on the status of the Discovery/Development Activities being performed for each Target;
(e) review and amend any Discovery Plans, Development Plans and the [***] Work Plan including with respect to any proposed revision or modification to such plan that (i) changes any timeline to complete any part of any such Discovery Plans, Development Plans or [***] Model Work Plan; (ii) alters the allocation of resources devoted by ZEALAND to any such Discovery Plans, Development Plans or [***] Model Work Plan; (iii) changes any of the Deliverables under any such any such Discovery Plans, Development Plans or [***] Model Work Plan; or (iv) changes any of the Candidate Product Criteria in any such Discovery Plan;
(f) consider and advise on any technical, quality or safety issues that arise under the Discovery/Development Programs or the [***] Work Plan;
(g) form such other committees as the JSC may deem appropriate, provided that such committees may make recommendations to the JSC but may not be delegated JSC decision-making authority;
(h) attempt to resolve any disputes on an informal basis; and
(i) address such other matters relating to the Discovery/Development Activities and the activities under the [***] Work Plan as either Party may bring before the JSC.
3.2.4 Decision Making Authority. A quorum for a meeting of the JSC shall require the presence of at least one (1) representative from each Party. The JSC shall endeavor to reach decisions by consensus, with each Party, through its representative members of the JSC, having one (1) vote. In the event that any subcommittee formed by the JSC does not reach a consensus agreement with respect to any material decision within [***] Business Days following a vote on such decision, the matter shall first be referred to the JSC for resolution. If the JSC cannot agree on a certain matter, then [***].
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
3.2.5 Limits on JSC Decision Making Authority. Except as provided under [***], neither Party shall have the authority to make any decision without the prior written consent of the other Party that would result in any revision or modification to any Discovery/Development Program (including any Discovery Plan or Development Plan thereunder) that (i) [***] or (ii) [***]. Further, except as expressly set forth herein, the JSC shall not have the power to amend the terms of, or waive compliance with, this Agreement.
3.3 Disbandment of the JSC. Upon the later of the expiration (or earlier termination) of the (i) last Discovery/Development Program for the last Product and (ii) the completion of activities under the [***] Work Plan, the JSC will have a final meeting to review the results of the overall collaboration and will thereafter be disbanded and will have no further authority with respect to the activities hereunder; provided that, in the event of an [***], the JSC shall, at [***], be disbanded and will have no further authority with respect to the activities hereunder.
4. DEVELOPMENT, REGULATORY APPROVALS AND COMMERCIALIZATION
4.1 Technology Transfer. In addition to the information, data and materials exchanged between the Parties through the JSC, on a Product-by-Product, Target-by-Target, and Candidate Product-by-Candidate Product basis, within [***] days after the conclusion of each Discovery Plan Term, Development Plan Term, and Discovery/Development Program Term in respect of the relevant Product, Target, and Candidate Products and at ALEXION’s reasonable request, ZEALAND shall transfer to ALEXION, at [***], all the data, information, materials, samples, know-how, and technology, in each case to the extent Controlled by ZEALAND and, in relation to the Manufacture of Products, from its CMOs (to the extent available to ZEALAND under (and subject to the terms of) a CMO Supply Agreement), set out as Results and Deliverables in the relevant Discovery Plan, Development Plan, and Discovery/Development Program. In addition to the foregoing technology transfer, ZEALAND shall provide ALEXION up to [***] FTE hours of support, including technical and quality support, to enable ALEXION to use such Results and Deliverables in the Development, Commercialization, Manufacture and other exploitation of the subject Product, Target, and Candidate Product.
4.2 Subsequent Development. Following the conclusion of the Development Plan Term in respect of a Product, ALEXION shall have sole control over, shall bear all costs and expenses of, and shall have sole discretion and decision-making authority with respect to, any further Development of such Product.
4.3 Regulatory Activities. ALEXION shall have sole control over, will own and control, and will bear all costs and expenses of, preparing and submitting applications for and obtaining and maintaining Regulatory Approvals for Products, including the filing of INDs for Products and communications with Regulatory Authorities. ZEALAND will [***] and provide [***] to ALEXION in its efforts to prepare and submit any submissions, documents or other correspondence to any Regulatory Authority in connection with obtaining, supporting, or maintaining Regulatory Approvals for any Products, including any preclinical packages, regulatory meeting requests, and any IND, NDA, sNDA and necessary drug master files (“DMFs”)
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
submissions (“Regulatory Documentation”). ZEALAND shall provide [***], and ALEXION will [***].
4.4 Commercialization. ALEXION shall have sole control over, shall bear all costs and expenses of, and shall have sole discretion and decision-making authority with respect to, the Commercialization of Products.
4.5 Diligence.
4.5.1 Development Diligence. ALEXION will, either itself or through an Affiliate or Sublicensee, use Commercially Reasonable Efforts in compliance with Applicable Law (including cGCP), to Develop and seek Regulatory Approval for [***]. ALEXION’s obligations under this Section begin on the date of the delivery by ZEALAND of Results and Deliverables containing all necessary data and information to prepare and submit an application for an IND to the applicable Regulatory Authority, in [***] in respect of such Product.
4.5.2 Deemed Satisfaction of ALEXION Diligence Obligations. On a Target-by-Target basis, and without in any way expanding ALEXION’s obligations under this Agreement, ALEXION’s expenditure of at least [***] dollars ($[***]) (including [***]) during any Calendar Year directly allocable to the Development, Regulatory Approval, or Commercialization of [***] Products directed to a given Target will be conclusive evidence that ALEXION has satisfied all ALEXION diligence obligations under this Agreement with respect to such Target for such Calendar Year.
4.5.3 Commercial Diligence. Upon receiving Regulatory Approval for a Product in [***], ALEXION will use, or will cause its Affiliates or Sublicensees to use, Commercially Reasonable Efforts to Commercialize such Product in such country in compliance with the requirements of all Applicable Laws. ALEXION shall have no other diligence obligations with respect to the Commercialization of Candidate Products.
4.5.4 ALEXION’s Development Reports. At least [***], ALEXION will prepare, and provide to ZEALAND, a report summarizing ALEXION’s [***] (the “Development Report”), including [***]. Upon ZEALAND’s reasonable request to ALEXION within [***] days after receipt of a Development Report, ZEALAND will [***]. The Development Report and [***] will be [***] under this Section 4.5.
4.5.5 Discontinuation. ALEXION reserves at all times the freedom and right, in its own discretion, to decide to discontinue any Development, Commercialization or Manufacturing of any Product, without any liability of ALEXION or its Affiliates to ZEALAND by reason of such discontinuation. In the event that ALEXION notifies ZEALAND there is a Discontinued Product, ZEALAND may request in writing that the Discontinued Product be returned to ZEALAND pursuant to Section 12.9.2, provided that ZEALAND shall consider in good faith ALEXION’s input with respect to any further Development or Commercialization by ZEALAND of such Discontinued Product.
4.5.6 Prudent Business Judgment. For the avoidance of doubt, nothing in this Section 4 will be construed to require ALEXION to act contrary to its prudent business judgment.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
4.6 Other [***]. [***] understands and acknowledges that [***] or [***], including [***], involving [***] this Agreement. [***] acknowledges and agrees that [***] this Agreement will be [***] that [***] will not [***] or [***] this Agreement.
4.7 ZEALAND Support. The Parties understand and agree that it may be necessary for ALEXION from time to time to seek guidance from ZEALAND following the Initial Development Period, and ZEALAND hereby agrees, upon reasonable request by ALEXION, to provide such guidance as a consultant. ZEALAND shall provide [***]. ZEALAND shall [***], and ALEXION shall [***].
5. MANUFACTURING
5.1 Clinical Trial and Commercial Supply. Subject to the agreement of (i) applicable CMO Supply Agreements to which ZEALAND is a Party and (ii) any supply agreement between ZEALAND and ALEXION, ZEALAND shall use diligent efforts to supply through permitted CMOs each Product for pre-clinical, clinical trial and commercial supply for a minimum period of [***] in accordance with this Article 5. ZEALAND and ALEXION shall agree prior to engaging any such CMO (which approval shall not be unreasonably withheld, conditioned or delayed), and ALEXION shall pay for such supply at a cost equal to [***]. Within [***] days following the Effective Date, the Parties shall discuss the basis on which any clinical and commercial supply of Candidate Products to ALEXION will be made including the terms that should be included in the agreement pursuant to which supply will be made and whether the relevant agreement with permitted CMOs (each a “CMO Supply Agreement”) will be entered into by ALEXION and/or ZEALAND and which of the Parties should lead such negotiations.
5.1.1 Clinical CMO Supply Agreement. Subject to Sections 5.1 and 5.4, the Parties shall negotiate diligently with permitted CMOs with a view to executing a CMO Supply Agreement for pre-clinical and Clinical Trial supply of each Product within [***] days following the Effective Date, such CMO Supply Agreement to contain mutually agreed supply terms and conditions consistent with the principles set forth on Schedule 5 hereto reasonably acceptable to ALEXION.
5.1.2 Commercial CMO Supply Agreement. Subject to Sections 5.1 and 5.4, the Parties shall negotiate diligently with permitted CMOs with a view to executing a CMO Supply Agreement for commercial supply of each Product within [***] following the Effective Date, such CMO Supply Agreement to contain mutually agreed supply terms and conditions consistent with the principles set forth on Schedule 5 hereto reasonably acceptable to ALEXION.
5.2 Where ZEALAND is to be the sole party to the CMO Supply Agreement, then ZEALAND shall lead negotiations provided that ZEALAND shall keep ALEXION fully informed of the progress of any such negotiations and shall provide copies of each draft of the CMO Supply Agreement received from such CMO. ZEALAND shall include all of ALEXION’s reasonable comments in such CMO Supply Agreement and shall use its reasonable efforts to include in such CMO Supply Agreement the provisions set out in Schedule 5, provided always that ZEALAND shall not be in breach of this Agreement if a CMO is not prepared to give such contract terms. ZEALAND will use its reasonable efforts to ensure that ALEXION is a third party beneficiary of all of
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
ZEALAND’s rights under the CMO Supply Agreement provided always that ZEALAND’s liability in relation to the supply of Product to ALEXION pursuant to such CMO Supply Agreement shall not [***] other than to the extent that [***]. In any event, ZEALAND shall not enter into any such CMO Supply Agreement without ALEXION’s prior written consent, both as to the identity of the CMO and the terms of the CMO Agreement.
5.3 Where ALEXION is to be a party to the CMO Supply Agreement, then ALEXION shall lead negotiations provided that ZEALAND shall give ALEXION all advice and cooperation reasonably requested by ALEXION, including on each draft of the CMO Supply Agreement received by ZEALAND from ALEXION. In the event that ZEALAND is also a party to the CMO Supply Agreement ZEALAND’s liability in relation to the supply of Product to ALEXION pursuant to such CMO Supply Agreement shall not [***] other than to the extent that [***]. In any event, ZEALAND shall not be required to be a party to a CMO Supply Agreement unless it has agreed to the identity of the CMO and the terms of the CMO Supply Agreement.
5.4 Manufacturing Step In. On a Product-by-Product basis, subject to the terms of the relevant CMO Supply agreement and any supply agreement between ALEXION and ZEALAND, ALEXION shall have the right, but not the obligation, at any time, for any reason or for no reason, to assume responsibility for Manufacturing any Product. Such Manufacturing responsibility may be executed directly by ALEXION or through one or more CMOs. ALEXION will provide ZEALAND [***] day notice of its intention to assume responsibility for Manufacturing and such notice will also act as a termination notice for the applicable CMO Supply Agreement, such termination to become effective upon the initiation of such Manufacturing by ALEXION or its CMOs (subject to the terms of the relevant CMO Agreement).
5.5 ZEALAND Support. The Parties understand and agree that it may be necessary for ALEXION from time to time to seek guidance from ZEALAND in relation to the Manufacture of Products, following the technology transfer contemplated by this Agreement, including with respect to scale-up activities, and ZEALAND hereby agrees to provide reasonable guidance as a consultant. ZEALAND shall provide [***]. ZEALAND shall [***], and ALEXION shall [***].
6. PAYMENTS AND ROYALTIES
6.1 One-time Upfront Payment; Equity Investment.
6.1.1 Upfront Payment. Within [***] Business Days following the Effective Date, ALEXION shall pay to ZEALAND a one-time and non-refundable, upfront payment of Twenty-Five Million Dollars ($25,000,000), which amount shall be payable in cash by wire transfer into an account designated in writing by ZEALAND.
6.1.2 Equity Investment. As of the Effective Date, the Parties have entered into a share issuance agreement, pursuant to which an ALEXION Affiliate will purchase ordinary shares of ZEALAND.
6.2 Target Option Exercise Fee. On an Optioned Target-by-Optioned Target basis, within [***] Business Days of ALEXION’s approval of a Discovery Plan under a Discovery/Development Program for such Optioned Target in accordance with Section 2.1.3 and 2.1.4, as full consideration for and payment of ZEALAND’s Discovery/Development Activities subject to Section 2.3 for
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
each such Optioned Target: ALEXION shall pay to ZEALAND a one-time non-refundable, non-reimbursable and non-creditable payment of Fifteen Million Dollars ($15,000,000) (“Target Option Exercise Fee”).
6.3 Milestone Payments.
6.3.1 Discovery/Development Program Milestones. On a Target-by-Target basis (except as expressly set forth below with respect to a [***] Product and a [***] Product), ALEXION shall make the one-time non-refundable milestone payments (each, a “Development Milestone Payment”) to ZEALAND set forth below in this Section 6.3.1 upon the first occurrence of the corresponding development or regulatory milestones events set forth below in this Section 6.3.1 (each, a “Development Milestone Event”) for the first Product directed to a given Target. As the [***] payment of Development Milestone Payments, such payments shall be payable with respect to each of a [***] Product] and a [***] Product that is the [***] Product and [***] Product to achieve the corresponding Development Milestone Events. In the event that one (1) or more Development Milestone Events in a given region are skipped for a Product, such skipped Development Milestone Events shall be payable upon achievement of the subsequent Development Milestone Event applicable to the same region. For clarity, a Development Milestone Event in one (1) region shall not be deemed to be skipped solely because of the achievement of any Development Milestone Event in a different region (e.g., a [***] in [***] shall not trigger any Development Milestone Payment applicable to [***] or [***] even if no Development Milestone Events had yet been achieved in [***] or [***]). ALEXION shall, within [***] days after ALEXION becomes aware of the achievement of a Development Milestone Event, notify ZEALAND in writing that such Development Milestone Event has occurred, and ZEALAND shall provide ALEXION with an Invoice related to the respective Development Milestone Payment. Each of the Development Milestone Payments shall be payable to ZEALAND by ALEXION within [***] days of receipt by ALEXION of such Invoice. The maximum amount payable by ALEXION with respect to the Lead Target, assuming there is [***], under this Section 6.3.1, if all Development Milestone Events occur, is [***] Dollars ($[***]). The maximum amount payable by ALEXION with respect to any Target other than the Lead Target under this Section 6.3.1, if all Development Milestone Events occur, is [***] Dollars ($[***]).
|
Development Milestone Event |
|
Development |
| |
|
Each of the following shall be a Development Milestone Event: [***] |
|
$ |
[***] |
|
|
Each of the following shall be a Development Milestone Event: [***] |
|
$ |
[***] |
|
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
|
Development Milestone Event |
|
Development |
| |
|
Each of the following shall be a Development Milestone Event: [***] |
|
$ |
[***] |
|
|
Each of the following shall be a Development Milestone Event: [***] |
|
$ |
[***] |
|
|
Each of the following shall be a Development Milestone Event: [***] |
|
$ |
[***] |
|
|
The first to occur of: (i) [***] or (ii) [***] shall be a Development Milestone Event for each of the following: [***] |
|
$ |
[***] |
|
|
Each of the following shall be a Development Milestone Event: [***] |
|
$ |
[***] |
|
|
Each of the following shall be a Development Milestone Event: [***] |
|
$ |
[***] |
|
|
Each of the following shall be a Development Milestone Event: [***] |
|
$ |
[***] |
|
6.3.2 Existing Lead Product Sales Milestones. ALEXION shall make a one-time, non-refundable sales milestone payment (each, an “Existing Product Sales Milestone Payment”) to ZEALAND upon the first occurrence of each of the corresponding milestone events based on worldwide, total Net Sales of all Products that are, contain or consist of any Existing Lead Molecules in a Calendar Year (each, an “Existing Product Sales Milestone Event”) as set forth in the table below in this Section 6.3.2. If a particular Existing Product Sales Milestone Event threshold triggering an Existing Product Sales Milestone Payment first occurs in the same Calendar Year as the first occurrence of one (1) or more lower Existing Product Sales Milestone Event thresholds, then any unpaid Existing Product Sales Milestone Payments for such lower Existing Product Sales Milestone Events shall be payable in addition to the Existing Product Sales Milestone Payment in respect of the higher Existing Product Sales Milestone Event. Net Sales of any Product not containing an Existing Lead Molecule shall not (1) count toward the achievement of any Existing Product Sales Milestone Event or (2) otherwise be considered in determining whether an Existing Product Sales Milestone Payment was triggered. For clarity: (i) no Existing Product Sales Milestone Payment shall be payable more than one (1) time for all Existing Lead Products, and (ii) the total of the Existing
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Product Sales Milestone Payments for the Lead Target payable during the Term shall not exceed $[***] Dollars ($[***]).
|
Existing Product Sales Milestone Events |
|
Existing Product |
| |
|
First Calendar Year in which aggregate annual Net Sales of Products containing Existing Lead Molecules in the Territory equal or exceed $[***] |
|
$ |
[***] |
|
|
First Calendar Year in which aggregate annual Net Sales of Products containing Existing Lead Molecules in the Territory $[***] |
|
$ |
[***] |
|
|
First Calendar Year in which aggregate annual Net Sales of Products containing Existing Lead Molecules in the Territory $[***] |
|
$ |
[***] |
|
|
First Calendar Year in which aggregate annual Net Sales of Products containing Existing Lead Molecules in the Territory $[***] |
|
$ |
[***] |
|
|
First Calendar Year in which aggregate annual Net Sales of Products containing Existing Lead Molecules in the Territory $[***] |
|
$ |
[***] |
|
6.3.3 Created Lead Product and Optioned Product Sales Milestones. ALEXION shall make on a Target-by-Target basis, a one-time, non-refundable sales milestone payment (each, a “Sales Milestone Payment”) to ZEALAND upon the first occurrence of each of the corresponding milestone events based on the worldwide, total Net Sales of all Created Lead Products or Optioned Products in each case as directed to a given Target, in a Calendar Year (each, a “Sales Milestone Event”) as set forth in the table below in this Section 6.3.3. If a particular Sales Milestone Event threshold triggering a Sales Milestone Payment first occurs in the same Calendar Year as the first occurrence of one (1) or more lower Sales Milestone Event thresholds, then any unpaid Sales Milestone Payments for such lower Created Lead Product or Optioned Product Sales Milestone Events shall be payable in addition to the Sales Milestone Payment in respect of the higher Sales Milestone Event. Net Sales of (a) any Product which is not a Created Lead Product shall not (1) count toward the achievement of any Sales Milestone Event for Created Lead Products or (2) otherwise be considered in determining whether a Sales Milestone Payment was triggered for Created Lead Products and (b) Optioned Products not directed to a given Target shall not (1) count toward the achievement of any Sales Milestone Event for such given Target or (2) otherwise be considered in determining whether a Sales Milestone Payment was triggered for such given Target. For clarity: (i) no Sales Milestone Payment for Created Lead Products shall be payable more than one (1) time for all Created Lead Products and no Sales Milestone Payments for Optioned Products shall be payable more than one (1) time for each Optioned Target, and (ii) the total of Sales Milestone Payments payable for all Created Lead Products shall not exceed [***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Dollars ($[***]), and (iii) on a Target-by-Target basis the Total of Sales Milestone Payments for all Optioned Products directed to a given Target shall not exceed [***] Dollars ($[***]).
Created Product and Optioned Product Sales
Milestone Events and Payments on a
Target-by-Target basis
|
Sales Milestone Event |
|
Sales Milestone |
| |
|
First Calendar Year in which aggregate annual Net Sales of (i) in the case of the Lead Target, all Created Lead Products or (ii) in the case of other Targets, all Optioned Products directed to such Target in the Territory equal or exceed $[***] |
|
$ |
[***] |
|
|
First Calendar Year in which aggregate annual Net Sales of (i) in the case of the Lead Target, all Created Lead Products or (ii) in the case of other Targets, all Optioned Products directed to such Target in the Territory equal or exceed $[***] |
|
$ |
[***] |
|
|
First Calendar Year in which aggregate annual Net Sales of (i) in the case of the Lead Target, all Created Lead Products or (ii) in the case of other Targets, all Optioned Products directed to such Target in the Territory equal or exceed $[***] |
|
$ |
[***] |
|
|
First Calendar Year in which aggregate annual Net Sales of (i) in the case of the Lead Target, all Created Lead Products or (ii) in the case of other Targets, all Optioned Products directed to such Target in the Territory equal or exceed $[***] |
|
$ |
[***] |
|
|
First Calendar Year in which aggregate annual Net Sales of (i) in the case of the Lead Target, all Created Lead Products or (ii) in the case of other Targets, all Optioned Products directed to such Target in the Territory equal or exceed $[***] |
|
$ |
[***] |
|
6.4 Sales Milestone Payment Terms. Subject to Sections 6.3.2 and 6.3.3, ALEXION will make any Existing Product Sales Milestone Payment or Sales Milestone Payment within [***] days after the end of the Calendar Quarter within which the corresponding Existing Product Sales Milestone Payment or Sales Milestone Event first occurred.
6.5 Royalties.
6.5.1 Existing Lead Products Royalties. On an Existing Lead Product-by-Existing Lead Product and country-by-country basis and subject to Section 6.9 ALEXION shall pay royalties to ZEALAND based on total worldwide Net Sales of a given Existing Lead Product (excluding Net Sales of all other Products including other Existing Lead
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Products) in a given Calendar Year (“Per Existing Lead Product Sales”) during the Royalty Term applicable to such Existing Lead Product and country at the rates set forth in the table set forth below. In the event that a Product contains or consists of an Existing Lead Product and a Created Lead Product and/or an Optioned Product, such Product shall be an Existing Lead Product for the duration of the Royalty Term applicable to such Existing Lead Product and country and, to the extent that the Royalty Term applicable to the relevant Created Lead Product and/or an Optioned Product contained in or comprising such Product and country has not expired, shall thereafter be a Created Lead Product or Optioned Product (as relevant) for the duration of the Royalty Term applicable to such Created Lead Product or Optioned Product (as relevant) and country.
|
Per Existing Lead Product Annual Net Sales |
|
Royalty Rate |
|
|
Up to and including [***] Dollars ($[***]) |
|
[***] |
% |
|
Portion that is greater than [***] Dollars ($[***]) up to and including [***] Dollars ($[***]) |
|
[***] |
% |
|
Portion that is greater than [***] Dollars ($[***]) |
|
[***] |
% |
6.5.2 Created Lead Products and Optioned Product Royalties. On a Created Lead Product-by-Created Lead Product or Optioned Product-by-Optioned Product basis and in each case on a country-by-country basis and subject to Section 6.9, ALEXION shall pay royalties to ZEALAND based on aggregate worldwide Net Sales of a given Created Lead Product or Optioned Product (excluding in each case Net Sales of all other Products including other Created Lead Products or Optioned Products) in a given Calendar Year (as applicable, “Per Created Lead Product Sales” or “Per Optioned Lead Product Sales”) during the Royalty Term applicable to such Created Lead Product or Optioned Product and country at the rates set forth in the following table.
|
Per Created Lead Product or Per Optioned Product Sales |
|
Royalty Rate |
|
|
Up to and including [***] Dollars ($[***]) |
|
[***] |
% |
|
Portion that is greater than [***] Dollars ($[***]) up to and including [***] Dollars ($[***]) |
|
[***] |
% |
|
Portion that is greater than [***] Dollars ($[***]) up to and including [***] Dollars ($[***]) |
|
[***] |
% |
|
Portion that is greater than [***] Dollars ($[***]) |
|
[***] |
% |
6.6 Royalty Calculation Example. By way of example, in a given Calendar Year and without giving effect to any potential reductions under this Agreement, if the Per Existing Lead Product Sales of a given Existing Lead Product for which a royalty is payable pursuant to Section 6.5.1 is $[***] (excluding any Net Sales attributable to any other Product whether or not an Existing Lead Product), the following royalty payment would be payable for those Net Sales: ([***]% of $[***]) + ([***]% of $[***]) = $[***].
6.7 Royalty Term. ALEXION’s obligation to pay royalties shall begin, on a country-by-country and Product-by-Product basis, with the First Commercial Sale of such Product in such country, and
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
shall expire, on a country-by-country and Product-by-Product basis, upon the expiry of the Royalty Term in such country. Upon expiration (but not upon the earlier termination of this Agreement) of the Royalty Term for a given Product in a given country (a) no further royalties shall be payable in respect of sales of such Product in such country, and (b) the licenses granted to ALEXION under Section 9.2.2, and 9.2.4 with respect to such Product in such country shall automatically become fully paid-up, irrevocable and royalty free on a non-exclusive basis. For the sake of clarity, no multiple royalties shall be payable even if more than one Valid Claim Covers a Product during the Royalty Term.
6.8 Royalty Reports; Payments.
6.8.1 Royalty Reports. Within [***] days following the end of each Calendar Quarter during which any royalty payments are owed, ALEXION shall submit to ZEALAND a written report on a Product-by-Product and country-by-country basis of Net Sales of Products sold, in the currency for which such Products were sold (and, if the currency of sale was not Dollars, also in Dollars), by or on behalf of ALEXION, its Affiliates and Sublicensees during such Calendar Quarter, and the royalty payments payable on such Net Sales in sufficient detail to permit confirmation of the accuracy of royalty payments payable hereunder.
6.8.2 Royalty Payments. Royalties will be payable on a Calendar Quarter basis and any such payments shall be made within [***] days after the end of the Calendar Quarter during which the applicable Net Sales of Products occurred.
6.8.3 Payment Method. All payments to be made between the Parties under this Agreement shall be made in Dollars and may be paid by wire transfer, or electronic funds transfer in immediately available funds to a bank account designated by ZEALAND or ALEXION, as applicable.
6.8.4 Currency Conversion. With respect to sales of Products invoiced in United States Dollars, all such amounts shall be expressed in United States Dollars. With respect to sales of Products invoiced in a currency other than United States Dollars, all such amounts shall be expressed both in the currency in which the sale is invoiced and in the United States Dollar equivalent. All royalties payable hereunder shall be calculated based on ALEXION Net Sales expressed in United States Dollars. For purposes of this Section 6.8.4, the United States Dollar equivalent shall be calculated using the rate of exchange at the close of business on the date ALEXION records the net revenue from the ALEXION customer. Each daily exchange rate will be the Bloomberg mid-market rate as of the 5:00 PM New York close, or, if not so available, as otherwise agreed by the Parties.
6.9 Royalty Adjustments.
6.9.1 Generic Competition. In the event that there is Generic Competition with respect to a Product in a country during [***], then royalties payable by ALEXION pursuant to Section 6.5 for such Product in such country shall in such [***] shall, subject to Section 6.9.4, be reduced to [***] percent ([***]%) of the royalty rate otherwise payable under
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Section 6.5 for such Product in such [***] and for so long as there is Generic Competition at such level with respect to such Product in such country.
6.9.2 No Valid Claim. During the applicable Royalty Term for a Product in a country, if such Product is sold in such country is not Covered by a Valid Claim of any Licensed Patent Right in the such country at the time of such sale, then the royalties payable for such Product in such country shall be reduced to a percentage which is [***] percent ([***]%) of the royalty rate otherwise payable.
6.9.3 Third Party Royalties Offset. In the event that ALEXION enters into an agreement with a Third Party to obtain a license under a Patent Right that is [***] a Candidate Product in a country, then, subject to Section 6.9.4, the amounts of any royalties due to ZEALAND for the respective Product in such country shall be reduced by [***] percent ([***]%) of the [***] paid to such Third Party in respect of such agreement.
6.9.4 Application of Royalty Adjustments. On a Product-by-Product and country-by-country basis, in any particular [***], the reduction under Section 6.9.1 shall be applied first to the amount of royalties otherwise owed under Section 6.5.1 and 6.5.2 for such Product, then the reduction under Section 6.9.2 shall be applied, and finally the offset under Section 6.9.3 shall be applied. In no event, shall the royalties payable to ZEALAND for a Product in a given country and [***], as reduced by Sections 6.9.1—6.9.3 above, be reduced to an amount less than a royalty of less than [***] percent ([***]%) of the amount otherwise owed under Section 6.5.1 and 6.5.2. ALEXION may carry forward any offsets permitted under Section 6.9.3 that are incurred or accrued in a [***] but are not applied against royalties due to ZEALAND for such Product in such country in such [***] as a result of the foregoing floor and apply such amounts against royalties due to ZEALAND for such Product in such country in any [***] (subject to the minimum floor set forth in this Section 6.9.4) until the amount of such offset under Section 6.9.3 has been fully applied against royalties due to ZEALAND for such Product in such country.
6.9.5 ZEALAND Third Party Agreements. ZEALAND shall [***]. In the event that ZEALAND [***], ZEALAND shall [***]. ALEXION shall [***]. To the extent that ALEXION [***], the [***].
6.10 Late Payments. If ALEXION fails to make any undisputed payment due under this Agreement as provided herein on or before the date that such payment is due, then the amount of such payment will bear interest at an annual rate equal to [***] (the “U.S. Prime Rate”) which applied on the first date on which such payment was delinquent or (ii) the maximum rate permitted by Applicable Law, in each case, [***]. If the U.S. Prime Rate is [***], the Parties will [***].
6.11 Taxes. All payments are exclusive of value added taxes, sales taxes, consumption taxes and other similar taxes as required by Applicable Law (the “Indirect Taxes”). If any Indirect Taxes are chargeable in respect of any payments, the paying Party shall pay such Indirect Taxes at the applicable rate in respect of such payments following receipt, where applicable, of an Indirect Taxes invoice in the appropriate form issued by the receiving Party in respect of those payments. The Parties shall issue invoices for all amounts payable under this Agreement consistent with Indirect Tax requirements and irrespective of whether the sums may be netted for settlement
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
purposes. If the Indirect Taxes originally paid or otherwise borne by the paying Party are in whole or in part subsequently determined not to have been chargeable, all necessary steps will be taken by the receiving Party to receive a refund of these undue Indirect Taxes from the applicable governmental authority or other fiscal authority and any amount of undue Indirect Taxes repaid by such authority to the receiving Party will be transferred to the paying Party within [***] days of receipt. Notwithstanding the foregoing, [***].
6.12 Withholding Taxes. The Parties agree that ALEXION and/or its Affiliates shall make each payment paid under this Agreement for the benefit of ZEALAND and/or its Affiliates from an account owned by an entity domiciled within [***] (as determined for Tax purposes under Applicable Law) to an account owned by ZEALAND and/or its Affiliates in [***]. Subject to the foregoing, if Applicable Law requires withholding by ALEXION and/or its Affiliates of any Taxes on account of any royalties or any other payments paid under this Agreement for the benefit of ZEALAND and/or its Affiliates, ALEXION and/or its Affiliates may deduct the amount of such Taxes from the applicable payment that is otherwise payable to ZEALAND and/or its Affiliates and such withheld amount shall be remitted by ALEXION and/or its Affiliates to the proper governmental authorities on behalf of ZEALAND and/or its Affiliates. Any Taxes that are so deducted, withheld and remitted to the proper governmental authorities will be treated for all purposes of this Agreement as having been paid to ZEALAND and/or its applicable Affiliate(s). Official receipts of the remittance by ALEXION and/or its Affiliates of any such withholding Tax shall be reasonably promptly secured and sent by ALEXION and/or its Affiliates to ZEALAND and/or its Affiliates as evidence of such payment. The Parties shall reasonably cooperate with each other in claiming exemptions from, or reductions of, any withholding Taxes imposed on ZEALAND and/or its Affiliates under the provisions of any Applicable Law. If ALEXION elects to make any payment, or to cause any of its Affiliates to make any payment, under this Agreement for the benefit of ZEALAND and/or its Affiliates from an account owned by an entity domiciled in a jurisdiction other than [***] for Tax purposes under Applicable Law, and such payment results in ZEALAND and/or its Affiliates being subject to a withholding tax that it would not have been subject to if the payment had instead been subject to [***] withholding tax (if any) under Applicable Law, as applicable, ALEXION shall pay to ZEALAND such an amount that would result in ZEALAND receiving the same amount under this Agreement as it would have received under this Agreement, had such payment been subject to [***] withholding tax (if any) under Applicable Law, as applicable.
6.13 Financial Audits.
6.13.1 Audits by ZEALAND. ALEXION shall [***] to permit ZEALAND to [***]. ZEALAND shall have the right to cause an independent, certified public accountant of nationally recognized standing selected by ZEALAND and reasonably acceptable to ALEXION (the “Auditor”) to [***]. The accounting firm shall [***]. The Auditor will [***]. The report of the Auditor will [***]. Any disputes with respect to the findings of such Auditor may be [***]. ALEXION shall [***]. If such final Auditor’s report [***], ALEXION shall have the right to [***]. Upon the expiration of [***], the calculation of [***] shall [***]. Any disclosures or reports disclosed to ZEALAND under this Section 6.13.1 shall [***].
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
6.13.2 Audits by ALEXION. Upon [***], ZEALAND shall [***]. An examination by ALEXION under this Section 6.13.2 shall [***]. The accounting firm shall [***]. The auditor will [***]. Upon completion of the audit, the accounting firm shall provide both ZEALAND and ALEXION with a written report disclosing [***]. Such report shall [***]. Any disputes with respect to the findings of such Auditor may be [***]. ALEXION shall [***]. ALEXION shall [***]. If such final Auditor’s report [***], ALEXION shall have the right to [***]. Any disclosures or reports disclosed to ALEXION under this Section 6.13.2 shall [***].
7. REPRESENTATIONS, WARRANTIES AND COVENANTS
7.1 Mutual Representations, Warranties and Covenants of the Parties. Each Party represents and warrants to the other Party as of the Effective Date, and as applicable, covenants to the other Party, that:
7.1.1 it is validly existing and in good standing under the Applicable Laws of the jurisdiction of its incorporation and has the full right, power and authority to enter into this Agreement, conduct the activities allocated to it under this Agreement, grant the licenses and assign the rights under this Agreement and disclose such information and Know-How that is disclosed in performance of its obligations under this Agreement;
7.1.2 this Agreement has been duly executed by it and is legally binding upon it, enforceable in accordance with its terms, and does not conflict with any agreement, instrument or understanding, oral or written, to which it is a party or by which it may be bound, nor violate any material Applicable Law of any court, governmental body or administrative or other agency having jurisdiction over it;
7.1.3 neither it, nor any of its Affiliates are party to any agreements, oral or written, that conflict with its obligations under this Agreement; and
7.1.4 neither it, nor any of its Affiliates, have been Debarred, and during the Term, neither it, nor any of its Affiliates shall use, in any capacity in connection with the obligations to be performed under this Agreement, any person or entity who has been Debarred.
7.2 Additional Representations and Warranties of ZEALAND. ZEALAND represents and warrants to ALEXION as of the Effective Date that:
7.2.1 all Licensed Patent Rights existing as of the Effective Date are set forth on APPENDIX 5 hereto, and all Licensed Patent Rights (i) are being diligently prosecuted in the respective patent offices in the Territory in accordance with Applicable Law, (ii) all applicable fees have been paid on or before the due date for such payments, and (iii) excluding any information submitted to or received from any patent office in the Territory or as disclosed to ALEXION, ZEALAND is not aware of information “material to the patentability” (as such term is used in 37 C.F.R 1.56) of any claim under the Licensed Patent Rights;
7.2.2 ZEALAND is the sole and exclusive owner of, or Controls, the Existing Lead Molecules and the Licensed Intellectual Property and none of the Licensed Intellectual Property or
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Existing Lead Molecules constitutes or results from any misappropriated Intellectual Property;
7.2.3 ZEALAND has the necessary rights to grant the licenses under the Licensed Intellectual Property to ALEXION under this Agreement;
7.2.4 ZEALAND has the necessary rights to the Licensed Intellectual Property licensed to ALEXION under this Agreement to conduct the Discovery/Development Programs (including any Discovery Plan and Development Plan thereunder), the [***] Work Plan, and the Manufacturing in the manner contemplated under the applicable Research Work Plans and Development Plans;
7.2.5 it is entitled to grant the licenses to ALEXION under this Agreement, and has taken all appropriate measures (including having appropriate agreements in place with any person or contractor that will be involved in Discovery/Development Activities, or Manufacturing) under all Applicable Laws to grant such licenses and assign such rights;
7.2.6 neither ZEALAND, nor any of its Affiliates have previously assigned, transferred, conveyed or otherwise encumbered its right, title or interest in or to the Existing Lead Molecules, the Licensed Intellectual Property in a manner that would prevent (i) ZEALAND from performing the Discovery/Development Activities in accordance with the Development Plans or the Discovery/Development Activities in accordance with the Development Plans, the Manufacturing obligations as they exist at the Effective Date, or assigning and granting the rights to ALEXION set forth in Section 9; or (ii) ALEXION or its Affiliates, subcontractors, CMOs and Sublicensees from researching, Developing, Manufacturing or Commercializing Products or from otherwise exploiting its rights and licenses granted or assigned by ZEALAND hereunder;
7.2.7 there are no pending claims, judgments or settlements against, or amounts owed to a Third Party by ZEALAND or any of its Affiliates in connection with a claim, judgment or settlement, involving the Existing Lead Molecules or Licensed Intellectual Property licensed by ZEALAND to ALEXION under this Agreement and ZEALAND has not received written notice threatening any such claims, judgments or settlements;
7.2.8 to ZEALAND’s knowledge, all information disclosed to ALEXION by ZEALAND relating to the Existing Lead Products, the Licensed Intellectual Property and the materials and methods to be employed by ZEALAND in the execution of the Development Plan and this Agreement is, at the time of disclosure, accurate in all material respects;
7.2.9 to ZEALAND’s knowledge, no person is infringing or threatening to infringe or misappropriate or threatening to misappropriate the Licensed IP;
7.2.10 each person who has or has had any rights in or to any the Existing Lead Molecules or Licensed IP in each case existing as of the Effective Date and owned by ZEALAND has assigned and has executed an agreement assigning its entire right, title and interest in and to such Licensed IP to ZEALAND and as far as ZEALAND is aware, each person who has or has had any rights in or to any Existing Lead Molecules or Licensed IP in each
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
case existing as of the Effective Date and licensed to ZEALAND by a Third Party has assigned and has executed an agreement assigning its entire right, title and interest in and to such Existing Lead Molecules or Licensed IP to such Third Party;
7.2.11 other than the Intellectual Property that is licensed or assigned by ZEALAND to ALEXION in this Agreement and as disclosed in Section 7.2.7, ZEALAND is not aware and has not received any written notice of any Intellectual Property (including any Intellectual Property Controlled by a Third Party) that would be infringed, either by ALEXION or ZEALAND, in the course of conducting Discovery/Development Activities, or Manufacturing, or ALEXION’s use, Manufacture, Commercialization or other exploitation of any Existing Lead Molecule, Candidate Product or Product as contemplated hereunder;
7.2.12 Schedule 1.63 sets forth a complete list of all Existing Lead Molecules owned or Controlled by ZEALAND as of the Effective Date.
7.3 Covenants. Each Party shall, and shall ensure that its Affiliates and Sublicensees, obtain written agreements from any and all persons involved in or performing any Discovery/Development Activities, Manufacturing or otherwise involved in the performance of a Party’s obligations under this Agreement assigning such persons’ right, title and interest in and to any Licensed IP, Results, or Collaboration IP to such Party prior to any such person performing such activities. During the Term, ZEALAND shall maintain sufficient resources to perform the Discovery/Development Activities and Manufacturing ascribed to it in accordance with this Agreement. Neither ZEALAND, nor any of its Affiliates, shall assign, transfer, convey or otherwise encumber during the Term, its right, title or interest in or to any Existing Lead Molecule, Candidate Product, the Licensed Intellectual Property, Collaboration IP, Results or Deliverables in a manner that would prevent (i) ZEALAND from performing the Discovery/Development Activities in accordance with the Development Plans or the Discovery/Development Activities in accordance with the Development Plans, or assigning and granting the rights to ALEXION set forth in Section 9; or (ii) ALEXION or its Affiliates, subcontractors and Sublicensees from researching, Developing, Manufacturing or Commercializing Products or from otherwise exploiting its rights and licenses granted or assigned by ZEALAND hereunder.
7.4 During the Term, ZEALAND shall ensure that all laboratories, rooms and equipment and the conduct of all activities to be carried out by on behalf of ZEALAND in connection with the Discovery/Development Programs or the Discovery/Development Programs, comply with the Development Plans, the Development Plans, and Applicable Laws (including, as applicable GLP).
7.5 DISCLAIMER OF WARRANTIES. EXCEPT AS OTHERWISE EXPRESSLY SET FORTH HEREIN, NEITHER PARTY MAKES ANY REPRESENTATION OR EXTENDS ANY WARRANTIES OF ANY KIND, EITHER EXPRESS OR IMPLIED, INCLUDING ANY IMPLIED WARRANTIES OF TITLE, NON-INFRINGEMENT, MERCHANTABILITY AND FITNESS FOR A PARTICULAR PURPOSE. IN PARTICULAR, NEITHER PARTY MAKES ANY REPRESENTATION OR EXTENDS ANY WARRANTY THAT THE CANDIDATE PRODUCTS OR PRODUCT(S) WILL BE SUCCESSFULLY DEVELOPED HEREUNDER.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
7.6 NO CONSEQUENTIAL DAMAGES. EXCEPT FOR DAMAGES RESULTING FROM BREACHES OF SECTION 2.5 (ADDITIONAL TARGETS), SECTION 2.6 (EXCLUSIVITY) OR ARTICLE 8 (CONFIDENTIALITY), OR INDEMNIFIABLE CLAIMS UNDER ARTICLE 11 (INDEMNIFICATION), IN NO EVENT WILL EITHER PARTY HAVE ANY CLAIMS AGAINST OR LIABILITY TO THE OTHER PARTY WITH RESPECT TO ANY INDIRECT, PUNITIVE, SPECIAL, INCIDENTAL, OR CONSEQUENTIAL DAMAGES (INCLUDING ANY CLAIMS FOR LOST PROFITS OR REVENUES) ARISING UNDER OR IN CONNECTION WITH THIS AGREEMENT UNDER ANY THEORY OF LIABILITY, EVEN IF SUCH PARTY HAS BEEN INFORMED OR SHOULD HAVE KNOWN OF THE POSSIBILITY OF SUCH DAMAGES.
8. CONFIDENTIALITY
8.1 Confidential Information. “Confidential Information” means all non-public Know-How or other information, including proprietary materials or information, transferred, disclosed or otherwise made available by or on behalf of a Party (the “Disclosing Party”) to the other Party or its permitted recipients (the “Receiving Party”) prior to, on or after the Effective Date, whether or not patentable and whether or not disclosed in written, oral graphical, machine-readable, electronic or other form or otherwise observed by the Receiving Party, and whether or not such information is marked as confidential or proprietary. It is understood and agreed by the Parties that:
8.1.1 The terms and conditions of this Agreement will be considered Confidential Information of both Parties and kept confidential by each of the Parties as set forth in this Article 8.
8.1.2 (i) the identities of the Targets, Existing Lead Molecules, Created Lead Molecules, the Candidate Products, and the Products and (ii) all [***], except insofar as any of the foregoing in (i) or (ii) solely relate to Discontinued Products (which shall be considered the Confidential Information of ZEALAND, with ZEALAND deemed to be the Disclosing Party and ALEXION deemed to be the Receiving Party with respect thereto), shall be considered the Confidential Information of ALEXION, with ALEXION deemed to be the Disclosing Party and ZEALAND deemed to be the Receiving Party with respect thereto.
8.2 Non-Disclosure and Non-Use Obligation. Except as otherwise expressly set forth herein, the Receiving Party shall keep the Confidential Information of the Disclosing Party confidential using at least the same degree of care with which the Receiving Party holds its own confidential information and shall not (i) disclose such Confidential Information to any person or entity without the prior written approval of the Disclosing Party, except, solely to the extent necessary to exercise its rights, perform its obligations under this Agreement, or to evaluate the economic value of and otherwise assess this Agreement (e.g., by sharing portfolio information) including any Results, Deliverables, Candidate Products and Products, to its employees, Affiliates, Sublicensees and contractors, consultants or agents who have a need to know such Confidential Information, all of whom will be similarly bound by the provisions of this Article 8 and for whom the Disclosing Party will be responsible, or (ii) use such Confidential Information for any purpose other than for the purposes contemplated by this Agreement. The Receiving Party will use diligent efforts to cause the foregoing entities and persons to comply with the restrictions on use and disclosure set
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
forth in this Section 8.2, and shall be responsible for ensuring that such entities and persons maintain the Disclosing Party’s Confidential Information in accordance with this Article 8.
8.3 Return of Confidential Information. Upon the expiration or termination of this Agreement, the Receiving Party shall return (or, as directed by the Disclosing Party, destroy) all Confidential Information of the Disclosing Party to the Disclosing Party that is in the Receiving Party’s possession or control, provided, however, one (1) copy may be retained and stored solely for the purpose of determining its obligations under this Agreement, provided that the non-disclosure and non-use obligation under this Article 8 shall continue to apply to any such copies. In addition, the Receiving Party shall not be required to return or destroy Confidential Information contained in any computer system back-up records made in the ordinary course of business; provided, that such Confidential Information may not be accessed without the Disclosing Party’s prior written consent or as required by Applicable Law.
8.4 Exemption. The foregoing confidentiality and non-use obligations shall not apply, to the extent that the Receiving Party can so demonstrate, to: (i) information already in the possession of the Receiving Party prior to its disclosure by the Disclosing Party as evidenced by written records, (ii) information which comes into the public domain by publication or otherwise through no breach of the obligations of confidentiality and non-use hereunder by the Receiving Party, including with respect to Section 10.1, (iii) information which has been disclosed to the Receiving Party from another source free from any obligation of confidentiality and which was not directly or indirectly obtained from the Disclosing Party, or (iv) information which is developed independently by employees, subcontractors, consultants or agents of the Receiving Party or any of its Affiliates without use of or reliance upon the Disclosing Party’s Confidential Information, as evidenced by contemporaneous written records.
8.5 Permitted Disclosures. In addition to the exceptions contained in Sections 8.2 and 8.4, the Receiving Party may disclose Confidential Information of the Disclosing Party to the extent (and solely to the extent) that such disclosure is reasonably necessary in the following instances:
8.5.1 to comply with Applicable Law (including any securities law or regulation or the rules of a securities exchange pursuant to Section 8.6 below) or the order of a court of competent jurisdiction, provided that, where legally permissible, the Receiving Party promptly notifies the Disclosing Party of such obligation sufficiently prior to making such disclosure, so as to allow the Disclosing Party adequate time to take whatever action it may deem appropriate to protect the confidentiality of the information to be disclosed, and the information to be disclosed and fully cooperates with the Disclosing Party, if so requested, in maintaining the confidentiality of such information by applying for a protective order or any similar legal instrument. In any event, the compelled Receiving Party shall only disclose such Confidential Information to the extent required under Applicable Law and shall continue to treat such information as Confidential Information for all other purposes under this Agreement;
8.5.2 to prosecute or defend litigation or to otherwise exercise its rights or perform its obligations in Section 9.4.2 below, Regulatory Approvals and other regulatory filings and communications, to file or prosecute patent applications or regulatory filings as contemplated by this Agreement so long as there is [***] days’ prior written notice before
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
filing and to enforce Patent Rights in connection with the Receiving Party’s rights and obligations pursuant to this Agreement; and
8.5.3 to allow the Receiving Party to exercise its rights and perform its obligations hereunder provided that such disclosure is covered by terms of confidentiality and non-use at least as restrictive as those set forth herein.
8.6 Disclosure of Agreement. Either Party may disclose the terms of this Agreement (a) to the extent required or advisable to comply with the rules and regulations promulgated by the United States Securities and Exchange Commission or any equivalent governmental agency in any country in the Territory, provided that such Party shall submit a confidential treatment request in connection with such disclosure and shall submit with such confidential treatment request only such redacted form of this Agreement as may be mutually agreed in writing by the Parties; (b) to bona fide prospective or actual acquirers, permitted assignees or merger candidates or to bona fide existing or potential investment bankers, investors and lenders, or financing sources, in a redacted form of this Agreement or its terms which shall be redacted in respect of (i) the identity of Targets, (ii) Development Milestone Events, Existing Product Sales Milestone Events or Sales Milestone Events and Development Milestone Payments, Existing Product Sales Milestone Payments or Sales Milestone Payments under Section 6.3 (other than total potential milestones per Target), and (iii) royalties and computations in Section 6.5; provided that, either Party may disclose an unredacted form of this Agreement (including the foregoing information regarding Targets and payments) to bona fide potential or actual acquirers and permitted assignees or merger candidates, but only at such time as the (x) Disclosing Party acting reasonably and in good faith believes, that the contemplated merger or acquisition is likely to take place with such potential transaction partner to whom ZEALAND wishes to disclose the Agreement to and (y) such transaction partner has executed with such Disclosing Party a confidentiality agreement with terms restricting such partner’s use solely for evaluating the potential transaction, restricting access to such individuals as may need to know the information for such evaluation and strictly prohibit disclosure of such terms by the partner and otherwise as protective with respect to Confidential Information as contained herein; and provided further, that in the case of disclosure by ZEALAND to a Competitor, ZEALAND may only disclose such unredacted Agreement or its terms in a secure data room accessible only on a need to know basis with watermarking and controlled access with respect to the proposed transaction with such Competitor and in accordance with the foregoing (x) and (y) and (c) to the extent necessary to perform obligations or exercise rights under this Agreement, any sublicensee, collaborator or potential sublicensee or potential collaborator of such Party, provided that any sublicensee, collaborator or potential sublicensee or collaborator agree in writing to be bound by obligations of confidentiality and non-use no less protective of the Disclosing Party than those set forth in this Article 8.
8.7 Use of Name and Logo. Subject to Section 10.2, neither ZEALAND nor ALEXION shall use the other Party’s or its Affiliates’ name or logo in any label, press release or product advertising, or for any other promotional purpose, without first obtaining the other Party’s written consent.
8.8 Engaging Individuals. Each Party hereby agrees that all individuals (including Sublicensees, subcontractors, and CMOs) engaged to perform any activities under this Agreement (including students, research assistants, etc.) shall be contractually bound by confidentiality obligations at
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
least as restrictive as the obligations of confidentiality and non-use set forth in this Article 8 prior to performing such activities.
8.9 Survival. This Article 8 shall survive the expiry or termination of this Agreement and shall remain in full force and effect for [***] years after the expiry or termination of this Agreement.
9. INTELLECTUAL PROPERTY
9.1 Ownership.
9.1.1 Background IP. Each Party shall be and shall remain the owner of any Intellectual Property that (i) it developed or owned prior to the Effective Date of this Agreement or (ii) it independently develops or acquires during the Term outside the scope of Discovery/Development Activities, or other performance of such Party’s obligations under this Agreement.
9.1.2 Ownership. As between the Parties (a) ZEALAND will own all right, title and interest in and to any Collaboration IP and Licensed IP conceived, reduced to practice or otherwise discovered solely by or on behalf of ZEALAND or its Affiliates or subcontractors and any Patent Rights that Cover or claim such Collaboration IP or Licensed IP and (b) the Parties will jointly own all right, title and interest in and to any Licensed IP or Collaboration IP conceived, reduced to practice or otherwise discovered jointly by or on behalf of ZEALAND or its Affiliates and subcontractors on the one hand and ALEXION or its Affiliates and subcontractors on the other hand including any Patent Rights that disclose or claim such joint Licensed IP or Collaboration IP (collectively, “Joint Patent Rights”). Subject to this Agreement including the licenses granted herein, each Party may exercise its ownership rights in and to any such jointly owned Licensed IP or Collaboration IP, including Joint Patent Rights, including the right to license and sublicense or otherwise to exploit, transfer or encumber its ownership interest, without an accounting or obligation to, or consent required from, the other Party. Without limiting a Party’s obligations hereunder, each Party shall require its employees (and Affiliates or subcontractors and employees thereof) performing the Discovery/Development Activities, Manufacturing or the Discovery/Development Programs on its behalf or with access to the other Party’s Background IP or Confidential Information to assign to such Party any Licensed IP or Collaboration IP and to cooperate with such Party in connection with obtaining patent protection therefor.
9.1.3 Disclosure of Inventions by Personnel. Where any personnel of a Party (including any employee, agent or consultant hired by a Party, and any person or entity performing work on behalf of a Party or such Party’s Affiliates) is involved in or performing activity that may result in Licensed IP or Collaboration IP, including in performing the Discovery/Development Activities, Manufacturing, or otherwise involved in the performance of a Party’s obligations under this Agreement, such Party shall ensure that such personnel informs such Party about and assigns to such Party any Licensed IP or Collaboration IP and such Party shall promptly inform the other Party of the same. The receiving Party shall treat any information on such Licensed IP or Collaboration IP as Confidential Information in accordance with Article 8 of this Agreement.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
9.1.4 Inventorship. Inventorship of patentable Inventions conceived, reduced to practice or otherwise discovered in the course of conducting the Discovery/Development Activities, Manufacturing, or otherwise arising out of a Party’s performance of its obligations under this Agreement shall be determined in accordance with U.S. patent laws.
9.2 Licenses.
9.2.1 Research License. Subject to the terms and conditions of this Agreement, ALEXION hereby grants to ZEALAND, during the Discovery/Development Program Term, a royalty-free, non-exclusive, worldwide license to use the ALEXION Background Intellectual Property solely for the purpose of conducting any Discovery/Development Activities expressly allocated to ZEALAND under this Agreement.
9.2.2 Research and Evaluation License. Subject to the terms and conditions of this Agreement, ZEALAND hereby grants to ALEXION and its Affiliates, during the Discovery/Development Program Term, a worldwide, royalty-free, co-exclusive (with ZEALAND) license, with the right to sublicense (subject to the provisions of Section 9.2.6 below), under the Licensed Intellectual Property solely for the purpose of evaluating of any Results, Deliverables, Candidate Products, or Products.
9.2.3 Manufacturing License. Subject to the terms and conditions of this Agreement, ALEXION hereby grants to ZEALAND and its Affiliates, a worldwide, royalty-free, non-exclusive license, without the right to sublicense (except to CMOs of ZEALAND as permitted under this Agreement or the applicable Clinical or Commercial Supply Agreement), under the ALEXION Background Intellectual Property, solely for the purpose of carrying out Manufacturing activities expressly allocated to ZEALAND pursuant to this Agreement, the Clinical Supply Agreement or the Commercial Supply Agreement.
9.2.4 Development and Commercialization License. Subject to the terms and conditions of this Agreement, ZEALAND hereby grants to ALEXION and its Affiliates a worldwide, royalty-bearing in accordance with Article 6 of this Agreement, exclusive (even as to ZEALAND, except to the extent necessary for ZEALAND to conduct any Discovery/Development Activities or Manufacturing activities expressly allocated to ZEALAND under this Agreement or the Clinical Supply Agreement or Commercial Supply Agreement) license, with the right to sublicense through multiple tiers (subject to the provisions of Section 9.2.6 below), under the Licensed Intellectual Property to Develop, have Developed, Manufacture, have Manufactured, use, have used, offer for sale, have offered for sale, sell, have sold, export, have exported, import, have imported, Commercialize and have Commercialized Products, or otherwise fully exploit Products in the Field in the Territory.
9.2.5 Collaboration IP License. Subject to the terms and conditions of this Agreement, ZEALAND hereby grants to ALEXION and its Affiliates a worldwide, royalty free, fully paid-up, non-exclusive license, with the right to sublicense through multiple tiers (subject to the provisions of Section 9.2.6 below), in and to any Collaboration IP to Develop, have Developed, Manufacture, have Manufactured, use, have used, offer for sale, have offered
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
for sale, sell, have sold, export, have exported, import, have imported, Commercialize and have Commercialized any and all products that, as its primary mechanism of action [***].
9.2.6 Sublicensing. ZEALAND may not grant any sublicense under Section 9.2.1 or Section 9.2.3 without the prior written consent of ALEXION, except to ZEALAND’s permitted subcontractors on APPENDIX 6 as approved by ALEXION, such approval not to be unreasonably withheld, conditioned or delayed. ALEXION and its Affiliates may grant sublicenses under Section 9.2.2 and Section 9.2.4 to any Affiliate or Third Party. A Party granting a sublicense under this Section 9.2.6 (the “Sublicensing Party”) shall promptly provide the other Party (the “Non-Sublicensing Party”) with a copy of each sublicense agreement within [***] days after execution of such sublicense agreement, which may be redacted to omit any terms not necessary to confirm the Sublicensing Party’s compliance with this Section 9.2.6. Any such sublicense agreement shall comply with the terms of this Agreement and shall include (a) confidentiality, non-disclosure and non-use provisions at least as restrictive or protective of the Parties as those set forth in this Agreement and (b) solely with respect to ALEXION as the Sublicensing Party, (i) an obligation to submit to ALEXION reports sufficient to enable ALEXION to comply with its obligations under Section 6.8.1, and (ii) audit requirements permitting audits by ALEXION of such Sublicensee consistent with the requirements set forth in Section 6.13.1 and (iii) indemnities that cover ZEALAND on terms substantially equivalent to those set out in Section 11.2. Each Party will remain responsible and liable for the performance of all Affiliates and Sublicensees under their respective sublicensed rights to the same extent as if such activities were conducted by the sublicensing Party. In no event will any sublicense relieve the sublicensing Party of any of its obligations under this Agreement.
9.2.7 [***]. By written notice delivered during the [***] Period applicable to any [***] Product, [***]. Subsequent to the delivery of such notice, [***]. If the Parties agree that such [***] Product is [***], the Parties shall [***]. Nothing in this Agreement or any course of conduct by either Party before or after the Effective Date of this Agreement, including during any [***] Period, shall give rise to any [***]. If [***], then [***] with respect to ZEALAND for such [***] Product under this Section 9.2.7.
9.2.8 No Implied Licenses. Except as expressly provided in this Agreement, neither Party shall be deemed to have granted the other Party any license or other right, title or interest, whether by implication, estoppel, reliance, or otherwise, with respect to any Intellectual Property of such Party.
9.3 Patent Prosecution and Maintenance.
9.3.1 Patent Prosecution by ALEXION. ALEXION shall have the first right (but not the obligation) to prosecute, defend and maintain all (i) Patent Rights that are [***] comprised in and to [***], including any [***] Patent Rights (collectively, “[***] Patent Rights”) and (ii) any [***] Patent Rights other than [***] Patent Rights (collectively, such [***] Patent Rights and other [***] Patent Rights “Agreement Patent Rights”) and ALEXION shall be responsible for and pay all future costs and expenses incurred for the
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
preparation, filing, prosecution, issuance and maintenance of such Agreement Patent Rights. ZEALAND shall provide ALEXION reasonable assistance and cooperation in respect thereto, including as specified in Section 9.3.4 below and including providing any necessary powers of attorney and executing any other required documents or instruments therefor. If ALEXION elects not to prosecute, defend or maintain any Agreement Patent Rights including electing not to file a patent application with respect thereto or to allow any such Patent Rights to lapse or become abandoned or unenforceable, then ALEXION shall promptly notify ZEALAND in writing (which such notice shall be at least [***] days prior to the lapse or abandonment of any such Patent Rights). Thereafter ZEALAND may, but is not required to, at its sole expense and in its sole discretion, prosecute, defend or maintain such Patent Rights through counsel of its choosing. In the event that ZEALAND undertakes such prosecution, defense or maintenance, ALEXION shall provide ZEALAND reasonable assistance and cooperation in respect thereto, including as specified in Section 9.3.4 below and including providing any necessary powers of attorney and executing any other required documents or instruments therefor.
9.3.2 Patent Prosecution by ZEALAND. ZEALAND shall have the first right (but not the obligation) to prosecute, defend and maintain all Patent Rights comprised in and to [***] that are not Agreement Patent Rights (collectively “ZEALAND Patent Rights”) and ZEALAND shall be responsible for and pay all future costs and expenses incurred for the preparation, filing, prosecution, issuance and maintenance of such ZEALAND Patent Rights. ALEXION shall provide ZEALAND reasonable assistance and cooperation in respect thereto, including as specified in Section 9.3.4 below and including providing any necessary powers of attorney and executing any other required documents or instruments therefor. If ZEALAND elects not to prosecute, defend or maintain any such ZEALAND Patent Rights including electing not to file a patent application with respect thereto or to allow any such Patent Rights to lapse or become abandoned or unenforceable, then ZEALAND shall promptly notify ALEXION in writing (which such notice shall be at least [***] days prior to the lapse or abandonment of any such Patent Rights). Thereafter ALEXION may, but is not required to, at its sole expense and in its sole discretion, prosecute, defend or maintain such ZEALAND Patent Rights through counsel of its choosing. In the event that ALEXION undertakes such prosecution, defense or maintenance, ZEALAND shall provide ALEXION reasonable assistance and cooperation in respect thereto, including as specified in Section 9.3.4 below and including providing any necessary powers of attorney and executing any other required documents or instruments therefor.
9.3.3 Comment Rights. In furtherance of Section 9.3.1 and 9.3.2, the prosecuting Party shall provide to the other Party or its designated patent counsel, reasonably in advance of each substantive submission to be filed in the United States, a Major European Market, Japan, China, or Canada, including any PCT application, a reasonable opportunity to review and comment on the proposed submission to any such patent office and consider in good faith all reasonable comments provided by such other the other Party, provided that the prosecuting Party shall have the final decision with respect thereto. The prosecuting Party will keep the other Party reasonably informed of the status of the applicable Patent Rights by timely providing the other Party with copies of filings or submissions relating to such
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Patent Rights and material communications relating to such Patent Rights that are received from any patent office.
9.3.4 Prosecution Cooperation. Each Party will cooperate with and reasonably assist the other Party in such preparation, filing, prosecution, issuance and maintenance, including making its relevant scientists and scientific records reasonably available. In addition, each Party will sign and deliver, or use reasonable efforts to have signed and delivered, at no charge to the requesting Party, all documents reasonably necessary for such preparation, filing, prosecution, issuance and maintenance of any Intellectual Property assigned to such requesting Party under this Agreement.
9.4 Patent Enforcement.
9.4.1 Third Party Infringement. During the Term, each Party shall promptly provide the other Party with a written notice if such Party becomes aware of any suspected, threatened or actual infringement by any Third Party of a [***] (in each case an “Infringed Patent”) and shall provide any available evidence of such suspected, threatened or actual infringement with such notification.
9.4.2 Infringement Actions.
(a) Enforcement. During the Term, ALEXION shall have the first right, but not the obligation, to enforce or defend any infringement proceedings, to the extent that the Infringed Patent is included in the [***] Patent Rights, against such Third Party, at ALEXION’s risk, cost and expense. During the Term, ZEALAND shall have the first right, but not the obligation, to enforce or defend any infringement proceedings in respect of any Patent Rights other than the [***] Patent Rights against such Third Party, at ZEALAND’s risk, cost and expense.
(b) Cooperation. Where a Party (the “Enforcing Party”) desires to enforce or defend such proceedings but may not – even as the co-owner or exclusive licensee of such Patent – do so due to Applicable Law or regulation, then the Enforcing Party may request that the other Party join as a named party in such action or itself enforce or defend such Infringed Patent against such Third Parties, at the Enforcing Party’s sole direction, cost and expense. The Enforcing Party shall take the lead in the control and conduct of any such enforcement or defense under this Section 9.4.2, in close coordination with the other Party, and the other Party shall reasonably assist the Enforcing Party at the Enforcing Party’s cost and expense, in any such enforcement or defense action under this Section 9.4.2.
(c) [***].
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
9.4.3 Third Party Patent Challenges. In the case of any objection, opposition or challenge by a Third Party to any [***] Patent Rights, ALEXION shall have the first right, but not the obligation, to defend such [***] Patent Rights against such Third Party, at ALEXION’s cost and expense, and the provisions of Section 9.4.2 shall apply as if such opposed, challenged or objected to [***] Patent Rights was an Infringed Patent. Objections, oppositions and challenges to a Patent Right under this Section include, for example, declaratory judgment proceedings, inter partes review proceedings, post grant review proceedings, patent interference proceedings, ex parte and inter partes reexamination proceedings, and patent opposition proceedings in a court, patent office or other administrative authority with competent jurisdiction in any country within the Territory.
9.4.4 Patent Listing. [***] shall [***] the listing of any [***] Patent Rights in the then-current edition of the United States Food and Drug Administration publication “Approved Drug Products with Therapeutic Equivalence Evaluations” (the “Orange Book”), or in equivalent patent listings in any other country within the Territory, in connection with the Regulatory Approval of any Product. If [***] desires to include in any such listing any such [***] Patent Rights, then [***] shall notify [***] and shall consider in good faith [***] reasonable comments with respect thereto, provided that [***] shall [***] with respect to any such listing. If [***] reasonably believes that a Patent Right other than any [***] Patent Rights should be included in any such listing, [***] shall consider in good faith [***] request with respect thereto.
9.4.5 Enforcement of Listed Patents. The provisions of Sections 9.4.2 and 9.4.3 notwithstanding, the following shall apply with respect to any notification provided by a Third Party to either ALEXION or ZEALAND under 21 U.S.C. § 355(j)(2)(B) making a certification described in 21 U.S.C. §355(j)(2)(A)(vii)(IV) with respect to any Agreement Patent Rights listed in the Orange Book for a Product and with respect to equivalent actions in the United States or in any other country within the Territory (a “Paragraph IV Certification”):
(a) The Party receiving a Paragraph IV Certification shall, without any avoidable delay and in any case within [***] Business Days after receiving such a Paragraph IV Certification, notify the other Party in writing and shall attach a copy of the Paragraph IV Certification to such notification.
(b) The rights to initiate or continue any infringement proceeding as a result of the Paragraph IV Certification with respect to a Product, including the commencement of a patent infringement action under 35 U.S.C. § 271(e)(2)(A), or under an equivalent statute or regulation within any other country in the Territory (a “Paragraph IV Proceeding”) shall be governed by Section 9.4.2, and any [***] Patent Rights (or other Patent Rights listed pursuant to Section 9.4.4) subject to a Paragraph IV Proceeding shall be considered an Infringed Patent under the [***] Patent Rights for purposes of interpreting Section 9.4.2.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
9.4.6 Recoveries. Any amount recovered in any action under Section 9.4.2, 9.4.3 or 9.4.5, including any amount recovered in any settlement of such action, shall first be used to [***] and shall thereafter be for [***] provided that such amount shall [***].
9.5 Defense of Claims. Each Party shall promptly inform the other in writing of any written notice to it of alleged infringement or misappropriation, based upon its performance of its obligations or exercise of its rights hereunder, of a Third Party’s Intellectual Property of which it shall become aware. Except as otherwise set forth herein (including under Article 11), each Party shall have the sole right to defend against any such claim brought against it, provided, however, that to the extent such action resulting from such claim involves an objection, opposition or challenge to any Agreement Patent Rights or ZEALAND Patent Rights by such Third Party or a counterclaim involving such Patent Rights, Section 9.4.2 or Section 9.4.3 shall apply, as applicable. Each Party shall each keep the other advised of all material developments in the conduct of any proceedings in defending any claim of such alleged infringement or misappropriation and shall reasonably cooperate with the other in the conduct of such defense. In no event may either Party settle any such infringement or misappropriation claim in a manner that would limit the rights of the other Party, admit the fault of the other Party or impose any obligation on the other Party, without such other Party’s prior written consent, which consent shall not be unreasonably withheld, conditioned or delayed.
9.6 Patent Term Extensions. [***] shall [***] all filings of requests for patent term extensions, supplementary protection certificates or equivalents thereto, in any country in the Territory, for any [***] and [***] shall have the rights to make such filing if [***] (hereinafter “Patent Term Extensions”). All costs and expenses relating to the Patent Term Extensions shall be born solely by [***]. Upon request of the filing Party and at the filing Party’s costs, the non-filing Party shall provide support, assistance and all necessary documents, in full executed form if needed, to the filing Party for the purpose of supporting, filing, obtaining and maintaining Patent Term Extensions.
10. PUBLICATIONS
10.1 Publication Rights. ALEXION shall [***] to publish on [***], subject to the prior review of ZEALAND. ALEXION shall submit to ZEALAND any such abstract, publication or presentation of [***] shall be subject to the prior review of ZEALAND and shall be provided by ALEXION at least [***] Business Days prior to its submission for publication or presentation. ZEALAND will use diligent efforts to complete its review at least [***] Business Days prior to ALEXION’s intended publication or presentation date. ALEXION shall, as reasonably requested by ZEALAND, (a) delete from such publication any of ZEALAND’s Confidential Information, or (b) upon a determination that such publication includes patentable material, delay the submission of such publication or presentation for an additional period of up to [***] Business Days in order to allow the appropriate Party to pursue patent protection.
10.2 Press Release. The Parties shall issue a press release following the execution of this Agreement describing the nature of the collaboration between ALEXION and ZEALAND in the form set out in Schedule 10.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
11. INDEMNIFICATION AND LIABILITY
11.1 Indemnification by ZEALAND. ZEALAND shall indemnify, defend, and hold harmless ALEXION and its Affiliates and their respective officers, directors, employees, licensees, sublicensees, and agents, and their respective successors, heirs and assigns and representatives (the “ALEXION Indemnitees”), from and against any and all damages, losses, suits, proceedings, liabilities, costs and expenses (including reasonable legal expenses, costs of litigation and reasonable attorney’s fees) or judgments, whether for money or equitable relief, of any kind (collectively, “Damages”) resulting from any and all suits, investigations, claims or demands of Third Parties (collectively, “Third Party Claims”) brought against an ALEXION Indemnitee, to the extent directly or indirectly arising out of or relating to: (i) the gross negligence or willful misconduct of ZEALAND, its Affiliates and its respective officers, directors, or employees in connection with ZEALAND’s performance of its obligations under this Agreement, (ii) any breach by ZEALAND of any obligation, representation, warranty or covenant set forth in this Agreement and (iii) the failure of ZEALAND or its Affiliates to comply with any Applicable Laws, in each case except to the extent ALEXION is liable for such Damages pursuant to Section 11.2.
11.2 Indemnification by ALEXION. ALEXION shall indemnify, defend, and hold harmless ZEALAND and its Affiliates, and its and their respective officers, directors, employees, and agents, and their respective successors, heirs and assigns and representatives (the “ZEALAND Indemnitees”), from and against any and all Damages resulting from Third Party Claims against a ZEALAND Indemnitee (including by ALEXION employees), to the extent directly or indirectly arising out of or relating to (i) the gross negligence or willful misconduct of ALEXION, its Affiliates, and its and their respective licensees, officers, directors, employees in connection with ALEXION’s performance of its obligations or exercise of its rights under this Agreement, (ii) any breach by ALEXION of any obligation, representation, warranty or covenant set forth in this Agreement, (iii) the failure to comply with any Applicable Laws by ALEXION, its Affiliates, or any of its licensees, or (iv) use, Development, Manufacture, Commercialization or other exploitation of any Candidate Product or Product by or on behalf of ALEXION or its Affiliates or Sublicensees, in each case except to the extent ZEALAND is liable for such Damages pursuant to Section 11.1.
11.3 Notification; Assumption of Defense; Cooperation and Assistance. In the event that a Party seeks indemnification hereunder with respect to a Third Party Claim, the Party seeking indemnification (the “Indemnified Party”) shall promptly notify the other Party (the “Indemnifying Party”) in writing (an “Indemnification Claim Notice”) of any Third Party Claim in respect of which it intends to claim indemnification under this Section 11.3 upon actual knowledge of any such claim or proceeding resulting in Damages, but in no event will the Indemnifying Party be liable for any Damages that result from any delay in providing such notice. The Indemnification Claim Notice must contain a description of the claim and the nature and amount of such Damages (to the extent that the nature and amount of such Damages is known at such time). The Indemnifying Party may, at its option, assume exclusive control of the defense and settlement of the Third Party Claim, subject to the limitations on settlement set forth below and in Section 9.5. If the Indemnifying Party assumes such defense, such assumption shall not be construed as an acknowledgement that the Indemnifying Party is liable to indemnify the Indemnified Party of any defenses it may assert against the Indemnified Party’s claim for indemnification. Following such assumption, the Indemnifying Party may appoint as lead counsel
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
in the defense of the Third Party Claim any legal counsel selected by the Indemnifying Party (the Indemnifying Party will consult with the Indemnified Party with respect to a possible conflict of interest of such counsel retained by the Indemnifying Party). The Indemnified Party will have the right to participate in the defense thereof and to employ counsel, at its own expense, separate from the counsel employed by the Indemnifying Party. If the Indemnifying Party does not commence actions to assume control of the defense of a Third Party Claim within [***] days after the receipt by the Indemnifying Party of the Indemnification Claim Notice required pursuant to this Section 11.3, or if shorter, prior to the date which is [***] Business Days before the expiry of any deadline to respond to a claim, the Indemnified Party will have the right to defend such claim in such manner as it may deem appropriate at the reasonable cost and expense of the Indemnifying Party. The Indemnified Party shall cooperate as may be reasonably requested by the Indemnifying Party (and at the Indemnifying Party’s expense) in order to ensure the proper and adequate defense of any action, claim or liability covered by this indemnification. The Indemnifying Party may not settle or otherwise dispose of any Third Party Claim without the prior written consent of the Indemnified Party unless such settlement includes only the payment of monetary damages (which are fully paid by the Indemnifying Party), does not impose any injunctive or equitable relief upon the Indemnified Party, does not require any admission or acknowledgment of liability or fault of the Indemnified Party and contains an unconditional release of the Indemnified Party in respect of such Third Party Claim. Regardless of whether the Indemnifying Party chooses to defend or prosecute any Third Party Claim, no Indemnified Party will admit any liability with respect to or settle or otherwise dispose of any Third Party Claim for which the Indemnifying Party may be liable for Damages under this Agreement without the prior written consent of the Indemnifying Party, such consent not to be unreasonably withheld, conditioned or delayed.
12. TERM AND TERMINATION
12.1 Term. This Agreement shall commence upon the Effective Date and, if not otherwise terminated earlier pursuant to this Article 12, shall continue in full force and effect on a Product-by-Product basis, until the expiration of the last payment obligation by ALEXION with respect to such Product (collectively, the “Term”).
12.2 Termination for Cause.
12.2.1 By ALEXION. In the event of a material breach of this Agreement by ZEALAND, which material breach remains uncured for [***] days measured from the date of written notice of such material breach by ALEXION as may be extended for the Mediation Period pursuant to Section 12.3, which notice identifies the material breach and the actions or conduct that it reasonably considers would be an acceptable cure of such material breach, ALEXION may terminate this Agreement in whole or with respect to one or more Candidate Products, Products or Targets, at any time during the Term of this Agreement by written notice of termination to ZEALAND.
12.2.2 By ZEALAND. In the event of a material breach of this Agreement by ALEXION, which material breach remains uncured for [***] days measured from the date of written notice of such material breach by ZEALAND as may be extended for the Mediation Period pursuant to Section 12.3, which notice identifies the material breach and the actions or conduct that it reasonably considers would be an acceptable cure of such material breach,
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
ZEALAND may terminate this Agreement in whole or with respect to one or more Candidate Products, Products or Targets to which such material breach relates, at any time during the Term of this Agreement by written notice of termination to ALEXION.
12.3 Disputes regarding Material Breach. In case the Party alleged to have committed the material breach (the “Defaulting Party”) disputes the material breach identified under Section 12.2.1 or 12.2.2 by the other Party (the “Non-Defaulting Party”), then the issue of whether the Non-Defaulting Party may properly terminate this Agreement on expiration of the Mediation Period in accordance with Section 13.8. This Agreement shall remain in full force and effect while any mediation pursuant to Section 13.8.2 is pending, such mediation shall not suspend any obligations of either Party hereunder, and each Party shall use reasonable efforts to mitigate any damage. If as a result of any dispute resolution proceeding (including litigation) it is determined that the Defaulting Party did not commit such material breach or such material breach was cured at any time during the Mediation Period in accordance with this Section 12.3, then no termination shall be effective, and this Agreement shall continue in full force and effect.
12.4 Termination for Insolvency. To the extent permitted by Applicable Laws, either Party may terminate this Agreement upon the filing or institution of bankruptcy, restructuring, reorganization, liquidation or receivership proceedings, upon the appointment of a receiver or trustee in respect of the other Party or over all or substantially all property, or upon an assignment of a substantial portion of the assets for the benefit of creditors by the other Party; provided, however, that in the case of any involuntary bankruptcy proceeding such right to terminate shall only become effective if (i) the other Party consents to the involuntary bankruptcy, (ii) a bankruptcy decree is rendered or (iii) the bankruptcy filing is not dismissed within [***] days after the filing thereof (an “Insolvency Event”).
12.4.1 All rights and licenses now or hereafter granted by ZEALAND to ALEXION under or pursuant to this Agreement are, for all purposes of Section 365(n) of the U.S. Bankruptcy Code, licenses of rights to “intellectual property” as defined in the U.S. Bankruptcy Code. Upon an Insolvency Event, ZEALAND agrees that ALEXION, as licensee of such rights under this Agreement, will retain and may fully exercise all of its rights and elections under the U.S. Bankruptcy Code. ZEALAND will, during the Term, create and maintain current copies or, if not amenable to copying, detailed descriptions or other appropriate embodiments, to the extent feasible, of all intellectual property licensed under this Agreement. Each Party acknowledges and agrees that “embodiments” of intellectual property within the meaning of Section 365(n) include laboratory notebooks, cell lines, product samples and inventory, research studies and data, all Regulatory Approvals (and all applications for Regulatory Approval) and rights of reference therein, the Licensed IP and Collaboration IP and all information related to the Licensed IP and Collaboration IP. If (i) a case under the U.S. Bankruptcy Code is commenced by or against ZEALAND, (ii) this Agreement is rejected as provided in section 365 of the U.S. Bankruptcy Code and (iii) ALEXION elects to retain its rights hereunder as provided in Section 365(n) of the U.S. Bankruptcy Code, ZEALAND (in any capacity, including debtor-in-possession) and its successors and assigns (including a trustee) will:
(a) provide ALEXION with embodiments of all Licensed IP and Collaboration IP held by ZEALAND and such successors and assigns,
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
or otherwise available to them, immediately upon ALEXION’s written request, and ALEXION will have the right to perform ZEALAND’s obligations hereunder and exercise all of the rights of a licensee of intellectual property under section 365(n) of the Bankruptcy Code, provided that neither such provision nor such performance by ALEXION will release ZEALAND from liability resulting from rejection of the license or the failure to perform such obligations; and
(b) not interfere with ALEXION’s rights under this Agreement, or any agreement supplemental hereto, to such intellectual property (including such embodiments), including any right to obtain such intellectual property (or such embodiments) from another entity, to the extent provided in Section 365(n) of the U.S. Bankruptcy Code.
12.4.2 All rights, powers and remedies of ALEXION provided herein are in addition to and not in substitution for any other rights, powers and remedies now or hereafter existing at law or in equity (including the U.S. Bankruptcy Code) in the event of the commencement of a case under the U.S. Bankruptcy Code with respect to ZEALAND. The Parties intend the following rights to extend to the maximum extent permitted by Applicable Law, and to be enforceable under U.S. Bankruptcy Code Section 365(n):
(a) the right of access to any intellectual property rights (including all embodiments thereof) of ZEALAND, or any Third Party with whom ZEALAND contracts to perform an obligation of ZEALAND under this Agreement to the extent that ZEALAND is not performing such obligation, and, in the case of any such Third Party, which is necessary for the Manufacture, use, sale, offer for sale, import or export of Products; and
(b) the right to contract directly with any Third Party to complete the contracted work to the extent that ZEALAND is not completing or having completed the contracted work.
12.5 [***]. [***] shall use all diligent efforts, as soon as practicable and not later than [***] months after the Effective Date, to [***]. Subject to the agreement of the Parties on [***], [***] shall [***], including [***]. The [***] shall be [a Danish limited liability company (in Danish: anpartsselskab) wholly owned by ZEALAND], and [***]. Concurrently with the [***], [***] shall negotiate the relevant [***], and the Parties will use their diligent efforts to finalise as soon as practicable the documentation for [***] (which the Parties intend [***] solely in connection with the [***]) within the foregoing [***] month period. The [***] documentation shall be in form and substance reasonably satisfactory to [***]. It is intended that, to the extent of [***], [***] will continue to perform this Agreement [***]. [***] acknowledges and agrees that a breach of its obligations under this Article 12 shall constitute a material breach of this Agreement, and any such breach would cause [***] irreparable harm and damage for which [***] would have no adequate remedy at law and for which monetary damages would be inadequate, and [***] further agrees
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
that in the event of a breach of such obligations [***] would be entitled to seek an injunction to compel [***] compliance with such obligations, in addition to any other remedies.
12.6 Termination at Will. ALEXION shall be entitled to terminate this Agreement, in whole or with respect to one or more Candidate Products, Products or Targets, at its sole discretion at any time upon [***] days prior written notice to ZEALAND.
12.7 Termination for Patent Challenge. In the event that ALEXION or any of its Affiliates or Sublicensees directly assert in its own respective name or directs or assists a Third Party to assert a Patent Challenge, ZEALAND will have the right to terminate this Agreement in its entirety upon [***] days written notice to ALEXION. For purposes of this Section 12.7, “Patent Challenge” means any challenges to a [***] Patent Right including, filing any opposition or objection with a patent office, declaratory judgment proceedings, inter partes review proceedings, post grant review proceedings, patent interference proceedings, ex parte and inter partes reexamination proceedings, and patent opposition proceedings in a court, patent office or other administrative authority with competent jurisdiction in any country within the Territory.
12.8 Termination for ZEALAND Change of Control. In the event of a Change of Control involving a Competitor, ALEXION may terminate this Agreement in whole or with respect to one or more Candidate Products, Products or Targets, within [***] days after receiving the Change of Control Notice, by sending written notice of such termination to ZEALAND within such [***] day period, with such termination being effective immediately prior to the closing of such Change of Control transaction; provided, however, that if ZEALAND [***], ALEXION shall [***].
12.9 Effects of Termination.
12.9.1 Generally. Upon termination (but not expiration) of the Agreement in whole or with respect to one or more Candidate Products, Products or Targets:
(a) The Receiving Party shall promptly return to the other Party or destroy all Confidential Information of the Disclosing Party that is solely related to any such terminated Candidate Product, Product or Target in accordance with Section 8.3 above;
(b) All licenses granted by ZEALAND to ALEXION under this Agreement with respect to any such terminated Candidate Product, Product or Target shall immediately terminate; and
(c) The licenses granted by ALEXION to ZEALAND pursuant to Section 9.2.1 and 9.2.3 with respect to any such terminated Candidate Product, Product or Target shall immediately terminate.
12.9.2 Return of Discontinued Product.
(a) Upon any Product becoming a Discontinued Product, any license rights granted by ZEALAND to ALEXION with respect to the Discontinued Product shall cease and revert to ZEALAND. ALEXION shall [***].
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
(b) ALEXION shall grant to ZEALAND an [***], and worldwide, which [***] shall [***]. Any [***] shall [***]. If permitted under [***], upon [***], such [***].
(c) Payments Regarding Discontinued Products. The milestones and royalty rates with respect to a Discontinued Product shall vary based on [***]:
|
[***] of the Product at |
|
Royalty Payments |
|
Less than [***] months after [***] applicable to such Discontinued Product |
|
[***] |
|
After the period above and prior to [***] |
|
[***] |
|
After [***] |
|
[***] |
12.10 Rights Accruing Prior to Expiration or Termination. Expiration or termination of this Agreement shall not relieve the Parties of any obligation accruing prior to such expiration or termination. Any expiration or termination of this Agreement shall be without prejudice to the rights of either Party against the other accrued or accruing under this Agreement prior to expiration or termination, including any payment obligation that accrued prior to the effective date of such expiration or termination.
12.11 Survival. In addition to any provisions specified in this Agreement as surviving as set forth therein, the provisions of Articles 1 (Definitions), 6 (Payments and Royalties) solely with respect to payment obligations accruing prior to the effective date of termination, 8 (Confidentiality, for the applicable time period set forth therein) and 11 (Indemnification and Liability, solely with respect to Third Party Claims arising out of circumstances occurring prior to the effective date of termination), Sections 4.6 (Other ALEXION Programs), 7.5 (Disclaimer of Warranties), 7.6 (No Consequential Damages), 9.1 (Ownership), 9.2.8 (No Implied Licenses), 9.3.4 (Prosecution Cooperation), 9.4.2(a) (Cooperation), Sections 12.4 (Termination for Insolvency) and 12.5 (Special Purpose Entity), 12.9 (Effects of Termination), 12.10 (Rights Accruing Prior to Expiration or Termination), this Section 12.11 (Survival) and Sections 13.1 (Assignment) through 13.13 (Further Assurances) shall survive any termination or expiration of the Agreement. In addition, all rights and obligations contained in this Agreement, which by their nature or effect contemplate performance or observance subsequent to expiration or termination of this Agreement will survive and remain binding upon and for the benefit of the Parties, their successors and permitted assigns.
13. CONCLUDING PROVISIONS
13.1 Assignment and Change of Control. Neither Party shall be entitled to assign or otherwise transfer its rights and/or obligations under this Agreement in whole or in part to any Third Party without the prior written consent of the other Party, which consent shall not be unreasonably withheld, conditioned or delayed; provided that, notwithstanding the foregoing, each Party may
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
assign this Agreement to (i) any Affiliate, or (ii) any successor corporation or entity resulting from any Change of Control of such Party; and provided further, that any such successor Affiliate, entity or Third Party to which this Agreement is assigned expressly agrees in writing to assume and be bound by all obligations of the assigning Party under this Agreement. Any Third Party involved in a Change of Control as set forth in the foregoing subclause (ii) of this Section 13.1 and such Third Party’s Affiliates immediately prior to such Change of Control shall be referred to herein as the “Acquirer”. Notwithstanding the foregoing, in the event that a Change of Control with respect to ZEALAND pursuant to the foregoing (ii) involves a Competitor, then ZEALAND shall promptly notify ALEXION of such contemplated Change of Control, but in any event within [***] Business Days prior to [***] with respect to such contemplated Change of Control, and ALEXION shall have the right to terminate this Agreement in accordance with Section 12.2.1. In the event of a Change of Control of ZEALAND, in addition to [***], ZEALAND shall implement effective walls and screens between personnel working on activities under this Agreement and activities performed on behalf of Third Parties.
13.2 Entire Agreement; Amendments. This Agreement sets forth the entire agreement between the Parties and supersedes all previous and contemporaneous negotiations, representations or agreements, written or oral, regarding the subject matter hereof. This Agreement may be amended only by an instrument in writing duly executed on behalf of the Parties. In case of inconsistencies between this Agreement and any Appendix hereof, the terms of this Agreement shall prevail unless agreed to explicitly that the Appendix should prevail.
13.3 Force Majeure. Neither Party shall be liable or deemed in default for failure to perform any duty or obligation that such Party may have under this Agreement where such failure has been occasioned by any act of God, fire, external strike, inevitable accidents, war, or any other cause outside the reasonable control of that Party, and occurring without its fault or negligence; provided that the Party affected will promptly notify the other of the force majeure condition and will exert all reasonable efforts to eliminate, cure or overcome any such causes and to resume performance of its obligations as soon as possible.
13.4 Waiver. The failure of either Party to require performance by the other Party of any of that other Party’s obligations under this Agreement shall in no manner affect the right of such Party to enforce the same at a later time. No waiver by any Party of any condition, or of the breach of any provision, term, representation or warranty contained in this Agreement shall be deemed to be or construed as a further or continuing waiver of any such condition or breach, or of any other condition or of the breach of any other provision, term, representation, or warranty hereof. The remedies provided in this Agreement are not exclusive and the Party suffering from a breach or default of this Agreement may pursue all other remedies, both legal and equitable, alternatively or cumulatively.
13.5 Severability. In the event that any provision or portion thereof in this Agreement is for any reason held to be invalid, illegal or unenforceable, the same shall not affect any other portion of this Agreement and its validity, as it is the intent of the Parties that this Agreement shall be construed in such fashion as to maintain its existence, validity and enforceability to the greatest extent possible. In any such event, this Agreement shall be construed as if such provision or portion thereof had never been contained in this Agreement, and there shall be deemed substituted therefore such provision as will most nearly carry out the intent of the Parties as expressed in this
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Agreement to the fullest extent permitted by Applicable Law unless doing so would have the effect of materially altering the right and obligations of the Parties in which event this Agreement may be terminated by mutual written agreement of the Parties.
13.6 Notices. Any notices and Invoices given under this Agreement shall be in writing and shall be addressed as follows (or at such other address for a Party as shall be specified by like notice, provided, however, that notices of a change of address shall be effective only upon receipt thereof):
If to ZEALAND:
Zealand Pharma A/S
Smedeland 36
2600 Glostrup Denmark
Attn: CEO
With copies (which is required but shall not constitute notice by themselves) to:
Zealand Pharma A/S
Smedeland 36
2600 Glostrup Denmark
Attn: General Counsel
and
Cooley (UK) LLP
Dashwood House, 69 Old Broad Street
London
EC2M 1QS
Attn: John Wilkinson
If to ALEXION:
ALEXION Pharma Holding Unlimited Company
22 Victoria Street
Hamilton HM 12 Bermuda
Attn: Secretary
With a copy to (which is required but shall not constitute notice by itself):
ALEXION Pharmaceuticals, Inc.
121 Seaport Blvd
Boston, MA 02210
Attn: General Counsel
13.7 Governing Law. This Agreement shall be construed in accordance with and governed exclusively by the law of [***], without reference to its rules of conflict of law.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
13.8 Dispute Resolution. Any dispute arising out of or in connection with this Agreement shall be settled, if possible, through good faith negotiations between the Parties. If the Parties are unable to settle such dispute within [***] days of first written notice by a Party of such dispute, such dispute shall:
13.8.1 be referred to the Chief Executive Officer of ZEALAND and the authorized designee of the [***] of ALEXION (the “Executive Officers”). Within [***] days of such notice, the Executive Officers of both Parties shall meet to attempt to resolve such dispute (the “Executive Meeting”). Such resolution, if any, of a referred issue shall be final and binding on the Parties. All negotiations pursuant to this Section 13.8 are confidential and shall be treated as compromise and settlement negotiations for purposes of applicable rules of evidence; and
13.8.2 if the Executive Officers cannot resolve such dispute within [***] days after either Party requests the Executive Meeting, then the Parties agree that the dispute shall be submitted to [***] for mediation under the [***]. Either party may commence mediation by providing to [***] and the other party a written request for mediation, setting forth the subject of the dispute and the relief requested. The parties will cooperate with [***] and with one another in selecting a mediator with relevant experience in the subject matter of the dispute. Appointment of appointment mediator is to be made within [***] days of the request for mediation, and the mediation is to take place within [***] days of such appointment. The mediation shall take place in [***] and both Parties shall make available sufficiently senior and suitably qualified and authorized representatives to resolve such dispute. The Mediation Period shall be the period from the Executive Meeting to [***] days after the application for the appoint of a mediator is made; and
13.8.3 if the dispute is not resolved within the Mediation Period, then either Party shall have the right to pursue any and all remedies available at law or equity.
13.9 Relationship of the Parties. Nothing in this Agreement is intended or will be deemed to constitute a partnership, agency, employer-employee or joint venture relationship between the Parties. No Party will incur any debts or make any commitments for the other. There are no express or implied third party beneficiaries hereunder (except for ALEXION Indemnitees and ZEALAND Indemnitees for purposes of Sections 11.1 or 11.2).
13.10 Headings. The captions to the Articles and Sections of this Agreement are not a part of this Agreement, but are merely for convenience to assist in locating and reading the several Sections of this Agreement.
13.11 Counterparts; Facsimiles. This Agreement may be executed in one or more counterparts, each of which will be deemed an original, and all of which together will be deemed to be one and the same instrument. Facsimile or PDF execution and delivery of this Agreement by either Party will constitute a legal, valid and binding execution and delivery of this Agreement by such Party.
13.12 Interpretation. Except where the context expressly requires otherwise, (a) the use of any gender herein shall be deemed to encompass references to either or both genders, and the use of the singular shall be deemed to include the plural (and vice versa), (b) the words and phrases e.g.,
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
“include”, “includes” and “including” shall be deemed to be followed by the phrase “without limitation”, (c) the word “will” shall be construed to have the same meaning and effect as the word “shall”, (d) any definition of or reference to any agreement, instrument or other document herein shall be construed as referring to such agreement, instrument or other document as from time to time amended, supplemented or otherwise modified (subject to any restrictions on such amendments, supplements or modifications set forth herein), (e) any reference herein to any person or entity shall be construed to include the person’s or entity’s successors and assigns, (f) the words “herein”, “hereof” and “hereunder”, and words of similar import, shall be construed to refer to this Agreement in its entirety and not to any particular provision hereof, (g) all references herein to Articles, Sections, Exhibits or Schedules shall be construed to refer to Articles, Sections, Exhibits or Schedules of this Agreement, and references to this Agreement include all Exhibits, Schedules and Appendixes hereto, (h) the word “notice” means notice in writing (whether or not specifically stated) and shall include notices, consents, approvals and other written communications contemplated under this Agreement, (i) provisions that require that a Party, the Parties or any committee hereunder “agree,” “consent” or “approve” or the like shall require that such agreement, consent or approval be specific and in writing, whether by written agreement, letter, approved minutes or otherwise (but excluding e-mail and instant messaging), (j) references to any specific law, rule or regulation, or article, section or other division thereof, shall be deemed to include the then-current amendments thereto or any replacement or successor law, rule or regulation thereof, (k) the term “or” shall be interpreted in the inclusive sense commonly associated with the term “and/or”, and (l) references to any Sections include Sections and subsections that are part of the related Section (e.g., a section numbered “Section 2.2” would be part of “Section 2”, and references to “Section 2.2” would also refer to material contained in the subsection described as “Section 2.2(a)”).
13.13 Further Assurances. Subject to the terms and conditions of this Agreement, each of ZEALAND and ALEXION agrees to duly execute and deliver, or cause to be duly executed or delivered, such further instruments and do and cause to be done such further acts, including the filing of additional assignments, agreements, documents and instruments, as the other Party may at any time and from time to time reasonably request in connection with this Agreement or to carry out more effectively the provisions and purposes of, or to better assure and confirm unto such other Party its rights and remedies under, this Agreement.
{Signature Page Follows}
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
IN WITNESS WHEREOF, the Parties have executed this Agreement in duplicate originals by their duly authorized representatives as of the date and year first above written
ALEXION Pharma Holding Unlimited Company
|
By: |
/s/ Patrick Riley |
|
|
|
|
|
|
Name: |
Patrick Riley |
|
|
|
|
|
|
Title: |
Senior Director of Research & Development, Director |
|
ZEALAND Pharma A/S
|
By: |
/s/ Martin Nicklasson |
|
|
|
|
|
|
Name: |
Martin Nicklasson |
|
|
|
|
|
|
Title: |
Chairman of the Board |
|
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Appendices:
|
APPENDIX 1: |
Discovery Plan and Development Plan for the Lead Target |
|
APPENDIX 2: |
Complement Pathway Targets |
|
APPENDIX 3: |
Discovery Plan and Development Plan(s) for any Selected Target(s), Additional Target(s), and Replacement Targets |
|
APPENDIX 4: |
Contact List |
|
APPENDIX 5: |
Licensed Patent Rights |
|
APPENDIX 6: |
Approved Subcontractors |
Schedules:
|
Schedule 1.63: |
Existing Lead Molecules |
|
Schedule 1.155: |
[***] Activities |
|
Schedule 5: |
Supply Agreement Key Terms |
|
Schedule 10: |
Press Release |
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
APPENDIX 1
Discovery Plan and Development Plan for the Lead Target
[***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
APPENDIX 2
Complement Pathway Targets
[***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
APPENDIX 3
Discovery Plan and Development Plan(s) for any Selected Target(s),
Additional Target(s), and Replacement Targets
[***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
APPENDIX 4
Contact List
[***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
APPENDIX 5
Licensed Patent Rights
[***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
APPENDIX 6
Approved Subcontractors
[***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
SCHEDULE 1.63
Existing Lead Molecules
[***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
SCHEDULE 1.155
[***] Activities
[***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
SCHEDULE 5
Supply Agreement Key Terms
Below are the terms that the Parties intend that the CMO Supply Agreement should contain, provided always that neither Party shall be in breach of this Agreement to the extent that a CMO will not agree such terms.
[***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
SCHEDULE 10
Press Release
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.

Company announcement — No. 06 / 2019
Alexion and Zealand Pharma Announce Collaboration to Discover and Develop Peptide Therapies for Complement-Mediated Diseases
· Collaboration furthers Alexion’s complement leadership with expansion into peptide therapies.
· Agreement provides Alexion with exclusive worldwide licenses for one preclinical target, with option for up to three additional targets, in the complement pathway.
· Zealand Pharma to receive upfront payment of $25 million and equity investment of $15 million, with potential for additional milestone-dependent and royalty payments.
· Collaboration further strengthens Zealand Pharma’s position as a leading developer of next generation peptide-based therapeutics.
BOSTON & COPENHAGEN, Denmark, March 20, 2019 - Alexion Pharmaceuticals, Inc. (NASDAQ: ALXN) and Zealand Pharma A/S (NASDAQ: ZEAL) today announced a collaboration to discover and develop novel peptide therapies for complement-mediated diseases. Peptides offer a number of advantages, including being highly selective and potent, allowing low dosage volumes for ease of administration, and having the potential to treat a broad range of complement-mediated diseases. The agreement provides Alexion with exclusive worldwide licenses, as well as development and commercial rights, for up to four targets within the complement pathway.
“We know that uncontrolled activation of the complement pathway is responsible for many devastating diseases, and through the development of the first two complement inhibitors — SOLIRIS and ULTOMIRIS — we have shown the transformative impact of C5 inhibition on several of these diseases,” said John Orloff, M.D., Executive Vice President and Head of Research & Development at Alexion. “We are excited by the potential to explore different targets in the complement pathway and look forward to building on Alexion’s more than 20 years of complement expertise with the development of next-generation peptide therapies in collaboration with Zealand Pharma, which may provide the opportunity to treat many additional diseases.”
“Collaborating with Alexion is further evidence of the strength of our peptide discovery platform, which has delivered both approved novel peptide-based therapeutics and a deep late-stage pipeline,” said Adam Steensberg, Interim CEO and Chief Medical and Development Officer at Zealand. “We believe that peptide-based therapies hold great promise in inhibiting complement activation. Alexion’s demonstrated expertise in treating rare and complement-mediated diseases makes this the ideal collaboration to advance Zealand’s complement-focused peptide programs.”
Under the terms of the agreement, Alexion and Zealand will enter into an exclusive collaboration for the discovery and development of subcutaneously delivered peptide therapies directed to up to four complement pathway targets. Zealand will lead the joint discovery and research efforts through the preclinical stage, and Alexion will lead development efforts beginning with IND filing and Phase 1 studies. The agreement provides Alexion with exclusive worldwide licenses and commercial rights to the peptide therapies developed in the collaboration. Zealand will receive an immediate upfront payment of $25 million for the first target, with Alexion making a concurrent $15 million equity investment in Zealand Pharma at a premium to the market price as of the collaboration effective date. For the lead target, the agreement provides the potential for development-related milestones of up to $115 million, as well as up to $495 million in sales-related milestones and the potential for high single- to low double-digit royalty payments. Each of the three subsequent targets can be selected for an option fee of $15 million and has the potential for additional development-milestones and sales-milestones and royalty payments at a reduced price to the lead target.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Alexion will discuss the collaboration further during today’s Investor Day event and webcast. Zealand will host a conference call on Thursday, March 21, at 4 p.m. CET.
About Alexion
Alexion is a global biopharmaceutical company focused on serving patients and families affected by rare diseases through the discovery, development and commercialization of life-changing therapies. As the global leader in complement biology and inhibition for more than 20 years, Alexion has developed and commercializes two approved complement inhibitors to treat patients with paroxysmal nocturnal hemoglobinuria (PNH) as well as the first and only approved complement inhibitor to treat atypical hemolytic uremic syndrome (aHUS) and anti-acetylcholine receptor (AchR) antibody-positive generalized myasthenia gravis (gMG), and is also developing it for patients with neuromyelitis optica spectrum disorder (NMOSD). Alexion also has two highly innovative enzyme replacement therapies for patients with life-threatening and ultra-rare metabolic disorders, hypophosphatasia (HPP) and lysosomal acid lipase deficiency (LAL-D). In addition, the company is developing several mid-to-late-stage therapies, including a second complement inhibitor, a copper-binding agent for Wilson disease and an anti-neonatal Fc receptor (FcRn) antibody for rare Immunoglobulin G (IgG)-mediated diseases as well as several early-stage therapies, including one for light chain (AL) amyloidosis and a second anti-FcRn therapy. Alexion focuses its research efforts on novel molecules and targets in the complement cascade and its development efforts on the core therapeutic areas of hematology, nephrology, neurology, and metabolic disorders. Alexion has been named to the Forbes list of the World’s Most Innovative Companies seven years in a row and is headquartered in Boston, Massachusetts’ Innovation District. The company also has offices around the globe and serves patients in more than 50 countries. This press release and further information about Alexion can be found at: www.alexion.com.
About Zealand Pharma
Zealand Pharma is a biotechnology company focused on the discovery and development of innovative peptide-based medicines. The company’s peptide discovery platform is built on 20 years of experience and has been extensively validated by their clinical pipeline, partnerships and marketed products; these include more than 10 drug candidates invented by Zealand that have advanced into clinical development, of which two have reached the market. Zealand’s current pipeline of internal product candidates focus on specialty gastrointestinal and metabolic diseases. Zealand’s portfolio also includes two clinical license collaborations with Boehringer Ingelheim. Zealand is based in Copenhagen (Glostrup), Denmark. For further information about the company’s business and activities, please visit www.zealandpharma.com.
Forward-Looking Statement
This press release includes forward-looking statements. Such forward-looking statements are subject to risks and uncertainties that could cause actual results to differ materially from those expressed or implied in such statements. Examples of forward-looking statements include, among others, statements we make regarding: (i) the therapeutic and commercial potential of peptide-based therapies; (ii) research and development plans related to peptide-based therapies; (iii) the potential of peptide-based therapies for the treatment of complement-mediated diseases; (iv) the potential for the collaboration between Alexion and Zealand; and (v) the closing of the equity investment by Alexion into Zealand, which is subject to standard closing conditions. The process by which early stage products such as Zealand’s peptide-based therapies could potentially lead to an approved product is long and subject to highly significant risks, particularly with respect to a preclinical research collaboration. Applicable risks and uncertainties include those relating to preclinical research and other risks identified under the heading “Risk Factors” included in Alexion’s and Zealand’s most recent Form 10-K and 20-F filings and in other future filings with the SEC. The forward-looking statements contained in this press release reflect Alexion’s and Zealand’s current views with respect to future events, and neither Alexion nor Zealand undertakes and specifically disclaims any obligation to update any forward-looking statements, except as required by law.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Alexion Contacts:
Media
Megan Goulart, 857-338-8634
Senior Director, Corporate Communications
Investors
Susan Altschuller, Ph.D., 857-338-8788
Vice President, Investor Relations
Zealand Pharma Contacts:
Adam Steensberg, +45 5060 3601
Interim CEO, Chief Medical and Development Officer
Mats Blom, +45 3153 7973
Chief Financial Officer
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
SHARE PURCHASE AGREEMENT
BETWEEN
EACH PERSON IDENTIFIED AS A “SELLER” IN SCHEDULE A
— and —
ZEALAND PHARMA A/S
— and —
ENCYCLE THERAPEUTICS INC.
OCTOBER 22, 2019
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
TABLE OF CONTENTS
|
|
|
|
Page |
|
|
|
|
|
|
ARTICLE 1 |
|
INTERPRETATION |
1 |
|
|
|
|
|
|
1.1 |
|
Definitions |
1 |
|
|
|
|
|
|
1.2 |
|
Certain Rules of Interpretation |
12 |
|
|
|
|
|
|
1.3 |
|
Governing Law |
13 |
|
|
|
|
|
|
1.4 |
|
Entire Agreement |
13 |
|
|
|
|
|
|
1.5 |
|
Schedules and Exhibits |
13 |
|
|
|
|
|
|
ARTICLE 2 |
|
PURCHASE AND SALE |
14 |
|
|
|
|
|
|
2.1 |
|
Agreement of Purchase and Sale |
14 |
|
|
|
|
|
|
2.2 |
|
Purchase Price |
14 |
|
|
|
|
|
|
2.3 |
|
Allocation of Payments |
19 |
|
|
|
|
|
|
2.4 |
|
Payment of Purchase Price |
19 |
|
|
|
|
|
|
2.5 |
|
Withholding Rights |
20 |
|
|
|
|
|
|
2.6 |
|
Capitalised Value |
20 |
|
|
|
|
|
|
ARTICLE 3 |
|
REPRESENTATIONS AND WARRANTIES |
20 |
|
|
|
|
|
|
3.1 |
|
Disclosure Schedule |
20 |
|
|
|
|
|
|
3.2 |
|
Representations Relating to the Sellers |
21 |
|
|
|
|
|
|
3.3 |
|
Representations Relating to the Corporation |
22 |
|
|
|
|
|
|
3.4 |
|
Representations Relating to the Buyer |
36 |
|
|
|
|
|
|
ARTICLE 4 |
|
COVENANTS |
37 |
|
|
|
|
|
|
4.1 |
|
Delivery of Books and Records |
37 |
|
|
|
|
|
|
4.2 |
|
Tax Returns |
37 |
|
|
|
|
|
|
4.3 |
|
Tax Refunds |
37 |
|
|
|
|
|
|
4.4 |
|
Amendments to Tax Returns |
37 |
|
|
|
|
|
|
ARTICLE 5 |
|
CLOSING ARRANGEMENTS |
38 |
|
|
|
|
|
|
5.1 |
|
Closing |
38 |
|
|
|
|
|
|
5.2 |
|
Closing Deliveries |
38 |
|
|
|
|
|
|
ARTICLE 6 |
|
INDEMNIFICATION AND SURVIVAL |
39 |
|
|
|
|
|
|
6.1 |
|
Indemnification by the Sellers |
39 |
|
|
|
|
|
|
6.2 |
|
Indemnification by the Buyer |
40 |
|
|
|
|
|
|
6.3 |
|
Survival Periods for Claims by Buyer Indemnified Parties |
40 |
|
|
|
|
|
|
6.4 |
|
Survival Periods for Claims by Seller Indemnified Parties |
41 |
|
|
|
|
|
|
6.5 |
|
Amount Limitations on Indemnification Obligations |
41 |
|
|
|
|
|
|
6.6 |
|
Rules Relating to Indemnification Obligations |
42 |
|
|
|
|
|
|
6.7 |
|
Notice of Indemnity Claims |
42 |
|
|
|
|
|
|
6.8 |
|
Procedure for Direct Claims |
43 |
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
TABLE OF CONTENTS
(continued)
|
|
|
|
Page |
|
|
|
|
|
|
6.9 |
|
Procedure for Third Party Claims |
43 |
|
|
|
|
|
|
6.10 |
|
Payment of Claims |
45 |
|
|
|
|
|
|
6.11 |
|
Indemnity Adjustments to Purchase Price |
45 |
|
|
|
|
|
|
6.12 |
|
Exclusive Remedy |
45 |
|
|
|
|
|
|
6.13 |
|
No Right of Subrogation |
46 |
|
|
|
|
|
|
6.14 |
|
Third Party Indemnification |
46 |
|
|
|
|
|
|
ARTICLE 7 |
|
GENERAL |
46 |
|
|
|
|
|
|
7.1 |
|
The Sellers’ Representative |
46 |
|
|
|
|
|
|
7.2 |
|
Time of Essence |
47 |
|
|
|
|
|
|
7.3 |
|
Notices |
47 |
|
|
|
|
|
|
7.4 |
|
Severability |
48 |
|
|
|
|
|
|
7.5 |
|
Submission to Jurisdiction |
49 |
|
|
|
|
|
|
7.6 |
|
Amendment and Waiver |
49 |
|
|
|
|
|
|
7.7 |
|
Further Assurances |
49 |
|
|
|
|
|
|
7.8 |
|
Assignment and Enurement |
49 |
|
|
|
|
|
|
7.9 |
|
Counterparts and Electronic Delivery |
49 |
|
|
|
|
|
|
7.10 |
|
Electronic Delivery |
50 |
|
|
|
|
|
|
7.11 |
|
Costs and Expenses |
50 |
|
|
|
|
|
|
7.12 |
|
Tender |
50 |
|
|
|
|
|
|
7.13 |
|
Payment and Currency |
50 |
|
|
|
|
|
|
7.14 |
|
Public Announcements |
51 |
|
|
|
|
|
|
7.15 |
|
Equitable Remedies |
51 |
|
|
|
|
|
|
7.16 |
|
No Contra Proferentem |
51 |
|
|
|
|
|
|
7.17 |
|
Independent Legal Advice |
51 |
|
|
|
|
|
|
7.18 |
|
Language |
51 |
|
|
|
|
|
|
7.19 |
|
Third Party Beneficiaries |
51 |
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
SHARE PURCHASE AGREEMENT
THIS AGREEMENT is dated as of October 22, 2019.
B E T W E E N :
Each Person identified as a “Seller” in Schedule A
(each, a “Seller” and, collectively, the “Sellers”)
- and -
ZEALAND PHARMA A/S, a company existing under the
laws of Denmark
(the “Buyer”)
- and -
ENCYCLE THERAPEUTICS INC., a corporation
incorporated under the laws of Ontario
(the “Corporation”)
CONTEXT:
A. The Sellers collectively own all of the issued and outstanding shares in the capital of the Corporation.
B. The Sellers want to sell to the Buyer and the Buyer wants to purchase from the Sellers all of the issued and outstanding shares in the capital of the Corporation.
THEREFORE, the Parties agree as follows:
ARTICLE 1
INTERPRETATION
1.1 Definitions
In this Agreement, in addition to terms defined elsewhere in this Agreement, the following terms have the following meanings:
1.1.1 “Accounting Principles” means the same accounting principles, methods, policies, practices and procedures, with consistent classifications, judgments and estimation methodology, as were used in the preparation of the Financial Statements.
1.1.2 “Affiliate” means an affiliate as that term is defined in the Business Corporations Act (Ontario).
1.1.3 “Agreement” means this agreement, including all Schedules and Exhibits, as it may be confirmed, amended, supplemented or restated by written agreement between the Parties.
1.1.4 “Arm’s Length” means arm’s length as that term is interpreted in connection with its use in the ITA.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
1.1.5 “Audited Balance Sheet” the audited balance sheet of the Corporation as at and for the year ended December 31, 2018.
1.1.6 “Books and Records” means all books, ledgers, files, lists, reports, plans, logs, deeds, surveys, correspondence, operating records, Tax Returns and other data and information, including all data and information stored on computer-related or other electronic media, of and related to the Corporation or the Business.
1.1.7 “Business” means the drug discovery business as is carried on by the Corporation including, without limitation, (a) the synthetic platform that has been developed by the Corporation enabling the generation of enhanced peptide macrocycles; and (b) the production of the Lead Product.
1.1.8 “Business Day” means any day excluding a Saturday, Sunday or statutory holiday in the Province of Ontario or the State of Denmark.
1.1.9 “Buyer” is defined in the recital of the Parties above.
1.1.10 “Buyer Indemnified Parties” means the Buyer, the Corporation, their respective Affiliates, and their respective directors, officers, shareholders, agents and employees, and the respective Successors of each of them.
1.1.11 “Benchmark Price Per Share” shall mean the volume weighted average closing price of Common Shares for the preceding ten (10) trading days on the Copenhagen Nasdaq Stock Exchange, converted to United States dollars using the spot foreign exchange rate published in the Wall Street Journal, European Edition on the day that such calculation is made.
1.1.12 “CFDA” means the Canada Food and Drugs Act and any rules and regulations promulgated thereunder.
1.1.13 “CFR” means the United Stated Code of Federal Regulations.
1.1.14 “Claim” means any claim, demand, action, cause of action, suit, arbitration, investigation, proceeding, complaint, grievance, charge, prosecution, assessment or reassessment, including any appeal or application for review.
1.1.15 “Claim Amount” is defined in Section 6.10.1.
1.1.16 “Claim Dispute Period” is defined in Section 6.8.1.
1.1.17 “Closing” means the completion of the sale to, and purchase by, the Buyer of the Purchased Shares under this Agreement.
1.1.18 “Closing Balance Sheet” means the unaudited trial balance of the Corporation as at and for the period ended as of the Closing Date.
1.1.19 “Closing Date” means the date of execution of this Agreement or any other date that the Parties may agree is the date upon which the Closing will take place.
1.1.20 “Closing Time” means 10:00 a.m. (Toronto time) on the Closing Date or any other time on the Closing Date as may be agreed by the Parties.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
1.1.21 “Commercial Sale” means a sale to an independent third party for value after a Product receives market authorization in a jurisdiction and for clarity does not include sampling or product sold under a special access program.
1.1.22 “Common Shares” means common shares of the Buyer.
1.1.23 “Communication” means any notice, demand, request, consent, approval or other communication which is required or permitted by this Agreement to be given or made by a Party.
1.1.24 “Composition of Matter Claim” means all patents and patent applications originating or derived from [***], including all provisional and non-provisional applications, continuations, continuations-in-part, divisions and renewals, and all patent rights granted thereon, reissues, re-examinations and extensions or restorations by existing or future extension or restoration mechanisms, including supplementary protection certificates and equivalents thereof anywhere in the world.
1.1.25 “Confidentiality Agreement” means the Confidential Disclosure Agreement between the Corporation and the Buyer dated as of May 30, 2019.
1.1.26 “Constating Documents” means the certificate and articles of incorporation of the Corporation dated January 24, 2012, the by-laws of the Corporation dated January 24, 2012 and the shareholders agreement between the Corporation and its shareholders made on January 24, 2012 and amended on July 14, 2014, March 17, 2015, August 21, 2015, January 29, 2016, November 24, 2016 and August, 2017.
1.1.27 “Contract” means any agreement, understanding, undertaking, commitment, licence or lease, whether written or oral.
1.1.28 “Corporation” is defined in the recital of the Parties above.
1.1.29 “Direct Claim” means an Indemnity Claim that does not arise as the result of a Third Party Claim.
1.1.30 “Disclosure Schedule” means the disclosure attached as Schedule B.
1.1.31 “Designated Paying Agent” means the paying agent designated from time to time by the Sellers’ Representative in writing.
1.1.32 “Earnout Payment” means any payment required to be made by the Buyer pursuant to Section 2.2.2.
1.1.33 “EMA” means the European Medicines Agency or any successor agency thereto.
1.1.34 “Employees” means all personnel and independent contractors employed, engaged or retained by the Corporation in connection with the Business, including any that are on medical or long-term disability leave, or other statutory or authorized leave or absence.
1.1.35 “Encumbrance” means any security interest, mortgage, charge, pledge, hypothec, lien, encumbrance, restriction, option, adverse claim or other encumbrance of any kind.
1.1.36 “Environment” means the ambient air, all layers of the atmosphere, all water including surface water and underground water, all land, all living organisms and the interacting natural
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
systems that include components of air, land, water, living organisms and organic and inorganic matter, and includes indoor spaces.
1.1.37 “Environmental Laws” means any Laws relating to the Environment and protection of the Environment, the regulation of chemical substances or products, health and safety including occupational health and safety, and the transportation of dangerous goods.
1.1.38 “ETA” means the Excise Tax Act (Canada).
1.1.39 “FDA” means the U.S. Food and Drug Administration or any successor agent thereto.
1.1.40 “FDCA” means the U.S. Federal Food, Drug and Cosmetic Act and any rules and regulations promulgated thereunder.
1.1.41 “Financial Statements” means:
1.1.41.1 the audited financial statements of the Corporation as at and for the year ended December 31, 2018;
1.1.41.2 the unaudited trial balance of the Corporation as at and for the period ended September 13, 2019; and
1.1.41.3 the Closing Balance Sheet.
1.1.42 “Former Employees” means all personnel and independent contractors who had been employed, engaged or retained by the Corporation in connection with the Business and who are no longer so employed, engaged or retained.
1.1.43 “Fundamental Representations” means the representations and warranties in Sections 3.2.1 (Corporate Existence of Seller), 3.2.2 (Capacity and Authority), 3.2.3 (Binding Obligation), 3.2.6 (Absence of Conflict), 3.3.1 (Capacity and Authority), 3.3.2 (Binding Obligation), 3.3.3 (Restrictive Covenants), 3.3.4 (Absence of Conflict), 3.3.8 (Corporate Existence of Corporation), 3.3.12 (No Purchase Rights), 3.3.18 (Absence of Undisclosed Liabilities) and 3.3.39 (Brokerage Fees).
1.1.44 “GAAP” means Canadian generally accepted accounting principles applicable to private enterprises under Part II of the CPA Canada Handbook of the Chartered Professional Accountants of Canada, applicable on the date of the relevant preparation or calculation or on the date of the relevant financial statements or records.
1.1.45 “Governmental Authority” means:
1.1.45.1 any federal, provincial, state, local, municipal, regional, territorial, aboriginal, or other government, governmental or public department, branch, ministry, or court, domestic or foreign, including any district, agency, commission, board, arbitration panel or authority and any subdivision of any of them exercising or entitled to exercise any administrative, executive, judicial, ministerial, prerogative, legislative, regulatory, or taxing authority or power of any nature; and
1.1.45.2 any quasi-governmental or private body exercising any regulatory, expropriation or taxing authority under or for the account of any of them, and any subdivision of any of them.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
1.1.46 “Hazardous Substance” means any substance, waste, liquid, gaseous or solid matter, fuel, micro-organism, sound, vibration, ray, heat, odour, radiation, energy vector, plasma, organic or inorganic matter which is or is deemed to be, alone or in any combination, hazardous, hazardous waste, solid or liquid waste, toxic, a pollutant, a deleterious substance, a contaminant or a source of pollution or contamination, regulated by any Environmental Laws.
1.1.47 “HPFB” means Health Canada’s Health Products and Food Branch.
1.1.48 “IND” means an investigational new drug application to the FDA of the United States pursuant to CFR title 21, Part 312, or other such national equivalent.
1.1.49 “Indebtedness” means, without duplication, all obligations as at immediately prior to the Closing Time in respect of:
1.1.49.1 borrowed money at the full amount payable, including with respect to deposits or advances of any kind;
1.1.49.2 indebtedness evidenced by notes, debentures or similar instruments;
1.1.49.3 leases required to be capitalized in accordance with GAAP;
1.1.49.4 the deferred purchase price of assets, services or securities;
1.1.49.5 interest rate swap, forward contract, foreign currency hedge or other hedging or similar arrangement;
1.1.49.6 all letters of credit or similar facilities (to the extent drawn);
1.1.49.7 seller expenses and change of control payments, both including all fees, expenses and other payments of the Corporation incurred, triggered by or otherwise payable in connection with the transactions contemplated by this Agreement and the consummation of the transactions contemplated hereby that are unpaid as of Closing, whether accrued or not;
1.1.49.8 any obligation in the foregoing Sections 1.1.49.1 to 1.1.49.7 guaranteed directly or indirectly;
1.1.49.9 royalty payments;
1.1.49.10 stock options;
1.1.49.11 promissory notes;
1.1.49.12 employee liabilities (contractual or discretionary) including taxes thereon;
1.1.49.13 all current liabilities in accordance with GAAP to the extent not captured as part of a working capital adjustment, if any;
1.1.49.14 management services payable;
1.1.49.15 vendor termination payments;
1.1.49.16 withholding tax on convertible debt;
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
1.1.49.17 Tax liabilities in respect of any period ending on the Closing Date including in respect or as a result of any debt forgiveness; and
1.1.49.18 interest, premium, fees, expenses, penalties (including prepayment and early termination penalties) and other amounts owing in respect of the line items in the foregoing Sections 1.1.49.1 to 1.1.49.17,
provided that Indebtedness shall not include up to US$50,000.00 owing to Wuxi AppTec (Hong Kong) Limited.
1.1.50 “Indemnified Party” means a Buyer Indemnified Party or a Seller Indemnified Party.
1.1.51 “Indemnifying Party” means the Party providing indemnification under any provision of Article 6.
1.1.52 “Indemnity Claim” is defined in Section 6.7.
1.1.53 “Indemnity Notice” is defined in Section 6.7.
1.1.54 “Insurance Policies” means the insurance policies maintained by the Corporation with respect to the Business.
1.1.55 “Intellectual Property” means all intellectual property rights of every kind and description throughout the world, including all U.S., Canadian and other foreign (i) patents, patent applications, invention disclosures, and all related continuations, continuations-in-part, divisionals, reissues, re-examinations, substitutions, and extensions thereof (“Patents”), (ii) trademarks, service marks, names, corporate names, trade names, domain names, logos, slogans, trade dress, design rights, and other similar designations of source or origin, together with the goodwill symbolized by any of the foregoing (“Trademarks”), (iii) copyrights and copyrightable subject matter (“Copyrights”), (iii) rights in computer programs (whether in source code, object code, or other form), algorithms, databases, compilations and data, technology supporting the foregoing, and all documentation, including user manuals and training materials, related to any of the foregoing (“Software”), (iv) trade secrets ideas, know- how, inventions, proprietary processes, data, formulae, models, and methodologies (“Trade Secrets”), (v) moral rights and rights of attribution and integrity and benefits waiver of moral rights, (vi) all rights in the foregoing and in other similar intangible assets, and (vii) all applications and registrations for the foregoing.
1.1.56 “Inventories” means all inventories of every kind owned by the Corporation and pertaining to the Business including raw materials and supplies.
1.1.57 “[***] Agreement” means the Collaborative Research Agreement made effective [***] between [***], [***] and the Corporation as the same may be amended from time to time.
1.1.58 “ITA” means the Income Tax Act (Canada).
1.1.59 “Key Personnel” means either or both of [***] and [***].
1.1.60 “Knowledge of the Corporation” means the knowledge that either of the Key Personnel either has, or would have obtained, after having made or caused to be made all reasonable inquiries necessary to obtain informed knowledge, including inquiries of the Corporation’s records.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
1.1.61 “Law” or “Laws” means all laws, statutes, codes, ordinances, decrees, rules, regulations, by-laws, statutory rules, principles of law, published policies and guidelines, judicial or arbitral or administrative or ministerial or departmental or regulatory judgments, orders, decisions, rulings or awards, including general principles of common and civil law, and the terms and conditions of any grant of approval, permission, authority or licence of any Governmental Authority.
1.1.62 “Lead Product” means the [***] developed by the Corporation to [***] and is known as [***], the chemical structure of which is described in Exhibit 1.1.62 and shall also include any back-up compounds listed in Exhibit 1.1.62 or that otherwise falls within the claims of [***].
1.1.63 “Loss” means any loss, liability, damage, cost, expense, charge, fine, penalty or assessment, including:
1.1.63.1 the reasonable costs and expenses of any action, suit, proceeding, demand, assessment, judgment, settlement or compromise;
1.1.63.2 all interest, fines and penalties; and
1.1.63.3 all reasonable professional fees and disbursements,
and including loss of value and the monetary value of lost opportunity.
1.1.64 “Marketing Approval” shall mean, with respect to the Lead Product, (a) any and all licenses, registrations, authorizations and approvals of the applicable Regulatory Authority, including NDAs or any foreign equivalent thereof, as applicable, and (b) if applicable, any and all pricing or reimbursement authorizations and approvals, in each case ((a) and (b)) that are necessary to manufacture, distribute, sell, obtain reimbursement for, market, use, store, and import such Product in such country.
1.1.65 “Material Adverse Effect” means an effect resulting from any change, event, occurrence or state of facts, either individually or in the aggregate, that:
1.1.65.1 is, or could reasonably be expected to be, material and adverse to the business, condition (financial or otherwise), properties, assets (tangible or intangible), liabilities (whether absolute, accrued, conditional, contingent or otherwise), capitalization, operations, prospects or results of operations of the Corporation; or
1.1.65.2 would, or would reasonably be expected to, materially impair or delay the consummation of the transactions contemplated by this Agreement,
except that:
1.1.65.3 any change, event, occurrence or state of facts relating to:
1.1.65.3.1. conditions affecting the pharmaceutical industry generally in jurisdictions in which the Corporation carries on business, including changes in commodity prices, generally accepted accounting principles, Laws or Taxes;
1.1.65.3.2. general economic conditions, or financial, credit, currency exchange, securities or commodities markets in general; or
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
1.1.65.3.3. war, armed hostilities or acts of terrorism,
will not result in a Material Adverse Effect unless it relates primarily to (or has the effect of relating primarily to) the Corporation, or adversely affects the Corporation disproportionately, compared to other businesses of similar size operating in the same industry as the Corporation.
1.1.66 “Material Contract” means a Contract to which the Corporation is a party or is bound that:
1.1.66.1 involves or may result in the payment of money or money’s worth by or to the Corporation in an amount in excess of $[***];
1.1.66.2 has an unexpired term of more than one year (including renewals);
1.1.66.3 cannot be terminated by the Corporation without penalty upon less than 60 days’ notice; or
1.1.66.4 the termination of which, or under which the loss of rights, would have a material and adverse effect on the business, condition (financial or otherwise), properties, assets (tangible or intangible), liabilities (whether absolute, accrued, conditional, contingent or otherwise), capitalization, operations, prospects or results of operations of the Corporation,
and in any event includes the Contracts set out in Exhibit 3.3.25.
1.1.67 “Milestone” is defined in Section 2.2.1.
1.1.68 “Milestone Payment” is defined in Section 2.2.1.
1.1.69 “Net Sales” means the gross amount invoiced for bona fide arms’ length sales of the Lead Product by or on behalf of the Buyer or its Affiliates or distributors to Third Parties, less the following deductions, determined in accordance with the Buyer’s standard accounting methods as generally and consistently applied by the Buyer at the time that such sales are recorded:
1.1.69.1 normal and customary trade, cash and/or quantity discounts allowed and taken directly with respect to sales of the Lead Product;
1.1.69.2 amounts repaid or credited by reason of defects, rejections, recalls, returns, rebates and allowances in respect of the Lead Product;
1.1.69.3 chargebacks and other amounts paid on sale or dispensing of the Lead Product;
1.1.69.4 Third Party cash rebates and chargebacks related to sales of the Lead Product, to the extent allowed;
1.1.69.5 retroactive price reductions that are actually allowed or granted in respect of the Lead Product;
1.1.69.6 compulsory payments and rebates directly related to the sale of the Lead Product, accrued, paid or deducted pursuant to agreements (including, but not limited to, managed care agreements) or governmental regulations;
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
1.1.69.7 freight, insurance and other transportation charges, to the extent included in the invoice price of the Lead Product;
1.1.69.8 tariffs, duties, excise, sales, value-added, consumption or other taxes (other than taxes based on income), to the extent included in the invoice price of the Lead Product;
1.1.69.9 amounts paid or payable to, or withheld by, any distributor of the Lead Product used or engaged by the Buyer or its Affiliates in connection with the sale of the Lead Product, and
1.1.69.10 any other specifically identifiable costs or charges included in the gross invoiced sales price of the Lead Product falling within categories substantially equivalent to those listed above.
1.1.70 “NDA” means a new drug application as described in §505(b) of the FDCA and subject to CFR Title 21, Part 314, including any amendments submitted pursuant to CFR Title 21, §314.60, or any analogous application or submission with any Regulatory Authority to obtain authorization to market a pharmaceutical product outside of the United States.
1.1.71 “NRC Agreement” means the agreement between the Corporation and the National Research Counsel of Canada dated effective October 3, 2014.
1.1.72 “Parties” means the Sellers, the Corporation and the Buyer, collectively, and “Party” means any one of them.
1.1.73 “Permits” means the authorizations, registrations, permits, certificates of approval, approvals, grants, licences, quotas, consents, commitments, rights or privileges (other than those relating to the Intellectual Property) issued or granted by any Governmental Authority to the Corporation.
1.1.74 “Permitted Encumbrances” means:
1.1.74.1 unregistered liens for Taxes, assessments or similar charges incurred by the Corporation in the ordinary course of the Business that are not yet due and payable or, if due and payable, are to be adjusted between the Sellers and the Buyer on Closing; and
1.1.74.2 inchoate mechanic’s, construction and carrier’s liens and other similar liens arising by operation of law or statute in the ordinary course of the Business for obligations which are not delinquent and will be paid or discharged in the ordinary course of the Business.
1.1.75 “Person” will be broadly interpreted and includes:
1.1.75.1 a natural person, whether acting in his or her own capacity, or in his or her capacity as executor, administrator, estate trustee, trustee or personal or legal representative, and the heirs, executors, administrators, estate trustees, trustees or other personal or legal representatives of a natural person;
1.1.75.2 a corporation or a company of any kind, a partnership of any kind, a sole proprietorship, a trust, a joint venture, an association, an unincorporated association, an unincorporated syndicate, an unincorporated organization or any other association, organization or entity of any kind; and
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
1.1.75.3 a Governmental Authority.
1.1.76 “Personal Information” means information about an individual who can be identified by the Person who holds that information.
1.1.77 “PIPEDA” means the Personal Information Protection and Electronic Documents Act (Canada).
1.1.78 “Phase 3 Clinical Trial” means a human clinical trial of a product the design of which is acknowledged by the FDA to be sufficient for such clinical trial to satisfy the requirements of 21 CFR § 312.21(c) or, if conducted in a country outside the United States, a similar clinical trial prescribed by the applicable Regulatory Authority in such country which is acknowledged by such Regulatory Authority to be sufficient for such clinical trial to satisfy the requirements of a pivotal efficacy and safety clinical trial.
1.1.79 “Plans” means all employee benefit plans, programs, agreements or arrangements, domestic or foreign, including all bonus, incentive, profit sharing, pension, retirement compensation, retirement savings, retirement income, deferred compensation, incentive compensation, welfare, fringe benefit, vacation, salary continuation, legal, health and other medical, dental, life, accident, disability, supplemental retirement, nonqualified trusts, profit sharing, stock purchase, stock option, restricted stock, phantom stock, stock appreciation rights or other equity incentive plans, programs, agreements or arrangements, and all termination, severance or other plans, programs, agreements or arrangements, whether formal or informal, written or unwritten, funded or unfunded, registered or unregistered, insured or self-insured, whether covering one person or more than one person, and all other benefit plans, programs, agreements or arrangements which are maintained, contributed to, required to be contributed to, or sponsored by the Corporation, or under which the Corporation has any liability or contingent liability, for the benefit of, or relating to, any Employee or Former Employee or their respective dependants or beneficiaries or which otherwise provide coverage for any current or Former Employees or their respective dependants and beneficiaries.
1.1.80 “PMDA” means the Pharmaceuticals and Medical Devices Agency of Japan and any successor thereto.
1.1.81 “Privacy Laws” means any Laws that regulate the collection, use or disclosure of Personal Information.
1.1.82 “Pro Rata Share” means, with respect to any Seller, the percentage set out in Schedule A opposite that Seller’s name under the heading “Pro Rata Share of Seller”.
1.1.83 “Purchase Price” is defined in Section 2.2.1.
1.1.84 “Purchase Price Instalment” has the meaning set forth in Section 2.4.2.
1.1.85 “Purchased Shares” means all of the issued and outstanding shares in the capital of the Corporation.
1.1.86 “Quarterly Earnout Payment” means a payment determined in accordance with Section
1.1.86.1.1. as may be adjusted pursuant to Section 2.2.2.2.7.
1.1.87 “Regulatory Authority” means (i) any Governmental Authority, notified body or other organization in a country or region that regulates the manufacture or sale of pharmaceutical
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
or medicinal products or medical devices, including the FDA, the PMDA and the EMA, and any successors thereto, and (ii) any other relevant bodies authorized by Applicable Law to review or otherwise exercise oversight over MAAs, other regulatory filings or Regulatory Approvals
1.1.88 “Release” means to release, spill, leak, pump, pour, emit, empty, discharge, deposit, inject, leach, dispose, dump or permit to escape.
1.1.89 “Remedial Order” means any remedial order, including any notice of non-compliance, order, other complaint, direction or sanction issued, filed or imposed by any Governmental Authority under Environmental Laws, with respect to any failure or neglect to comply with Environmental Laws.
1.1.90 “Representatives” means the advisors, agents, consultants, directors, officers, management, employees, subcontractors, and other representatives, including accountants, auditors, financial advisors, lenders and lawyers of a Person.
1.1.91 “Second Milestone” is defined in Section 2.2.1.2.
1.1.92 “Second Milestone Payment” is defined in Section 2.2.1.2.
1.1.93 “Securities” has the meaning given to that term in the Securities Act (Ontario).
1.1.94 “Seller” and “Sellers” are defined in the recital of the Parties above.
1.1.95 “Seller Indemnified Parties” means each Seller and, as applicable, its Affiliates, and their respective directors, officers, shareholders, agents and employees, and the respective Successors of each of them.
1.1.96 “Seller Indemnifying Party” means any Seller providing indemnification under any provision of Article 6.
1.1.97 “Sellers’ Representative” is defined in Section 7.1.
1.1.98 “Straddle Period” means any taxation period of the Corporation ending after the Closing Date which commenced before the Closing Date and includes a period before the Closing Date.
1.1.99 “Stub Period Returns” is defined in Section 4.2.
1.1.100 “Successful Read-Out of Phase 2 Clinical Trial” means the earlier of (i) the first dose administered to a patient in a second Phase 2 Clinical Trial for the same compound as was administered in the first Phase 2 Clinical Trial, and (ii) the receipt of written final minutes from an end of phase 2 meeting with a Regulatory Authority confirming that safety and efficacy data for the Lead Product are sufficient to initiate a Phase 3 Clinical Trial.
1.1.101 “Successful Read-Out of Phase 3 Clinical Trial” means shall mean the first to occur of (i) a press release issued by the Buyer stating that the results of a completed Phase 3 Clinical Trial combined with other completed clinical and non-clinical studies are sufficient for the Buyer to submit an NDA for the Lead Product, and (ii) receipt of written confirmation of acceptance for filing of an NDA by a Regulatory Authority for evaluation of the Lead Product.
1.1.102 “Successors” means, as applicable, the heirs, executors, administrators, estate trustees, trustees, personal or legal representatives, successors and permitted assigns of a Person.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
1.1.103 “Tax” or “Taxes” means all taxes, duties, fees, premiums, assessments, imposts, levies, rates, withholdings, dues, government contributions and other charges of any kind imposed by any Governmental Authority, whether direct or indirect, together with all interest, penalties, fines, additions to tax or other additional amounts imposed in respect thereof, including those levied on, or measured by, or referred to as income, gross income, gross receipts, net proceeds, profits, capital gains, alternative or add-on, or minimum, capital, transfer, land transfer, sales, retail sales, consumption, use, goods and services, harmonized sales, value- added, ad valorem, turnover, excise, stamp, non-resident withholding, business, franchising, business licences, real and personal property (tangible and intangible), environmental, payroll, employee withholding, employment, health, employer health, social services, development, occupation, education or social security, and all contributions, premiums, surtaxes, all customs duties, countervail, anti-dumping, special import measures and import and export taxes, all licence, franchise and registration fees, all provincial workers’ compensation payments, and all employment insurance, health insurance and Canada, Québec and other government pension plan contributions.
1.1.104 “Tax Law” means any Law that imposes Taxes or that deals with the administration or enforcement of liabilities for Taxes.
1.1.105 “Tax Return” means any return, report, declaration, designation, election, undertaking, waiver, notice, filing, information return, statement, form, certificate or any other document or materials relating to Taxes, including any related or supporting information with respect to any of those documents or materials listed above in this Section 1.1.105, filed or to be filed with any Governmental Authority in connection with the determination, assessment, collection or administration of Taxes.
1.1.106 “Technology” shall mean all know-how, patents, patent applications and other Intellectual Property owned or controlled by Encycle as of the Effective Date.
1.1.107 “Third Party” shall mean any Person other than the Buyer or any Affiliate of the Buyer.
1.1.108 “Third Party Claim” means a Claim made against an Indemnified Party by a Person who is not a Party.
1.1.109 “TPD” means the Therapeutic Products Directorate of Health Canada.
1.1.110 “[***] Agreement” means the Technology License Agreement dated as of [***] between [***] and the Corporation (as amended by a [***] Agreement dated [***]).
1.2 Certain Rules of Interpretation
1.2.1 Gender, etc. In this Agreement, words signifying the singular number include the plural and vice versa, and words signifying gender include all genders.
1.2.2 Including. Every use of the words “including” or “includes” in this Agreement is to be construed as meaning “including, without limitation” or “includes, without limitation”, respectively.
1.2.3 Division and Headings. The division of this Agreement into Articles and Sections, the insertion of headings and the inclusion of a table of contents are for convenience of reference only and do not affect the construction or interpretation of this Agreement.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
1.2.4 Articles, Sections, etc. References in this Agreement to an Article, Section, Schedule or Exhibit are to be construed as references to an Article, Section, Schedule or Exhibit of or to this Agreement unless otherwise specified.
1.2.5 Time Periods. Unless otherwise specified in this Agreement, time periods within which or following which any calculation or payment is to be made, or action is to be taken, will be calculated by excluding the day on which the period begins and including the day on which the period ends. If the last day of a time period is not a Business Day, the time period will end on the next Business Day.
1.2.6 Statutory Instruments. Unless otherwise specified, any reference in this Agreement to any statute includes all regulations and subordinate legislation made under or in connection with that statute at any time, and is to be construed as a reference to that statute as amended, restated, supplemented, extended, re-enacted, replaced or superseded at any time.
1.3 Governing Law
This Agreement is governed by, and is to be construed and interpreted in accordance with, the Laws of the Province of Ontario and the Laws of Canada applicable therein without giving effect to any choice or conflict of law provision or rule (whether of the Province of Ontario or any other jurisdiction).
1.4 Entire Agreement
This Agreement, and any other agreements and documents to be delivered under this Agreement, constitutes the entire agreement between the Parties pertaining to the subject matter of this Agreement and supersedes all prior agreements, understandings, negotiations and discussions, whether oral or written, of the Parties, other than the provisions of the Confidentiality Agreement, and there are no representations, warranties or other agreements between the Parties in connection with the subject matter of this Agreement except as specifically set out in this Agreement, or in any other agreements and documents delivered under this Agreement. No Party has been induced to enter into this Agreement in reliance on, and there will be no liability assessed, either in tort or contract, with respect to, any warranty, representation, opinion, advice or assertion of fact, except to the extent it has been reduced to writing and included as a term in this Agreement, or in any other agreements and documents delivered under this Agreement.
1.5 Schedules and Exhibits
The following is a list of Schedules and Exhibits:
Schedule Subject Matter
A Sellers and Purchased Shares
B Disclosure Schedule
|
Exhibit |
|
Subject Matter |
|
|
|
|
|
1.1.62 |
|
Lead Product |
|
|
|
|
|
5.2.1.3 |
|
Form of Consulting and Transitional Services Agreement |
|
|
|
|
|
5.2.1.5 |
|
Form of Release |
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
ARTICLE 2
PURCHASE AND SALE
2.1 Agreement of Purchase and Sale
Subject to the terms and conditions of this Agreement, on the Closing Date the Sellers will sell, and the Buyer will purchase, the Purchased Shares.
2.2 Purchase Price
2.2.1 The aggregate purchase price (the “Purchase Price”) payable by the Buyer to the Sellers in accordance with Section 2.4 is the sum of the Quarterly Earnout Payments provided for in Section 2.2.2, and the following one-time payments (each, a “Milestone Payment”) upon achievement of the following milestones (each, a “Milestone”):
2.2.1.1 an amount equal to US$[***] upon the Buyer or any of its Affiliates, or a Third Party on behalf of and as directed by the Buyer or any of its Affiliates, first administering a dose of the Lead Product to an animal in a preclinical toxicity study (the “First Milestone”);
2.2.1.2 an amount equal to US$[***] (the “Second Milestone Payment”) upon the acceptance by the FDA of an IND (or other such national equivalent) in respect of the Lead Product (the “Second Milestone”);
2.2.1.3 an amount equal to US$[***] upon a Successful Read-Out of Phase 2 Clinical Trial in respect of the Lead Product;
2.2.1.4 an amount equal to US$[***] upon a Successful Read-Out of Phase 3 Clinical Trial in respect of the Lead Product;
2.2.1.5 an amount equal to US$[***] upon the first Commercial Sale in [***] of the Lead Product;
2.2.1.6 an amount equal to US$[***] upon the first Commercial Sale in either [***] or [***] of the Lead Product; and
2.2.1.7 an amount equal to US$[***] the first time that annual global Net Sales total at least US$[***] in a single calendar year.
For the avoidance of doubt, each Milestone may only be achieved one time and each Milestone Payment may only be paid one time, if at all.
2.2.2 Earnout.
2.2.2.1 The Buyer shall pay, or cause to be paid, to the Sellers:
2.2.2.1.1. in respect of sales of the Lead Product in any jurisdiction in which the Corporation has an issued patent having at least one Composition of Matter Claim that effectively and legally prevents a generic or interchangeable product entry (or competition) in the respective jurisdiction, an amount equal to [***]% of the [***] Net Sales in such jurisdiction commencing on the date of the first Commercial Sale of the Lead Product in such jurisdiction, and ending on the later of (i) the 10th anniversary of the first Commercial
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Sale of the Lead Product in such jurisdiction and (ii) the expiration of the issued patent of the Lead Product in such jurisdiction; and
2.2.2.1.2. in respect of sales of the Lead Product in any jurisdiction in which the Corporation does not have an issued patent having at least one Composition of Matter Claim that effectively and legally prevents a generic or interchangeable product entry (or competition) in the respective jurisdiction, an amount equal to [***]% of the [***] Net Sales in such jurisdiction during a period commencing on the date of the first Commercial Sale of the Lead Product in such jurisdiction, and ending on the tenth anniversary of the first Commercial Sale of the Lead Product in such jurisdiction.
2.2.2.2
2.2.2.2.1. The Buyer shall, within 90 days following the end of each quarter of the financial year of the Buyer, provide a report (each an “Quarterly Earnout Report”) to the Sellers’ Representative detailing the Buyer’s calculation of such payment as at the relevant quarter end and pay the amount of the Quarterly Earnout Payment set forth in the Quarterly Earnout Report to the Designated Payment Agent on behalf of the Sellers in accordance with Section 2.4.2.2.
2.2.2.2.2. The Buyer shall provide the Sellers’ Representative with reasonable access to the applicable records of the Buyer and its Affiliates to the extent reasonably requested by the Sellers’ Representative during the dispute resolution process described in this Section 2.2.2.2 and to the extent necessary for the Sellers’ Representative to review the applicable calculations.
2.2.2.2.3. If the Sellers’ Representative objects to any Quarterly Earnout Report, the Sellers’ Representative shall notify the Buyer in writing of such objection(s) within 120 days of the end of the relevant financial year of the Buyer (a “Notice of Objection”). The Notice of Objection shall describe, in reasonable detail, the basis for such dispute and shall include all disputes related to any Quarterly Earnout Report provided by the Buyer in the relevant financial year.
2.2.2.2.4. If the Sellers’ Representative does not deliver a Notice of Objection within the time limited therefor, then each Quarterly Earnout Report provided by the Buyer in the relevant financial year of the Buyer will be conclusive, final and binding in its entirety on all of the parties.
2.2.2.2.5. If the Sellers’ Representative does deliver a Notice of Objection in accordance with Section 2.2.2.2.3, then the Sellers’ Representative and the Buyer shall attempt to resolve such disputed items, and to the extent that the Sellers’ Representative and the Buyer are unable to resolve any such disputes, the items set forth in the Notice of Objection shall be resolved in accordance with the procedures set forth in Section 2.2.2.2.6 below.
2.2.2.2.6. In the event that the Sellers’ Representative and the Buyer are unable to resolve the disputed items within 20 Business Days after delivery of the Notice of Objection, either the Buyer or the Sellers’
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Representative may demand that such disputed items be referred to an accounting firm mutually agreed by the Sellers’ Representative and the Buyer (the “Accounting Firm”) to finally resolve such disputed items. The Accounting Firm shall act as an expert and not as an arbitrator to determine only the disputed items, and the determination of each disputed item shall be within the range established by the relevant Quarterly Earnout Report or Quarterly Earnout Reports and the Notice of Objection, if any. The determination of the Accounting Firm shall be made as promptly as possible and shall be final and binding upon the parties. Each party hereto shall be permitted to submit such data and information to the Accounting Firm as such party deems appropriate. The fees and expenses of the Accounting Firm incurred in resolving the disputed matter shall be borne by each of the Buyer, on the one hand, and the Sellers, on the other hand, in proportion to the difference between their respective position(s) on the matter(s) under dispute and the ultimate determined amount. For all other costs and expenses, the Sellers’ Representative and the Buyer shall each pay their own costs and expenses incurred under this Section 2.2.2.2.
2.2.2.2.7. Once the Accounting Firm has determined the disputed items, the amount that the Accounting Firm determines in respect of each disputed Quarterly Earnout Report shall be the deemed to be the Quarterly Earnout Amount payable to the Sellers. If the relevant Quarterly Earnout Amount determined by the Accounting Firm is greater than the amount paid by the Buyer pursuant to Section 2.2.2.2.1 in respect of the relevant Quarterly Earnout Report, the Buyer shall, within 10 Business Days of receipt of the Accounting Firm’s determination, pay to the Designated Paying Agent on behalf of the Sellers, in accordance with Section 2.4.2.2, an amount equal to the difference between the amount actually paid in respect of the relevant Quarterly Earnout Report and the relevant Quarterly Earnout Amount as determined by the Accounting Firm. If the relevant Quarterly Earnout Amount determined by the Accounting Firm is less than the amount paid by the Buyer pursuant to Section 2.2.2.2.1 in respect of the relevant Quarterly Earnout Report, the Buyer shall, without limiting its other remedies, be entitled to set-off such amount from any subsequent payments due by the Buyer on account of the Purchase Price.
2.2.3 The payments to the Sellers pursuant to Section 2.2.1 and Section 2.2.2 shall, notwithstanding the provisions thereof, be net of and reduced by, and the Purchase Price shall be reduced by, any amounts paid or payable by the Corporation or any of its Affiliates pursuant to the [***] Agreement, the [***] Agreement and, if applicable, the [***] Agreement by any amounts paid or payable by the Corporation or any of its Affiliates to any other Third Party in connection with such Third Party’s rights or entitlements (including Intellectual Property rights) in or to the Lead Product, where the existence of such rights or entitlement constitute a breach by the Corporation of the Corporation’s representations and warranties in this Agreement, or where the Buyer is entitled to indemnification from the Sellers as a result of the existence of such Third Party’s rights or entitlement.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
2.2.4 Commercially Reasonable Efforts.
2.2.4.1 Subject to Section 2.2.4.2, the Buyer shall, and shall cause its Affiliates to, (i) act in good faith and use Commercially Reasonable Efforts to achieve the First Milestone and the Second Milestone, where “Commercially Reasonable Efforts” means such commercially reasonable efforts as are typically used by a similarly situated pharmaceutical company in comparable circumstances for the development and commercialization of prescription pharmaceutical products of similar commercial potential at a similar stage in product lifecycle, taking into consideration the safety and efficacy of such product, the development work that is to be completed by the Buyer and its Affiliates (or any Third Party), the development of other products in the pipeline of the Buyer and its Affiliates, its competitiveness compared to alternative Third Party products, the proprietary position of the product (including scope and duration of relevant patents), the scope, timing and likelihood of regulatory approval, the regulatory status of the product, whether the product is subject to a clinical hold, recall or market withdrawal, and the anticipated reimbursability and pricing of the product, and (ii) refrain from taking any action in bad faith or without reasonable basis which is primarily intended to prevent, or the primary effect of which is to prevent, the realization of the achievement of the First Milestone and the Second Milestone.
2.2.4.2 In the event that the Buyer breaches any of its obligations pursuant to Section 2.2.4.1 prior to achieving the Second Milestone and has failed to cure such breach within 15 Business Days of its receipt of notice in writing from the Sellers Representative of such breach, the Buyer, if requested by Sellers’ Representative in writing within 10 Business Days of the expiry of such Buyer cure period, shall, and shall cause its Affiliates, including the Corporation, to (i) assign all rights, title and interest in and to the Lead Product held by it or its Affiliates to an entity then specified in writing by the Sellers’ Representative, and (ii) grant to such entity all of its right, title and interest in and to: (A) all data relating to non-clinical and clinical studies conducted on, and all filings made with regulatory agencies specifically with respect to, the Lead Product and (B) any regulatory approvals and regulatory documentation specifically relating to the Lead Product. Such transfer of the Lead Product shall be on an “as is, where is” basis with no representations, warranties, indemnities from the Buyer or the Corporation whatsoever, all of which being expressly disclaimed, and will be in full satisfaction of any Buyer, Corporation, or any Affiliate, liability or obligation, if any, to the Sellers on account of the Purchase Price and/or pursuant to this Section 2.2, other than such amounts, if any, then due and owing to the Sellers pursuant to the express provisions of Section 2.2.1 or Section 2.2.2 of this Agreement that have not been paid in full or set-off in accordance with the provisions of this Agreement; provided that, and for greater certainty, the amounts then due and owing to the Sellers shall only comprise the Milestone Payments and Quarterly Earnout Payments, if any, then due and owing at the time of such breach, and shall not include any amount not then due and owing to the Sellers including any damages or similar amounts resulting from such breach or any acceleration of any amounts on account of the Purchase Price. As a condition, and in consideration, of the grant and transfer of rights by the Buyer pursuant to this section, the Sellers covenant and agree to (a) pay contemporaneous with and as a condition precedent to any such grant and transfer any and all reasonable costs and expenses incurred by the Buyer or the Corporation (including, without limitation, any sales or transfer Tax that may arise) in connection with any such transfer or assignment; (b) assume all obligations of the Buyer or Corporation relating to the Lead Product arising pursuant to the [***] Agreement and the [***] Agreement and any other obligations previously approved in writing by the Sellers’ Representative or that
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
are otherwise acceptable to the Sellers’ Representative, and the Sellers shall indemnify and hold harmless the Buyer and the Corporation for such obligations; and (c) deliver a release to the Buyer and the Corporation of any claims against the Buyer or the Corporation on terms reasonably acceptable to the Buyer and the Corporation.
2.2.4.3 Notwithstanding any other provision in this Agreement including, without limitation, Article 6, the Sellers’ rights and recourse pursuant to this Section 2.2.4 shall be the Sellers’ sole and exclusive remedy for a breach by the Buyer of its obligations pursuant to Section 2.2.4.1.
2.2.5 Return of Rights to the Lead Product if Cessation of Development.
2.2.5.1 The Buyer may, at any time and in the Buyer’s sole and absolute discretion, decide to cease or discontinue all development activities with regard to the Lead Product.
2.2.5.2 In the event that the Buyer decides to cease or discontinue all development activities with regard to the Lead Product prior to achieving the Second Milestone, the Buyer shall notify the Sellers’ Representative of its decision and, if requested by Sellers’ Representative in writing within 20 Business Days of the receipt of such notice, shall, and shall cause its Affiliates, including the Corporation, to, (i) assign all rights, title and interest in and to the Lead Product held by it or its Affiliates to an entity then specified in writing by the Sellers’ Representative, and (ii) grant to such entity all of its right, title and interest in and to: (A) all data relating to non-clinical and clinical studies conducted on, and all filings made with regulatory agencies specifically with respect to, the Lead Product and (B) any regulatory approvals and regulatory documentation specifically relating to the Lead Product. Such transfer of the Lead Product shall be on an “as is, where is” basis with no representations, warranties, indemnities from the Buyer or the Corporation whatsoever, all of which being expressly disclaimed, and will be in full satisfaction of any Buyer, Corporation, or any Affiliate, liability or obligation, if any, to the Sellers on account of the Purchase Price and/or pursuant to this Section 2.2, other than such amounts, if any, then due and owing to the Sellers pursuant to the express provisions of Section 2.2.1 or Section 2.2.2 of this Agreement that have not been paid in full or set-off in accordance with the provisions of this Agreement; provided that, and for greater certainty, the amounts then due and owing to the Sellers shall only comprise the Milestone Payments and Quarterly Earnout Payments, if any, then due and owing at the time of such determination by the Buyer, and shall not include any amount not then due and owing to the Sellers including any damages or similar amounts resulting from such determination by the Buyer or any acceleration of any amounts on account of the Purchase Price. As a condition, and in consideration, of the grant and transfer of rights by the Buyer pursuant to this section, the Sellers covenant and agree to (a) pay contemporaneous with and as a condition precedent to any such grant and transfer any and all reasonable costs and expenses incurred by the Buyer or the Corporation (including, without limitation, any sales or transfer Tax that may arise) in connection with any such transfer or assignment; and (b) assume all obligations of the Buyer or Corporation relating to the Lead Product arising pursuant to the [***] Agreement and the [***] Agreement and any other obligations previously approved in writing by the Sellers’ Representative or that are otherwise acceptable to the Sellers’ Representative, and the Sellers shall indemnify and hold harmless the Buyer and the Corporation for such obligations.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
2.2.5.3 In the event that the Buyer decides to cease or discontinue all development activities with regard to the Lead Product following achieving the Second Milestone, none of the Buyer, the Corporation or any of their Affiliates shall have any further obligation or liability whatsoever to the Sellers on account of the Purchase Price and/or pursuant to this Section 2.2 as a result of the Buyer’s decision to cease or discontinue such development activities with regard to the Lead Product, other than such amounts, if any, then due and owing to the Sellers pursuant to the express provisions of Section 2.2.1 or Section 2.2.2 of this Agreement that have not been paid in full or set-off in accordance with the provisions of this Agreement; provided that, and for greater certainty, the amounts then due and owing to the Sellers shall only comprise the Milestone Payments and Quarterly Earnout Payments, if any, then due and owing at the time of such determination by the Buyer, and shall not include any amount not then due and owing to the Sellers including any damages or similar amounts resulting from such determination by the Buyer or any acceleration of any amounts on account of the Purchase Price.
2.2.5.4 Notwithstanding any other provision in this Agreement including, without limitation, Article 6, the Sellers’ rights and recourse pursuant to Section 2.2.4 or this Section 2.2.5 shall be the Sellers’ sole and exclusive remedy for any breach by the Buyer of its obligations under Section 2.2.4.1 or any discontinuance by the Buyer or the Corporation of such development activities for any reason whatsoever.
2.3 Allocation of Payments
The Purchase Price will be paid to the Designated Paying Agent who will [***]. The Buyer’s obligations with respect to the payment of the Purchase Price will be fully and finally discharged upon the Buyer delivering to the Designated Paying Agent, from time to time, the full amount of any portion of the Purchase Price that is then due and the Buyer shall have no obligation to oversee, and no liability to any Seller in respect of, any allocation of the Purchase Price to and among the Sellers (or any of them), or any failure to allocate all or any portion of the Purchase Price, by the Designated Paying Agent. For the avoidance of any doubt, neither the Buyer nor any of its Affiliates (including the Corporation) shall have any obligation with respect to the allocation and distribution of any portion of the Purchase Price among the Sellers and shall not be liable for any determination made by the Designated Paying Agent with respect to such allocations and distributions, or any dispute among the Sellers and/or between the any of the Sellers and the Designated Paying Agent with respect to the distribution of or failure to distribute all or any portion of the Purchase Price. The Sellers acknowledge and agree that the Designated Paying Agent is the agent of the Sellers and will be receiving payments of the Purchase Price from the Buyer in its capacity as agent of the Sellers and in no other capacity.
2.4 Payment of Purchase Price
2.4.1 The Buyer will pay and satisfy the Purchase Price by paying each Milestone Payment (within 30 days of attainment of the applicable Milestone) and each Quarterly Earnout Payment (upon determination of the applicable amount in accordance with Section 2.2.2.2), subject to adjustment after the Closing in accordance with Section 6.11 (Indemnity Adjustments to Purchase Price), as follows:
2.4.1.1 the Buyer will pay to the Designated Payment Agent on behalf of the Sellers, in aggregate:
2.4.1.1.1. the amount of any Milestone Payment that is due pursuant to Section 2.2.1 (Purchase Price), and;
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
2.4.1.1.2. each Quarterly Earnout Payment determined in accordance with Section 2.2.2.2.
2.4.2 Each payment of the Purchase Price (each a “Purchase Price Instalment”):
2.4.2.1 that is a Milestone Payment may be satisfied by any of the following as determined by the Buyer from time to time in its absolute discretion:
2.4.2.1.1. by wire transfer of immediately funds to the account designated by the Designated Paying Agent; or
2.4.2.1.2. by an issuance to each Seller or as each such Sellers may, in writing direct of such number of Common Shares in the share capital of the Buyer as have a value (calculated by reference to the Benchmark Price Per Share), as at the date of the payment, equal to the portion of the Milestone Payment then due to such Seller, provided that share certificates evidencing such issued Common Shares shall be delivered by the Buyer to Designated Paying Agent; or
2.4.2.1.3. partly in accordance with Section 2.4.2.1.1 and partly in accordance with Section 2.4.2.1.2; and
2.4.2.2 that is a Quarterly Earnout Payment shall be satisfied by wire transfer of immediately funds to the account designated by the Designated Paying Agent.
2.5 Withholding Rights
The Buyer shall be entitled to deduct and withhold from the consideration otherwise payable to any Person pursuant to this Article 2, and is hereby irrevocably directed by the Sellers to withhold, such amounts as may be required to be deducted and withheld with respect to the making of such payment under any provision of applicable Law. To the extent that amounts are so deducted and withheld by the Buyer and remitted to the applicable Governmental Authority, such amounts shall be treated for all purposes of this Agreement as having been paid to the Person in respect of which the Buyer made such deduction and withholding.
2.6 Capitalised Value.
The parties agree that the capitalised value of the payments of the Purchase Price pursuant to this Article 2 is, according to the Section 12B of the Danish tax assessment act (Ligningsloven), CDN$[***]. The parties further agree that they will each prepare and file any Tax Return related to the transactions contemplated by this Agreement in a manner consistent with that value provided that, in the event that a relevant Governmental Authority finally determines that [***], then any Tax Return related to the transactions contemplated by this Agreement will be prepared and filed in a manner consistent with the value as finally determined by such Governmental Authority.
ARTICLE 3
REPRESENTATIONS AND WARRANTIES
3.1 Disclosure Schedule
Each exception to the representations and warranties that is set out in the Disclosure Schedule is identified by reference to one or more specific individual Sections of this Agreement and is only effective to create an exception to each specific individual Section listed. Any statement in this Agreement that is not expressly qualified by a reference to an exception in the Disclosure Schedule will prevail, despite
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
anything to the contrary that is disclosed in the Disclosure Schedule. The exceptions to representations and warranties set out in the Disclosure Schedule are intended only to qualify and limit the representations and warranties of the Corporation and the Sellers, and will not be construed to expand in any way the scope or effect of any of those representations and warranties, and will not be construed to constitute a new representation, warranty or covenant of the Corporation or any Seller. The disclosure of any matter in the Disclosure Schedule will not be construed as an admission or indication that the matter is material or that the matter is necessarily required to be disclosed in order for any representation or warranty in this Agreement to be true and correct, and will not be construed as an admission of any obligation or liability to any third party. No disclosure in the Disclosure Schedule relating to any possible breach or violation of any Contract or Law will be construed as an admission or indication that any breach or violation exists or has actually occurred.
3.2 Representations Relating to the Sellers
Each Seller, on its own behalf, represents and warrants to the Buyer as follows, and acknowledges that the Buyer is relying upon these representations and warranties in connection with the purchase of the Purchased Shares, despite any investigation made by or on behalf of the Buyer.
3.2.1 Corporate Existence of Seller. If the Seller is not an individual, the Seller is a corporation duly incorporated and validly existing under the Laws of the jurisdiction set out in Schedule A opposite that Seller’s name under the heading “Jurisdiction”.
3.2.2 Capacity and Authority. If the Seller is not an individual, the Seller has all necessary corporate power, authority and capacity to enter into and perform its obligations under this Agreement. If the Seller is an individual, the Seller is of the full age of majority and has the legal capacity and competence to enter into and perform its obligations under this Agreement.
3.2.3 Binding Obligation. This Agreement has been duly executed and delivered by the Seller and constitutes a valid and binding obligation of the Seller, enforceable against the Seller in accordance with its terms, subject to applicable bankruptcy, insolvency and other Laws of general application limiting the enforcement of creditors’ rights generally and to the fact that equitable remedies, including specific performance, are discretionary and may not be ordered in respect of certain defaults.
3.2.4 Title to Purchased Shares. The Seller is the legal and beneficial owner of the number of Purchased Shares set out in Schedule A opposite that Seller’s name under the heading “Number of Purchased Shares Owned by Seller”, and has good title to them, free and clear of any Encumbrance except for any restriction on transfer contained in the Constating Documents. At Closing, the Seller will have the absolute and exclusive right to sell those Purchased Shares to the Buyer as contemplated by this Agreement.
3.2.5 Residence of Seller. The Seller is resident in the jurisdiction set out in Schedule A opposite that Seller’s name under the heading “Jurisdiction”, and, except as disclosed in the Disclosure Schedule, is not a non-resident of Canada for purposes of the ITA.
3.2.6 Absence of Conflict. None of the execution and delivery of this Agreement by the Seller, the performance of the Seller’s obligations under this Agreement, or the completion by the Seller of the transactions contemplated by this Agreement will:
3.2.6.1 result in or constitute a breach of any term or provision of, or constitute a default under, any Contract to which the Seller is a party or which affects the Purchased Shares owned by the Seller;
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
3.2.6.2 if the Seller is not an individual, result in or constitute a breach of any term or provision of, or constitute a default under, the Constating Documents of the Seller;
3.2.6.3 contravene any applicable Law; or
3.2.6.4 contravene any judgment, order, writ, injunction or decree of any Governmental Authority.
3.3 Representations Relating to the Corporation
The Corporation represents and warrants to the Buyer as follows, and acknowledges that the Buyer is relying upon these representations and warranties in connection with the purchase of the Purchased Shares, despite any investigation made by or on behalf of the Buyer.
3.3.1 Capacity and Authority. The Corporation has all necessary corporate power, authority and capacity to enter into and perform its obligations under this Agreement, to own or lease its assets and to carry on the Business as currently being conducted.
3.3.2 Binding Obligation. This Agreement has been duly executed and delivered by the Corporation and constitutes a valid and binding obligation of the Corporation, enforceable against the Corporation in accordance with its terms, subject to applicable bankruptcy, insolvency and other Laws of general application limiting the enforcement of creditors’ rights generally and to the fact that equitable remedies, including specific performance, are discretionary and may not be ordered in respect of certain defaults.
3.3.3 Restrictive Covenants. The Corporation is not a party to, or bound or affected by, any Contract containing any covenant expressly limiting its ability to compete in any line of business, or limiting its ability to transfer or move any of its assets or operations.
3.3.4 Absence of Conflict. None of the execution and delivery of this Agreement by the Corporation, the performance of the Corporation’s obligations under this Agreement, or the completion by the Corporation of the transactions contemplated by this Agreement will:
3.3.4.1 result in or constitute a breach of any term or provision of, or constitute a default under, the Constating Documents;
3.3.4.2 result in or constitute a breach of any term or provision of, or constitute a default under, any Contract to which the Corporation is a party or which affects the Purchased Shares, except as disclosed in the Disclosure Schedule;
3.3.4.3 constitute an event that would permit any party to any Contract with the Corporation to amend, cancel, terminate or sue for damages with respect to that Material Contract, or to accelerate the maturity of any Indebtedness of the Corporation, or other obligation of the Corporation, under that Material Contract, except as disclosed in the Disclosure Schedule;
3.3.4.4 result in the creation or imposition of any Encumbrance on the Purchased Shares;
3.3.4.5 contravene any applicable Law; or
3.3.4.6 contravene any judgment, order, writ, injunction or decree of any Governmental Authority.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
3.3.5 Consents. Except as disclosed in the Disclosure Schedule, there is no requirement to obtain any consent, approval or waiver of a party under any Material Contract in order to complete the transactions contemplated by this Agreement.
3.3.6 Regulatory Matters and Approvals.
3.3.6.1 No authorization, approval, order or consent of, or filing with, any Governmental Authority is required on the part of the Corporation in connection with the execution, delivery and performance of this Agreement or any other documents and agreements to be delivered under this Agreement.
3.3.6.2 The Lead Product is being and has been developed, tested, manufactured, stored, imported, exported, and distributed in compliance in all material respects with all applicable Laws, except such non-compliance as would not have a Material Adverse Effect.
3.3.6.3 The Corporation is not subject to any pending or, to the Knowledge of the Corporation, threatened Claim by the HPFB or FDA alleging that any operation or activity of the Corporation is in violation of the FDCA, the CFDA or any other Law, or any equivalent Governmental Authority outside Canada pursuant to any foreign Law.
3.3.6.4 The Corporation has made available and provided to the Buyer as of the date of this Agreement a complete and correct copy of all material communications with regulatory authorities (including, without limitation, the FDA and Health Canada and comparable Regulatory Authorities).
3.3.6.5 The Corporation has not commenced or conducted any IND-enabling or clinical investigations, studies, trials, or other studies and tests and none have been conducted on behalf of the Corporation.
3.3.6.6 The Corporation has timely filed all reports, statements, documents, registrations, filings, amendments, supplements and submissions required to be filed by it under applicable Laws. Each such filing complied and complies in all material respects with applicable Laws and any legally necessary or required updates, changes, corrections, amendments, supplements or modifications to such filings have been submitted to the applicable Governmental Authority.
3.3.6.7 The Corporation has complied in all material respects with all applicable security and privacy standards under (i) the Health Insurance Portability and Accountability Act of 1996, as amended by the Health Information Technology for Economic and Clinical Health Act of 2009, including the regulations promulgated thereunder, (ii) any comparable Laws in any other relevant jurisdiction relating to the security or privacy of medical information including PIPEDA and other substantially similar laws in any province of Canada, and (iii) any applicable state and provincial privacy Laws, except in each such case such breaches or violations as would not have a Material Adverse Effect.
3.3.6.8 There have been no manufacturing activities conducted by or on behalf of the Corporation of any products for human use, trials, administration or consumption.
3.3.6.9 No data generated by the Corporation with respect to the Lead Product that has been provided to any third party or otherwise made public is the subject of
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
any regulatory or other action, either pending or threatened, by any Governmental Authority relating to the truthfulness of such data.
3.3.6.10 Neither the Corporation nor any of its Representatives, has been convicted of any crime or engaged in any conduct that has resulted, or would reasonably be expected to result, in debarment or exclusion from clinical trials under applicable Laws. No Claims that would reasonably be expected to result in such a material debarment or exclusion of the Corporation are pending or threatened against the Corporation or any of its Representatives.
3.3.6.11 The Corporation is not a party to any consent decree, settlement order, or similar agreement with, or imposed by, any Governmental Authority.
3.3.7 Subsidiaries and Investments. The Corporation has no subsidiaries and does not own or hold, directly or indirectly, any Securities of, and does not have any other interest in, any Person and the Corporation has not entered into any agreement to acquire any such interest.
3.3.8 Corporate Existence of Corporation. The Corporation has been duly incorporated and organized, and is validly existing and in good standing as a corporation under the Business Corporations Act (Ontario). No proceedings have been taken or authorized by the Corporation in respect of the bankruptcy, insolvency, liquidation, dissolution or winding up of the Corporation.
3.3.9 Constating Documents. The Constating Documents include all of the charter documents of the Corporation and are in full force and effect. No action has been taken to amend the Constating Documents and no changes to the Constating Documents are planned.
3.3.10 Jurisdictions. The Corporation is qualified to do business in the Province of Ontario. The nature of the Business does not require qualification to do business in any other jurisdiction.
3.3.11 Authorized and Issued Capital. The authorized capital of the Corporation consists of an unlimited number of common shares, of which 13,974,378 common shares, and no other shares, are issued and outstanding as fully paid shares.
3.3.12 No Purchase Rights. No Person has any written or oral agreement or option or any right or privilege (whether by Law, pre-emptive, contractual or otherwise) capable of becoming an agreement or option, including Securities, warrants or convertible obligations of any kind, for:
3.3.12.1 the purchase of any Securities of the Corporation; or
3.3.12.2 the purchase of any of the assets of the Corporation, other than in the ordinary course of the Business.
3.3.13 Corporate Records. The corporate records and minute books of the Corporation have been maintained in accordance with all applicable statutory requirements and are complete and accurate in all material respects. All those corporate records and minute books of the Corporation have been made available to the Buyer.
3.3.14 Books and Records. The Financial Statements fairly and correctly set out and disclose in accordance with GAAP and in all material respects the financial position of the Corporation, and all material financial transactions of the Corporation have been accurately recorded in the Books and Records.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
3.3.15 Financial Statements. Copies of the Financial Statements are included in the Disclosure Schedule. The Financial Statements have been prepared in accordance with GAAP and present fairly:
3.3.15.1 the assets, liabilities (whether accrued, absolute, contingent or otherwise) and financial condition of the Corporation as at the respective dates of the Financial Statements; and
3.3.15.2 the sales, earnings and results of the operations of the Corporation during the periods covered by the Financial Statements,
but the unaudited interim financial statements:
3.3.15.3 do not contain all notes required under GAAP; and
3.3.15.4 are subject to normal year-end audit adjustments, which individually or in the aggregate would not be material to any buyer contemplating the purchase of the Purchased Shares.
3.3.16 Tax Matters.
3.3.16.1 Except as disclosed in the Disclosure Schedule:
3.3.16.1.1. all Tax Returns required by applicable Tax Law to be filed on or before the Closing Date by or on behalf of the Corporation have been or will be duly filed on a timely basis in compliance with their due dates under applicable Tax Law with the appropriate Governmental Authorities on or before the Closing Date. Each of those Tax Returns is or will be true, correct and complete in all material respects and none of them has been or will be amended;
3.3.16.1.2. the Corporation has duly, and on a timely basis, paid or remitted all Taxes required to be paid or remitted by it on or before the Closing Date, including all Taxes shown as due and owing on all Tax Returns, all Taxes assessed or reassessed by any Governmental Authority, all Taxes held in trust or deemed to be held in trust for any Governmental Authority, and all instalments on account of Taxes for the current year. The Corporation will not have any liability for Taxes for any period ending on or before the Closing Date, or that portion of any Straddle Period up to and including the Closing Date, other than those liabilities for Taxes reflected as reserves on the Financial Statements. The liabilities reflected as reserves for Taxes on the Financial Statements are sufficient for the payment or remittance of all Taxes which may become payable or remittable by the Corporation, whether or not disputed, in respect of any period ending on or before the Closing Date;
3.3.16.1.3. there are no liens for Taxes (other than for Taxes not yet due and payable) on any of the properties or assets of the Corporation, nor are those properties or assets the subject of any trust arising under Tax Law; and
3.3.16.1.4. the Corporation has not requested, executed, received, or entered into any Contract relating to any waiver, which is still outstanding
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
and which provides for any extension of time in respect of: (i) the assessment, reassessment or collection of any Taxes by any Governmental Authority; (ii) the filing of any Tax Returns in respect of any Taxes for which the Corporation is or may be liable; or (iii) the payment or remittance of any Taxes or amounts on account of Taxes.
3.3.16.2 The Corporation has not been required, and is not currently required, to file any Tax Returns with any Governmental Authority outside Canada or outside the Province of Ontario. No Claims have ever been made by any Governmental Authority that the Corporation is or may be subject to Tax in a jurisdiction where the Corporation does not file Tax Returns. There is no basis for a Claim that the Corporation is subject to Tax in a jurisdiction in which it does not file Tax Returns.The Corporation has received notices of assessment and reassessment from all relevant Governmental Authorities with respect to its Tax liabilities for all taxation years and other periods ending [***].
3.3.16.3 Except as disclosed in the Disclosure Schedule, there are no Tax deficiencies that have been claimed, proposed or asserted in writing against the Corporation that have not been fully paid or finally settled and there are no discussions, audits, assertions or Claims now pending, or to the Knowledge of the Corporation, threatened, in respect of Taxes due from the Corporation. No Governmental Authority has challenged, disputed or questioned in writing any Taxes of or any Tax Returns filed by the Corporation or indicated that an assessment, reassessment or determination in respect of Taxes is proposed.
3.3.16.4 All Taxes required to be deducted, withheld or remitted by the Corporation under any applicable Tax Law from amounts paid or credited by it to or for the account or benefit of any Person, including Taxes on payments to any of its present or Former Employees, officers or directors and Taxes on payments to any Person who is a non-resident of Canada, have been properly deducted, withheld and remitted on a timely basis to the appropriate Governmental Authorities.
3.3.16.5 Copies of all Tax rulings pertaining to the Corporation have been provided to the Buyer. There is no Claim or, to the Knowledge of the Corporation, threatened Claim to revoke any such Tax ruling. Except as disclosed in the Disclosure Schedule, there are no Tax rulings or requests for Tax rulings pertaining to the Corporation that could affect the liability for Taxes or the amount of the taxable income or loss for any taxation year or period ending after the Closing Date.
3.3.16.6 Except as disclosed in the Disclosure Schedule, there are no amounts outstanding and unpaid for which the Corporation has previously claimed a deduction from income under the ITA or any other applicable Tax Law and which may be included in the Corporation’s income for any taxation year ending after the Closing Date.
3.3.16.7 The Corporation has not, directly or indirectly, transferred property to or acquired property from or provided services to or received services from any Person with whom the Corporation was not dealing at Arm’s Length, for consideration the fair market value of which was less than the fair market value of the property or service at the time of (in the case of property) the disposition or acquisition of the property or (in the case of services) the provision or receipt
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
of the services, nor been a party to any contract or transaction that could result in a liability for Tax under section 160 of the ITA or any substantially similar provisions of other applicable Tax Laws. The Corporation has provided the Buyer with copies of all Contracts and any other relevant documents relating to transactions entered into by it before the Closing Date with Persons with whom the Corporation was not dealing at Arm’s Length at the time the transaction occurred.
3.3.16.8 There are no transactions or events that have resulted, and no circumstances existing, which could result, in the application to the Corporation of sections 80, 80.01, 80.02, 80.03, 80.04 of the ITA or any analogous provision of any comparable Law of any province or territory of Canada.
3.3.16.9 The Corporation is registered for purposes of the ETA and the Corporation’s goods and services tax or harmonized sales tax registration number is [***]. The Corporation has complied on a timely basis with all registration, reporting, collection, remittance and other requirements in respect of all applicable commodity and sales taxes. The Corporation has provided to the Buyer all invoices, purchase orders and all other documents as are necessary to make any claim for input tax credits, refunds or rebates claimed or to be claimed under the applicable commodity and sales taxes.The Corporation has provided to the Buyer copies of all Tax Returns for all fiscal periods for which the relevant limitation period has not expired and all working papers, calculations, and schedules relating to those Tax Returns, together with all written communications relating to those Tax Returns from any Governmental Authority and the written response, if any, of the Corporation to those communications.
3.3.16.10 For all transactions between the Corporation and any non-resident person with whom it was not dealing at Arm’s Length, the Corporation has made or obtained records or documents that meet the transfer pricing requirements of subsection 247(4) of the ITA. Except as disclosed in the Disclosure Schedule, the Corporation has not made any elections or designations for the purposes of the ITA or the ETA or other applicable Tax Law, or for the purposes of any administrative rulings or notices or administrative practices under any Tax Law.
3.3.16.11 The Corporation has not acquired any property on a Tax-deferred or rollover basis and no election under sections 13, 44, 83 or 85 of the ITA (or any substantially similar provision of any applicable Tax Law) has been made or filed by or on behalf of the Corporation with respect to the acquisition or disposition of property.
3.3.16.12 Except for elections under section 167 of the ETA and under section 75 of An Act respecting the Québec sales tax, the Corporation is not a party to any elections made under the ETA or An Act respecting the Québec sales tax.
3.3.16.13 The Corporation has not made or authorized any payments to its directors, officers, former directors, shareholders or Employees or to any Person not dealing at Arm’s Length with any of its directors, officers, former directors, shareholders or Employees, except in the ordinary course of the Business and at the regular rates payable to them of salary, pension, bonuses, rents or other payments of any nature.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
3.3.16.14 The Corporation has no loans or Indebtedness outstanding made to any Employee or to any director, former director, officer or shareholder of the Corporation or to any Person not dealing at Arm’s Length with any of those Persons.
3.3.16.15 The Corporation has been since its incorporation a Canadian-controlled private corporation as defined in the ITA.
3.3.16.16 The Corporation has not been and is now not associated, as determined for the purposes of the ITA, with any corporations. All non-capital losses not deducted but still deductible by the Corporation have been incurred and are derived from the carrying on of the Business. The Business has been carried on by the Corporation since the date of its inception for profit and with a reasonable expectation of profit.
3.3.16.17 In the three years before the date of this Agreement, the Corporation has not received or taken advantage of, directly or indirectly, any grants, Tax benefits other than as may be provided for in the ITA, subsidies, loan guarantees, government contracts, or other forms of preferential treatment or assistance from any Governmental Authority.
3.3.16.18 For purposes of the ITA, the taxation year end of the Corporation is December 31 of each year. The taxation year end of the Corporation has not changed since its incorporation.
3.3.17 Absence of Changes. Except as disclosed in the Disclosure Schedule, since December 31, 2018, there has not been any change, event or occurrence that, either individually or in the aggregate, has resulted in, or would reasonably be expected to result in, a Material Adverse Effect.
3.3.18 Absence of Undisclosed Liabilities.
3.3.18.1 Except as set forth in the Disclosure Schedule and except for liabilities and obligations specifically and adequately reserved in the Audited Balance Sheet, the Corporation does not have and has not incurred any liabilities or obligations (whether accrued, absolute, contingent or otherwise, including under any guarantee of any debt).
3.3.18.2 Except as set forth in the Disclosure Schedule, the Corporation does not have any outstanding Indebtedness.
3.3.19 Absence of Unusual Transactions. Except as disclosed in the Disclosure Schedule, since December 31, 2018, the Corporation has not:
3.3.19.1 given any guarantee of any debt, liability or obligation of any Person;
3.3.19.2 subjected, or permitted to be subjected, any of its assets to any Encumbrance other than the Permitted Encumbrances;
3.3.19.3 acquired, sold, leased or otherwise disposed of or transferred any assets, other than in the ordinary course of the Business;
3.3.19.4 made or committed to any capital expenditures, other than in the ordinary course of the Business;
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
3.3.19.5 declared or paid any dividend or otherwise made any distribution or other payment of any kind to any of its shareholders or any other Person, or taken any corporate proceedings for that purpose;
3.3.19.6 redeemed, purchased or otherwise retired any of its shares or otherwise reduced its stated capital;
3.3.19.7 entered into or become bound by any Contract, other than in the ordinary course of the Business;
3.3.19.8 amended or terminated any Contract (except for Contracts which expire by the passage of time) resulting, collectively or individually, in a material and adverse effect on the business, condition (financial or otherwise), properties, assets (tangible or intangible), liabilities (whether absolute, accrued, conditional, contingent or otherwise), capitalization, operations, prospects or results of operations of the Corporation;
3.3.19.9 waived or released any rights which it has or had, or any debts owed to it, resulting, collectively or individually, in a material and adverse effect on the business, condition (financial or otherwise), properties, assets (tangible or intangible), liabilities (whether absolute, accrued, conditional, contingent or otherwise), capitalization, operations, prospects or results of operations of the Corporation;
3.3.19.10 increased any compensation arrangement or agreement with any Employee or officer, director or shareholder of the Corporation;
3.3.19.11 changed any method of accounting or auditing practice; or
3.3.19.12 agreed or offered to do any of the things described in this Section 3.3.19.
3.3.20 Title to and Condition of Assets. The Corporation owns, possesses and has good and valid title to all of its undertakings, property and assets not otherwise the subject of specific representations and warranties in this Section 3.3, including all the undertakings, property and assets reflected in the most recent balance sheet included in the Financial Statements, free and clear of all Encumbrances other than Permitted Encumbrances. The undertakings, property and assets of the Corporation comprise all of the undertakings, property and assets necessary for the Corporation to carry on the Business as it is currently operated. All machinery, equipment, fixtures and other tangible assets owned, leased or used by the Corporation are in good operating condition and repair, ordinary wear and tear excepted, and are reasonably fit and usable for the purposes for which they are being used.
3.3.21 Real Property. The Corporation does not now hold, nor has it ever held, any interest in any real property other than right to use office space at [***] and laboratory spaced provided by [***], which arrangements have been terminated and the Corporation has no obligations, liabilities or other Indebtedness related to such arrangements. The Corporation is not currently a party to nor does it have any obligations, liabilities or other Indebtedness in respect of any leases or other arrangements in respect of real property.
3.3.22 Intellectual Property.
3.3.22.1 The Disclosure Schedule sets forth a correct and complete list of all (i) issued Patents and Patent applications, (ii) Trademark registrations and applications, (iii) Copyright registrations and applications, and (iv) material Software, in each
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
case which is owned by the Corporation, or which the Corporation has rights in (including license rights and options, other than licenses granting rights to use readily available shrink wrap or click wrap Software having a replacement cost and annual license fee of less than US$[***]), in any jurisdiction in the world. The Corporation is the sole and exclusive beneficial and, with respect to applications and registrations (including Patents), recorded owner of all of the Intellectual Property items set forth in the Disclosure Schedule which are identified as owned by the Corporation, and all such Intellectual Property that is pending or registered is subsisting. Further, the Corporation is the sole and exclusive beneficial rights owner with respect to the applications and registrations (including Patents) of all of the Intellectual Property items set for in the Disclosure Schedule which are identified as owned by the Corporation and all such Intellectual Property that is pending or registered is subsisting. For any Intellectual Property that is registrable (e.g. Patents, Trademarks, Copyright, Domain Names) and for which an application has been filed or registration has been obtained and in which the Corporation owns or has rights in, all applicable Laws have been complied with, there have been no technical or formality breaches of applicable Law (including for Patents, any obligation to submit all known prior art and payment of fees and responses to any office actions), and is not aware of any reasons nor received any communications that would indicate that any such registrations would not be valid or enforceable or that any pending applications for same would not be registered.
3.3.22.2 Copies of the Corporation’s standard form(s) of proprietary information, confidentiality and assignment agreements have been delivered to the Buyer or its counsel. All Employees, all current and former consultants of the Corporation and all other individuals who have been involved in the creation, invention or development of Corporation Intellectual Property for or on behalf of the Corporation (each, a “Contributor”) have executed the applicable form of agreement or other written agreements, all of which have been provided to the Buyer or its counsel, that assign (to the extent permitted by applicable Law) to the Corporation all rights in and to, any Intellectual Property created by such Employees and consultants in the course of their employment or consulting relationship with the Corporation. Without limiting the foregoing, no Contributor owns or has any right, including the right to assert any moral rights (except to the extent such rights are non-waivable under applicable Law) to the products of the Corporation or Intellectual Property, nor has any Contributor made any assertions with respect to any alleged ownership or rights over the foregoing.
3.3.22.3 The Disclosure Schedule sets forth a true, correct, and complete list of all Contracts (i) pursuant to which the Corporation (A) is granted or obtains or agrees to obtain any right to use any material Intellectual Property (other than standard form Contracts granting rights to use readily available shrink wrap or click wrap Software having a replacement cost and annual license fee of less than US$25,000 for all such related Contracts in the aggregate), (B) is restricted in its right to use or register any material Intellectual Property, or (C) permits or agrees to permit any other Person, to use, obtain, enforce, or register any material Intellectual Property, including any license agreements, coexistence agreements, and covenants not to sue, (ii) that would require, following the Closing, the Buyer to license, assign, or make available its or its Affiliates’ (other than Corporation) Intellectual Property to any other Person, or restrict the use by the Buyer or its Affiliates of such Intellectual Property as a result of the transactions contemplated hereby, in the case of each of clauses (i) and (ii), excluding all non-disclosure, material transfer, clinical trial and similar Contracts.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
3.3.22.4 Except as set forth in the Disclosure Schedule:
3.3.22.4.1. The Corporation owns, or has a valid right to use, or can develop or obtain rights to use on commercially reasonable terms, free and clear of all Liens, all material Intellectual Property used or held for use in the business of the Corporation.
3.3.22.4.2. Except for research use exceptions in any jurisdiction worldwide, (A) the conduct of the business of the Corporation (including the products and services of the Corporation), as conducted in the past three years, as currently conducted, and as currently contemplated to be conducted for future commercialization (including the practice of any Intellectual Property in which it owns or has rights and the commercialization of the Lead Product) has not, does not and will not infringe, misappropriate, or otherwise violate, in each case in a manner that is or would be material to the Corporation, any Person’s Intellectual Property, and (B) there has been no such claim asserted or threatened in writing (including in the form of offers or invitations to obtain a license) in the past three (3) years against the Corporation.
3.3.22.4.3. To the Knowledge of the Corporation, no Person is materially infringing, misappropriating, or otherwise violating any Intellectual Property owned, used, or held for use by the Corporation (or in which the Corporation has rights), and no such claims have been asserted or threatened in writing against any Person by the Corporation in the past three (3) years.
3.3.22.4.4. There has been no Claim asserted or threatened in writing by any Person against the Corporation challenging the scope, validity, or enforceability of any applications or registrations for Patents or Trademarks owned by the Corporation. The Corporation has not granted any Person any right to control the prosecution or registration of any material Intellectual Property owned by the Corporation or to commence, defend, or otherwise control any claim with respect to such Intellectual Property.
3.3.22.4.5. The Corporation takes reasonable measures to protect the confidentiality of material Trade Secrets. To the Knowledge of the Corporation, there has not been any disclosure of any material Trade Secret of the Corporation (including any such information of any other Person disclosed in confidence to the Corporation) to any Person in a manner that has resulted or is likely to result in the loss of such Trade Secret or other rights in and to such information.
3.3.22.4.6. Each Employee and Former Employee and officer of the Corporation is bound by proprietary information provisions contained in their employment or contracting agreements with the Corporation substantially in the form made available to the Buyer prior to the date hereof.
3.3.22.4.7. Except as disclosed in the Disclosure Schedule, no current or former director, shareholder, officer, or Employee of the Corporation will, after giving effect to the transactions contemplated hereby,
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
own, license, or retain any proprietary rights of the Corporation in and to Intellectual Property owned, used, or held for use (including for defensive purposes) by the Corporation.
3.3.22.4.8. The consummation of the transactions contemplated by this Agreement will not result in the loss or impairment of or payment of any additional amounts with respect to, nor require the consent of any other Person in respect of, the Corporation’s right to own, use, or hold for use any of the Intellectual Property as owned, used, or held for use (including for defensive purposes) as of the date hereof.
3.3.22.4.9. The Corporation has at all times complied in all material respects with all applicable material Legal Requirements relating to privacy, data protection, and the collection and use of personal information collected, used, or held for use by or on behalf of the Corporation. No claims have been asserted or threatened in writing against the Corporation alleging a violation of any Person’s privacy or personal information or data rights and the consummation of the transactions contemplated hereby will not breach or otherwise cause any violation of any Legal Requirement or procedure related to privacy, data protection, or the collection and use of personal information collected, used, or held for use by or on behalf of the Corporation, except such breaches or violations as would not have a Corporation Material Adverse Effect. The Corporation takes reasonable measures to ensure that such information is protected against unauthorized access, use, modification, or other misuse.
3.3.22.5 With respect to the [***] Agreement and except as set forth in the Disclosure Schedule:
3.3.22.5.1. There have been no activities conducted and there are no ongoing activities being conducted or approved or consented to by the Corporation under Part II of the Project, and there have been no Targets selected and no Compounds screened;
3.3.22.5.2. No Project IP has been developed or has arisen from Part II of the Project and all Project IP relating or pertaining to the Lead Project has been developed under Part I of the Project; and
3.3.22.5.3. The Corporation has been granted a perpetual worldwide, exclusive license in whole and without limitations, and for no other consideration than that set out at Article 3.0 of the [***] Agreement for any and all Project IP relating or pertaining to the Lead Product and any Lead Targets (including any art, process, machine, manufacture or composition of matter, or any new and useful improvement in any art, process, machine, manufacture or composition of matter relating or pertaining to the Lead Product or the commercialization thereof, including the exclusive right, privilege and liberty of making, constructing and using the Project IP relating or pertaining to any of the foregoing and selling it to others to be used).
The terms “Part I”, “Part II”, “Project”, “Targets”, “Compounds”, and “Project IP” as used in this Section 3.3.22.5 have the meanings as ascribed to them in the [***] Agreement.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
3.3.23 Accounts Receivable. All accounts receivable of the Corporation reflected in the Financial Statements, or which have come into existence since the date of the most recent Financial Statements, were created in the ordinary course of the Business from bona fide arm’s length transactions, and, except to the extent that they have been paid in the ordinary course of the Business since the date of the Financial Statements, are valid and enforceable and payable in full, without any right of set-off or counterclaim or any reduction for any credit or allowance made or given, except to the extent of the allowance for doubtful accounts reflected in the Financial Statements and, in the case of accounts receivable which have come into existence since the date of the most recent Financial Statements, of a reasonable allowance for doubtful accounts, which allowances are, and will as of the Closing Date be, adequate and calculated in a manner consistent with GAAP.
3.3.24 Inventories. The Inventories have been accumulated by the Corporation for use in the ordinary course of the Business. The present levels of the Inventories are consistent with those maintained in the ordinary course of the Business.
3.3.25 Material Contracts. The Disclosure Schedule lists all Material Contracts. The Corporation is not in default or breach of any Material Contract, and, to the Knowledge of the Corporation, there exists no state of facts which, after notice or lapse of time or both, would constitute a default or breach under any Material Contract. To the Knowledge of the Corporation, no counterparty to any Material Contract is in default of any of its obligations under any Material Contract, the Corporation is entitled to all benefits under each Material Contract, and the Corporation has not received any notice of termination of any Material Contract.
3.3.26 Accounts and Powers of Attorney. The Disclosure Schedule lists:
3.3.26.1 the name of each bank or other depository in which the Corporation maintains any bank account, trust account or safety deposit box and the names of all individuals authorized to draw on them or who have access to them; and
3.3.26.2 the name of each Person holding a general or special power of attorney from the Corporation and a summary of its terms.
3.3.27 Compliance with Laws. The Corporation is, and at all times since the date of its incorporation has been, conducting the Business in compliance with all applicable Laws, and the Corporation has not received notice of any violation by the Corporation of any Laws.
3.3.28 Permits. The Corporation does not have any material Permits and no such Permits are required to enable the Corporation to carry on the Business as currently conducted and to enable the Corporation to own, lease and operate its assets.
3.3.29 Environmental Conditions.
3.3.29.1 The Corporation, the conduct by the Corporation of the Business has been and are in compliance with all applicable Environmental Laws and there are no facts that would reasonably be expected to give rise to any non-compliance by the Corporation with any Environmental Laws, either in the conduct by the Corporation of the Business, or in Corporation’s prior use of office or laboratory premises provided by third parties.
3.3.29.2 The Corporation has all Permits required by all Environmental Laws for the conduct by the Corporation of the Business, and the Corporation is in compliance with all those Permits.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
3.3.29.3 The Corporation, and any Person for whom the Corporation is responsible under all Environmental Laws, have imported, manufactured, processed, distributed, used, treated, stored, disposed of, transported, exported or handled Hazardous Substances in strict compliance with all Environmental Laws.
3.3.29.4 There has been no Release of any Hazardous Substance in the course of the Business, except in compliance with all Environmental Laws.
3.3.29.5 There has been no Remedial Order issued to the Corporation in respect of the Business and, to the Knowledge of the Corporation, no Remedial Orders are threatened, and there are no facts that facts that would reasonably be expected to give rise to any Remedial Orders.
3.3.29.6 The Corporation has not received any notice of Claim, summons, order, direction or other communication relating to non-compliance with any Environmental Laws from any Governmental Authority or other third party.
3.3.29.7 There is no pending or, to the Knowledge of the Corporation, threatened matter, act or fact that could cause the Corporation or the conduct of the Business to be no longer in compliance with all applicable Environmental Laws.
3.3.30 Suppliers. The Disclosure Schedule lists each material supplier of goods and services from whom the Corporation has purchased goods or services in the 12 months immediately preceding the date of this Agreement.
3.3.31 Rights to Use Personal Information.
3.3.31.1 All Personal Information in the possession of the Corporation has been collected, used and disclosed in compliance with all applicable Privacy Laws in those jurisdictions in which the Corporation conducts, or is deemed by operation of law in those jurisdictions to conduct, the Business.
3.3.31.2 The Corporation has disclosed to the Buyer all Contracts and facts concerning the collection, use, retention, destruction and disclosure by the Corporation of Personal Information, and there are no other Contracts or facts which, on completion of the transactions contemplated by this Agreement, would restrict or interfere with the use of any Personal Information by the Corporation in the continued operation of the Business as conducted before the Closing.
3.3.31.3 There are no Claims pending or, to the Knowledge of the Corporation, threatened with respect to the Corporation’s collection, use or disclosure of Personal Information.
3.3.32 Product Warranties. The Corporation has not given any warranties to buyers or users of products or services supplied by or on behalf of the Corporation and there are no Claims against the Corporation on account of warranties or with respect to the production or sale of products or the provision of services, nor is there any basis for any possible prospective Claim against, or Loss on the part of, the Corporation arising from, relating to, or in connection with the production or sale of products or the provision of services.
3.3.33 Employees and Employment Contracts.
3.3.33.1 The Corporation has no Employees. [***] and [***] are the only individuals currently providing services to the Corporation.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
3.3.33.2 There are no employment Law-related Claims pending, commenced or, to the Knowledge of the Corporation, threatened, and there exists no state of facts that could reasonably be expected to give rise to any such Claims. There are no outstanding employment Law-related orders, awards or rulings involving the Corporation.
3.3.33.3 The employment, engagement or retainer of all Former Employees has been terminated in accordance with their respective agreements and applicable Law. The Corporation has complied with all of its obligations to such employees and has no outstanding or undischarged liability to any such Former Employee and each such Former Employee has executed and delivered an unconditional release to the Corporation in respect of all obligations and liabilities that the Corporation owed to such Former Employee.
3.3.34 Employee Confidentiality Agreements. The Corporation has entered into enforceable confidentiality agreements, true and complete copies of which have been provided to the Buyer, with all of its relevant Employees that protect confidential information relating to, and the intellectual property of, the Corporation and third party licensors. All current and former officers and Employees who have been involved in the development, modification or use of Intellectual Property have assigned all of their rights, title and interest in and to the Intellectual Property to the Corporation, have expressly waived any moral rights in the Intellectual Property, and have executed and delivered written agreements to the Corporation to that effect.
3.3.35 Unions. There are no apparent or, to the Knowledge of the Corporation, threatened union organizing activities involving Employees, and there have not been any union organizing activities since the Corporation’s incorporation.
3.3.36 Pension and Benefit Plans.
3.3.36.1 The Disclosure Schedule lists and describes each Plan. True and complete copies of each written Plan, as amended to the date of this Agreement, have been made available to the Buyer. There have been no promised improvements, increases or changes to the benefits provided under any Plan, whether legally binding or not.
3.3.36.2 The Corporation has never (i) sponsored or participated in a defined benefit pension plan, or (ii) participated in a multi-employer pension plan as that term is defined under the Pension Benefits Act (Ontario) or the Pension Benefits Standards Act, 1985 (Canada), or in any similar type of plan for purposes of any other applicable pension benefits standards legislation.
3.3.36.3 All liabilities of the Corporation (whether accrued, absolute, contingent or otherwise) related to all Plans have been fully disclosed in the Financial Statements.
3.3.37 Insurance Policies. The Disclosure Schedule lists all Insurance Policies in force as of the date of this Agreement, copies of which have been made available to the Buyer. All Insurance Policies are in full force and effect and the Corporation (i) is not in default, whether as to the payment of premiums or otherwise, under any material term or condition of any of the Insurance Policies and (ii) has not failed to give notice or present any Claim under any of the Insurance Policies in a due and timely fashion.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
3.3.38 Litigation.
3.3.38.1 There are no Claims, whether or not purportedly on behalf of or against the Corporation, pending, commenced, or, to the Knowledge of the Corporation, threatened, that might reasonably be expected to have a Material Adverse Effect or that might result in an Encumbrance against the undertakings, property or assets of the Corporation.
3.3.38.2 There is no outstanding judgment, decree, order, ruling or injunction in favour of, against or otherwise involving the Corporation or relating in any way to the transactions contemplated by this Agreement.
3.3.39 Brokerage Fees. The Corporation has not retained any financial advisor, broker, agent or finder, or entered into any agreement entitling any Person to any broker’s commission, finder’s fee or similar payment, relating to this Agreement or the transactions contemplated by this Agreement.
3.3.40 Disclosure. No representation or warranty or other statement made by the Corporation or the Sellers in this Agreement or otherwise in connection with the transactions contemplated by this Agreement contains any untrue statement of material fact or omits to state a material fact necessary to make those statements, in light of the circumstances in which they were made, not misleading.
3.4 Representations Relating to the Buyer
The Buyer represents and warrants to the Sellers as follows, and acknowledges that the Sellers are relying upon these representations and warranties in connection with the sale of the Purchased Shares, despite any investigation made by or on behalf of any of the Sellers.
3.4.1 Corporate Existence of Buyer. The Buyer is a company duly incorporated and validly existing under the laws of Denmark.
3.4.2 Capacity and Authority. The Buyer has all necessary corporate power, authority and capacity to enter into and perform its obligations under this Agreement.
3.4.3 Binding Obligation. This Agreement has been duly executed and delivered by the Buyer and constitutes a valid and binding obligation of the Buyer, enforceable against the Buyer in accordance with its terms, subject to applicable bankruptcy, insolvency and other Laws of general application limiting the enforcement of creditors’ rights generally and to the fact that equitable remedies, including specific performance, are discretionary and may not be ordered in respect of certain defaults.
3.4.4 Absence of Conflict. None of the execution and delivery of this Agreement by the Buyer, the performance of the Buyer’s obligations under this Agreement, or the completion by the Buyer of the transactions contemplated by this Agreement, will result in or constitute a breach of any term or provision of, or constitute a default under, the constating documents of the Buyer or any agreement or other commitment to which the Buyer is a party.
3.4.5 Regulatory Approvals. No authorization, approval, order or consent of, or filing with, any Governmental Authority is required on the part of the Buyer in connection with the execution, delivery and performance of this Agreement or any other documents and agreements to be delivered under this Agreement.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
ARTICLE 4
COVENANTS
4.1 Delivery of Books and Records
At the Closing Time, the Sellers will cause to be delivered to the Buyer all of the Books and Records, including copies of all of the Insurance Policies. The Buyer agrees that it will preserve the Books and Records so delivered to it for a period of two years after the Closing Date, or for any longer period required by any applicable Law, and will permit the Sellers or their authorized Representatives reasonable access to them in connection with the affairs of the Sellers, but the Buyer will not be responsible or liable to the Sellers for or as a result of any accidental loss or destruction of or damage to any Books and Records.
4.2 Tax Returns
The Buyer will cause to be prepared and filed on a timely basis all Tax Returns for the Corporation for any period that ends on or before the Closing Date and for which Tax Returns have not been filed as of the Closing Date. The Buyer will also cause to be prepared and filed on a timely basis all Tax Returns for the Corporation for all Straddle Periods (all these Tax Returns together with the Tax Returns referred to in the first sentence of this Section 4.2 being referred to as “Stub Period Returns”). The Buyer shall prepare each Stub Period Return on a basis consistent with (i) applicable Law and GAAP, and, to the extent not inconsistent with applicable Law and GAAP, (ii) the Closing Balance Sheet, and (iii) the past practices and procedures of the Corporation. The Sellers and the Buyer will cooperate fully with each other and make available to each other in a timely fashion all data and other information as may reasonably be required for the preparation of all Stub Period Returns and will preserve that data and other information until the expiration of any applicable limitation period for maintaining books and records under any applicable Tax Law with respect to the Stub Period Returns. The Buyer will provide to the Sellers for their review, at least 30 Business Days before the relevant filing deadline, a copy of the Stub Period Returns and, before filing the Stub Period Returns, will take into account, acting reasonably, any comments of the Sellers on the Stub Period Returns. The Buyer will pay all of the costs of the preparation and filing of the Stub Period Returns. In addition, the Buyer will cause the Corporation to make, and file, an election under subsection 256(9) of the ITA in its Tax Return under the ITA for the taxation year ending immediately before the Buyer’s acquisition of control of the Corporation on the Closing Date, such that subsection 256(9) of the ITA does not apply, and the Buyer’s acquisition of control of the Corporation for the purposes of the ITA occurs at the Closing Time, and not at the beginning of the Closing Date.
4.3 Tax Refunds.
Any refunds or credits of Taxes (including any interest paid or credited with respect thereto but net of any Taxes paid or payable by the Corporation on such interest, and net of any Taxes paid or payable by the Corporation on such refunds or credits) of, or with respect to, the Corporation for any period ending on or before the Closing Date will be for the account of the Sellers (except to the extent such refunds or credits were reflected on the Closing Balance Sheet). The Buyer shall promptly inform the Sellers of any such refunds or credits to which the Sellers may be entitled hereunder and shall pay to the Sellers an amount equal to the amount of any such refunds or credits within 10 days following the date such refunds or credits were paid or credited by the relevant Governmental Authority to the Corporation. Any payment made pursuant to this Section 4.3 shall constitute an increase to the Purchase Price.
4.4 Amendments to Tax Returns.
Except as required by applicable Law, none of the Buyer or the Corporation shall, or cause or permit their Affiliates to, without the prior written consent of the Sellers not to be unreasonably withheld, (a) refile, amend or otherwise modify any Tax Return of the Corporation filed for a period ending on or before the Closing Date or for any Straddle Period, or (b) make any Tax election that would have any
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
retroactive effect to any such period. The Sellers shall have no liability whatsoever resulting from any action by the Buyer, the Corporation or any Affiliate contrary to this Section.
ARTICLE 5
CLOSING ARRANGEMENTS
5.1 Closing
The Closing will take place at the Closing Time. All required documents may be delivered as originals or may be delivered by electronic transmission, except that the share certificates representing the Purchased Shares must be delivered in original form.
5.2 Closing Deliveries
At the Closing Time:
5.2.1 the Sellers will deliver or cause to be delivered to the Buyer the following documents and instruments:
5.2.1.1 original share certificates representing all of the issued and outstanding shares in the capital of the Corporation, duly endorsed by an effective endorsement for transfer to the Buyer;
5.2.1.2 certified copies of resolutions of the directors of the Corporation authorizing the transfer of the Purchased Shares to the Buyer;
5.2.1.3 a consulting and transitional services agreement executed by each of the Key Personnel, substantially in the form attached as Exhibit 5.2.1.3;
5.2.1.4 executed resignations effective as at the Closing Time of each director or officer of the Corporation;
5.2.1.5 releases from each Seller and each of the individuals specified in Section 5.2.1.4 of all Claims they may have against the Corporation, substantially in the form attached as Exhibit 5.2.1.5;
5.2.1.6 the Closing Balance Sheet;
5.2.1.7 the consents, if any, required under Material Contracts, on terms acceptable to the Buyer, acting reasonably;
5.2.1.8 an agreement amending the terms of the [***] Agreement, substantially in the form attached as Exhibit 5.2.1.8;
5.2.1.9 evidence of the exercise, conversion, exchange or termination in full of all contracts and options for the purchase or grant of any options, warrants, rights, additional securities, debentures, loans or notes convertible or exchangeable for any shares or other securities of the Corporation, on terms acceptable to the Buyer, acting reasonably;
5.2.1.10 all Books and Records, including copies of all of the Insurance Policies;
5.2.1.11 a full and final release from each Seller in favour of the Corporation and the Buyer, its directors, officers, employees and representatives and their
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
respective Affiliates, of all Claims (excluding obligations owing by the Buyer under this Agreement) and including in the case of Sellers who were former holders of debt, options or other convertible obligations Claims with respect to such obligations and any security therefore, substantially in the form attached as Exhibit 5.2.1.11; and
5.2.1.12 all other documentation and evidence reasonably requested by the Buyer in order to establish the due authorization and completion by each of the Sellers and the Corporation of the transactions contemplated by this Agreement, including the taking of all corporate proceedings by the boards of directors and shareholders of each non-individual Seller and the Corporation required to effectively carry out their respective obligations under this Agreement; and
5.2.2 the Buyer will deliver or cause to be delivered to the Sellers all documentation and other evidence reasonably requested by any Seller in order to establish the due authorization and completion by the Buyer of the transactions contemplated by this Agreement, including the taking of all corporate proceedings by the board of directors and the shareholders of the Buyer required to effectively carry out the obligations of the Buyer under this Agreement.
ARTICLE 6
INDEMNIFICATION AND SURVIVAL
6.1 Indemnification by the Sellers
Subject to the other provisions of this Article 6, each Seller will:
6.1.1 Indemnify and hold harmless each Buyer Indemnified Party from and against any Loss that a Buyer Indemnified Party may suffer as a result of:
6.1.1.1 any breach of any representation or warranty made by the Corporation in this Agreement;
6.1.1.2 any non-performance of any covenant or agreement of the Corporation contained in this Agreement;
6.1.1.3 any Taxes of the Corporation for any taxation year or period ending on or before the Closing Date or related to the portion of any Straddle Period up to and including the Closing Date including without limitation any Taxes (a) arising due to the conversion of any convertible debentures issued by the Corporation, (b) arising in connection with any debt forgiveness by the Corporation, and (c) due as withholding tax in respect of any payment made by the Corporation to a non- resident of Canada; and
6.1.1.4 any Indebtedness of the Corporation outstanding on the Closing Date; and
6.1.2 Indemnify and hold harmless each Buyer Indemnified Party from and against any Loss that a Buyer Indemnified Party may suffer as a result of:
6.1.2.1 any breach of any representation or warranty made by that Seller in Section 3.2 (Representations Relating to the Sellers); and
6.1.2.2 any non-performance of any covenant or agreement of that Seller contained in this Agreement.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
6.1.3 In respect of any Buyer claim for indemnification pursuant to Section 6.1.1, the Buyer shall be entitled to:
6.1.3.1 set-off all or any portion of the Buyer’s Indemnity Claim against any Purchase Price Instalment that is then or thereafter payable to the Designated Paying Agent on behalf of the Sellers; and/or
6.1.3.2 pursue from and satisfy against each Seller on a several basis, and not a joint and several basis, all or any portion of the Buyer’s Indemnity Claim in an amount equal to such Seller’s Pro Rata Share of the Purchase Price that has previously been paid to the Designated Paying Agent, less any amount previously paid by such Seller to the Buyer pursuant to any previous Indemnity Claims.
6.1.4 In respect of any Buyer claim for indemnification against a Seller pursuant to Section 6.1.2, the Buyer shall be entitled to:
6.1.4.1 set-off all or any portion of the Buyer’s Indemnity Claim against any Purchase Price Instalment that is then or thereafter payable to the Designated Paying Agent on behalf of the Sellers; and/or
6.1.4.2 pursue from and satisfy against that Seller all or any portion of such Buyer’s Indemnity Claim in an amount equal to such Seller’s Pro Rata Share of the Purchase Price that has previously been paid to the Designated Paying Agent, less any amount previously paid by such Seller to the Buyer pursuant to any previous Indemnity Claims.
6.2 Indemnification by the Buyer
Subject to the other provisions of this Article 6, the Buyer will, if the Closing occurs, indemnify and hold harmless each Seller Indemnified Party from and against any Loss that a Seller Indemnified Party may suffer as a result of:
6.2.1 any breach of any representation or warranty made by the Buyer in this Agreement or in any agreement or document delivered under this Agreement; and
6.2.2 any non-performance of any covenant or agreement of the Buyer contained in this Agreement or in any agreement or document delivered under this Agreement.
6.3 Survival Periods for Claims by Buyer Indemnified Parties
6.3.1 The covenants, representations and warranties made by the Corporation and the Sellers in this Agreement, in the relevant Closing Certificates and in any other agreements and documents delivered under this Agreement will survive the Closing and completion of the transactions contemplated by this Agreement, and will continue in full force and effect for the periods specified in this Section 6.3.
6.3.2 Subject to Section 6.3.3, a Buyer Indemnified Party may make an Indemnity Claim by delivering an Indemnity Notice in respect of such Indemnity Claim to the Sellers’ Representative:
6.3.2.1.1.1 if the Indemnity Claim relates to a breach of any representation and warranty made in Section 3.2.4 (Title to Purchased Shares), Section 3.3.11 (Authorized and Issued Capital), Section 3.3.18
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
(Absence of Undisclosed Liabilities) or Section 3.3.22 (Intellectual Property), at any time after the Closing without limitation; or
6.3.2.1.1.2 if the Indemnity Claim relates to any breach of the Fundamental Representations and Warranties, prior to the later of (i) the third anniversary of the satisfaction of the Milestone set out in Section 2.2.1.4, and (ii) the fifth anniversary of the Closing Date; or
6.3.2.1.1.3 if the Indemnity Claim relates to any breach of the representations and warranties made in Section 3.3.16 (Tax Matters), or if the Indemnity Claim is made under Section 6.1.1.3 (Taxes), prior to the later of (i) the third anniversary of the satisfaction of the Milestone set out in Section 2.2.1.4, and (ii) on or before 90 days after the last day upon which any of the relevant Governmental Authorities is entitled to assess or reassess the Corporation (or to raise Claims against the Buyer or the Corporation relating to that assessment or reassessment) with respect to any Tax for any taxation year ending on or before the Closing Date or related to the portion of any Straddle Period up to and including the Closing Date; or
6.3.2.1.1.4 in all other cases, prior to the third anniversary of the Closing Date.
6.3.3 The notice periods set out in Section 6.3.2 will not apply to, and there will be an indefinite survival period for, an Indemnity Claim based on intentional misrepresentation or fraud by the Corporation or the Sellers, or any of them, relating to this Agreement.
6.3.4 Notwithstanding anything to the contrary contained herein, provided the Buyer has made an Indemnity Claim prior to the expiry of any applicable survival period, the Buyer shall retain and be able to exercise its set-off rights herein or at Law against any portion of the Purchase Price that relates to any period, or is payable, subsequent to the expiry of such applicable survival period.
6.4 Survival Periods for Claims by Seller Indemnified Parties
6.4.1 The covenants, representations and warranties made by the Buyer in this Agreement or in any agreements or documents delivered under this Agreement will survive the Closing and completion of the transactions contemplated by this Agreement, and will continue in full force and effect for the periods specified in this Section 6.4.
6.4.2 Subject to Section 6.4.3, a Seller Indemnified Party may make an Indemnity Claim under Section 6.2.1 only if an Indemnity Notice of that Indemnity Claim is delivered to the Buyer as Indemnifying Party within 24 months after the Closing Date.
6.4.3 The notice period set out in Section 6.4.2 will not apply to, and there will be an indefinite survival period for, an Indemnity Claim based on intentional misrepresentation or fraud by the Buyer relating to this Agreement or any document delivered under this Agreement.
6.5 Amount Limitations on Indemnification Obligations
6.5.1 The indemnification obligations of the Sellers under this Article 6 are subject to the limitations, if any, set out in Section 6.1.3 and Section 6.1.4. The Sellers shall not be liable to the Buyer Indemnified Parties for indemnification in respect of Indemnity Claims under this Article 6 (other than Indemnity Claims for any breach of the representations and warranties
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
made in Section 3.3.18 (Absence of Undisclosed Liabilities) or Section 3.3.16 (Tax Matters), or if the Indemnity Claim is made under Section 6.1.1.3 (Taxes) or under Section 6.1.1.4 (Indebtedness)), until the aggregate amount of all Losses in respect of all Indemnity Claims exceeds US$50,000 (the “Basket”), in which event Sellers shall be required to pay or be liable such Losses from the first dollar without regard to the Basket or any deductible or similar credit in respect of the Basket. For the avoidance of doubt, the Sellers shall be liable for Indemnity Claims relating to any breach of the representations and warranties made in Section 3.3.18 (Absence of Undisclosed Liabilities) or Section 3.3.16 (Tax Matters) or for any Indemnity Claim made under Section 6.1.1.3 (Taxes) or under Section 6.1.1.4 (Indebtedness) from the first dollar of such Indemnity Claims without regard to the Basket or any deductible or similar credit in respect of the Basket.
6.5.2 The indemnification obligations of the Buyer under this Article 6 are limited in the aggregate to the sum of Milestone Payments and Earnout Payments that are or become due and owing by the Buyer and that have not been paid. For greater certainty, the amount of Milestone Payments and Earnout Payments that are due and owing by the Buyer at any time will be reduced by the amount of any Indemnity Claims of the Buyer that have been set-off against such payment amounts.
6.6 Rules Relating to Indemnification Obligations
The following will apply to the indemnification obligations under this Article 6.
6.6.1 Effect of Waiver. The waiver of any condition relating to any representation, warranty or covenant will not affect the right to indemnification under this Article 6 based on that representation, warranty or covenant.
6.6.2 No Double Recovery. No Indemnified Party is entitled to double recovery for any Indemnity Claim even though the Indemnity Claim may have resulted from the breach or inaccuracy of more than one of the representations, warranties, covenants and obligations of the Indemnifying Party under this Agreement, the relevant Closing Certificates or in any other agreements and documents delivered under this Agreement.
6.6.3 Materiality. In the case of an Indemnity Claim by a Buyer Indemnified Party under this Article 6 for breach by the Corporation or by the Sellers or any of them of a representation or warranty that is qualified by materiality or Material Adverse Effect, the Buyer Indemnified Party will be entitled to claim the full amount of the Loss resulting from that breach without regard to the materiality or Material Adverse Effect qualifier. However, the determination under this Article 6 of whether there has been a breach of a representation or warranty that is qualified by materiality or Material Adverse Effect will be made having regard to the materiality or Material Adverse Effect qualifier.
6.6.4 Remoteness and Mitigation. The quantum of Losses that can be recovered by an Indemnified Party under this Article 6 will not be affected by the application of principles of remoteness of damages, or the duty to mitigate.
6.7 Notice of Indemnity Claims
If an Indemnified Party becomes aware of a Loss or potential Loss in respect of which an Indemnifying Party has agreed to indemnify it under this Article 6, the Indemnified Party will promptly give written notice (an “Indemnity Notice”) of its Claim or potential Claim for indemnification (an “Indemnity Claim”) to the Indemnifying Party. The Buyer’s obligation to deliver an Indemnity Notice, and any information, documentation or notices with respect thereto provided therefor in Sections 6.7, 6.8 or 6.9 to the Sellers or any of them shall be satisfied by the Buyer delivering the Indemnity Notice or such information,
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
documentation or notices to the Sellers’ Representative, and the Buyer shall have no obligation to deliver such Indemnity Notice or such information, documentation or notices to a Seller or the Sellers. The Buyer shall have no liability or obligation for the failure of the Sellers’ Representative to deliver an Indemnity Notice or such information, documentation or notices to any of the Sellers. An Indemnity Notice must specify whether the Indemnity Claim arises as the result of a Third Party Claim or whether the Indemnity Claim is a Direct Claim, and must also specify with reasonable particularity (to the extent that the information is available):
6.7.1 the factual basis for the Indemnity Claim;
6.7.2 the amount of the Indemnity Claim, if known; and
6.7.3 if the Indemnified Party is a Buyer Indemnified Party, the portion of such Indemnity Claim that the Indemnified Party intends to set-off against Milestone Payments and Earnout Payments, in each case if known and subject to the Indemnified Party’s right to modify that determination at any time that any portion of the Indemnity Claim remains due and outstanding.
If, through the fault of the Indemnified Party, the Indemnifying Party does not receive an Indemnity Notice of an Indemnity Claim in time to effectively contest the determination of any liability capable of being contested, the Indemnifying Party will be entitled to set off against the amount claimed by the Indemnified Party the amount of any Loss incurred by the Indemnifying Party resulting from the Indemnified Party’s failure to give an Indemnity Notice on a timely basis. For greater certainty, an Indemnified Party that is a Seller will be deemed to have received an Indemnity Notice from the Buyer once the Indemnity Notice is delivered to the Sellers’ Representative.
6.8 Procedure for Direct Claims
6.8.1 Following receipt of an Indemnity Notice from the Indemnified Party of a Direct Claim, the Indemnifying Party will have 20 Business Days, or any other period of time agreed to by the Indemnifying Party and the Indemnified Party, (in either case, the “Claim Dispute Period”) to make any investigations it considers necessary or desirable. For the purpose of those investigations, the Indemnified Party will make available to the Indemnifying Party the information relied upon by the Indemnified Party to substantiate the Direct Claim, together with all other information that the Indemnifying Party may reasonably request.
6.8.2 If the Indemnifying Party disputes the validity or amount of the Direct Claim, the Indemnifying Party may provide written notice of the dispute to the Indemnified Party within the Claim Dispute Period. That dispute notice must describe in reasonable detail the nature of the Indemnifying Party’s dispute. Upon receipt of a dispute notice, the Indemnified Party may pursue all rights and remedies available to it, subject to this Agreement.
6.8.3 If the Indemnifying Party does not provide written notice of a dispute with respect to the Direct Claim within the Claim Dispute Period, the Indemnifying Party will be deemed to have agreed to the validity and amount of the Direct Claim.
6.9 Procedure for Third Party Claims
6.9.1 The Indemnified Party will promptly deliver to the Indemnifying Party copies of all correspondence, notices, assessments or other written communication received by the Indemnified Party in respect of any Third Party Claim that is or might become the basis of an Indemnity Claim.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
6.9.2 The Indemnified Party will not negotiate, settle, compromise or pay any Third Party Claim that is or might become the basis of an Indemnity Claim without the prior written consent of the Indemnifying Party, which consent will not be unreasonably withheld; provided that the foregoing does not preclude the exercise of any right of set off provided for in this Agreement or at Law, which exercise of rights are permitted notwithstanding the absence of such consent.
6.9.3 The Indemnified Party will not cause or permit the termination of any right to defend or right of appeal in respect of any Third Party Claim which is or might become the basis of an Indemnity Claim without giving the Indemnifying Party written notice of the contemplated or potential termination in time to grant the Indemnifying Party an opportunity to contest the Third Party Claim.
6.9.4 Each of the Indemnified Party and the Indemnifying Party will make all commercially reasonable efforts to make available to the Party (or other Indemnified Party) who has assumed carriage and control of the negotiation, defence or settlement of a Third Party Claim which is or might reasonably be expected to become the basis of an Indemnity Claim all employees and other Persons under its control whose assistance or evidence is necessary to assist that Party (or other Indemnified Party) in evaluating and defending that Third Party Claim and all documents, records and other materials in its possession or control required for use in the negotiation, defence or settlement of that Third Party Claim.
6.9.5 If the Indemnifying Party acknowledges in writing its obligation, subject to the limits in this Article 6, to satisfy an Indemnity Claim to the extent of any binding determination or settlement in connection with a Third Party Claim, then:
6.9.5.1 the Indemnifying Party will have the right, by written notice delivered to the Indemnified Party within 20 Business Days of receipt by the Indemnifying Party of the Indemnity Notice, and subject to the right of any insurer or other Person to assume carriage and control of the negotiation, defence or settlement of the Third Party Claim, to assume carriage and control of the negotiation, defence or settlement of the Third Party Claim and the conduct of any related legal or administrative proceedings at the expense of the Indemnifying Party and by its own counsel;
6.9.5.2 if the Indemnifying Party elects to assume carriage and control, the Indemnified Party will have the right to participate at its own expense (which will not be indemnified under this Article 6) in the negotiation, defence or settlement of the Third Party Claim assisted by its own counsel; and
6.9.5.3 despite Section 6.9.5.1, the Indemnifying Party will not, except with the Indemnified Party’s prior written consent, settle the Third Party Claim or conduct any related legal or administrative proceeding in a manner that would, in the opinion of the Indemnified Party, acting reasonably, have a material adverse effect on the Indemnified Party.
6.9.6 Despite Section 6.9.2, if the Indemnified Party is, before the completion of related settlement negotiations or legal proceedings, required by applicable Law to make a payment into court, into escrow, or to any third party, with respect to a Third Party Claim which is the basis of an Indemnity Claim, the Indemnified Party may make the required payment and the Indemnifying Party will, promptly after demand by the Indemnified Party, reimburse the Indemnified Party for the required payment made, subject to the limits in this Article 6. If the Indemnifying Party makes that reimbursement, and if the amount of the Indemnity Claim relating to the Third Party Claim in respect of which the required payment was made, as
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
finally determined, is less than the amount that was paid by the Indemnifying Party to the Indemnified Party, the Indemnified Party will promptly pay that surplus amount to the Indemnifying Party. A reimbursement made by the Indemnifying Party under this Section
6.9.7 will reduce the related Claim Amount, if any, payable by the Indemnifying Party under Section 6.10 (Payment of Claims) and, despite Section 6.10, will not be paid to a Buyer Indemnified Party out of the Indemnity Escrow Fund.
6.9.8 If the Indemnified Party has been permitted by the Indemnifying Party to retain or assume the carriage and control of the negotiation, defence or settlement of a Third Party Claim which is the basis of an Indemnity Claim, the Indemnifying Party will not contest the amount of the Loss with respect to that Third Party Claim.
6.9.9 The Indemnifying Party will not be required to indemnify for any Loss relating to a Third Party Claim that is settled or contested in violation of the terms of this Section 6.9.
6.10 Payment of Claims
6.10.1 Once the validity and amount of an Indemnity Claim has been finally determined (the “Claim Amount”), by agreement between the Indemnifying Party and the Indemnified Party or by binding determination or settlement, then:
6.10.1.1 if the Indemnifying Party is a Seller, the Claim Amount will be satisfied, at the option of the Indemnified Party, by either (i) setting off such Claim Amount against the next Purchase Price Instalment(s) due to be made to the Sellers if and to the extent the Buyer has not previously exercised its set off rights with respect to such Claim Amount, or (ii) by direct recourse to the Indemnifying Party whereby the Seller Indemnifying Party will promptly pay the Claim Amount to the Buyer Indemnified Party subject to the limitations set out in Sections 6.1.3.2 and 6.1.4.2; and
6.10.1.2 if the Indemnifying Party is the Buyer, the Indemnifying Party will promptly pay the Claim Amount to the Indemnified Party subject to the limitations set out in Section 6.5.2.
6.11 Indemnity Adjustments to Purchase Price
Any payment made to, or set off right exercised by, a Buyer Indemnified Party under this Article 6 will constitute a decrease to the Purchase Price, and any payment made to a Seller Indemnified Party under this Article 6 will constitute an increase to the Purchase Price, in each case to the extent consistent with applicable Law. Despite Section 2.3 (Allocation of Payments), any decrease in the Purchase Price under this Article 6 will be allocated to the Seller or Sellers making the payment or being subject to the right of set off, and any increase in the Purchase Price under this Article 6 will be allocated to the Seller or Sellers receiving the payment (or to the Seller whose related Seller Indemnified Party is receiving the payment).
6.12 Exclusive Remedy
6.12.1 Subject to Section 2.2.5 and Section 6.12.2, the rights of indemnity in this Article 6 will be the sole and exclusive remedy of any Indemnified Party for any breach of a representation or warranty, or non-performance of any covenant or agreement, contained in this Agreement, or any agreement and document delivered under this Agreement and each Indemnified Party waives any other recourse or remedy it may have in contract, tort or otherwise.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
6.12.2 Nothing in this Section 6.12 will limit or restrict an Indemnified Party from seeking:
6.12.2.1 equitable remedies, under Section 7.15 (Equitable Remedies) or otherwise; or
6.12.2.2 any remedies that may be available to an Indemnified Party in the case of fraud.
6.13 No Right of Subrogation
The Sellers shall have no right of recourse against the Corporation, including without limitation any subrogated right, by virtue of the Sellers’ obligation to provide indemnification in relation to Losses arising from any matter referred to in Section 6.1.1.
6.14 Third Party Indemnification
To ensure that the indemnities provided by each of the Sellers and the Buyer to any Indemnified Parties that are not parties to this Agreement are enforceable, it is agreed by the Parties that each of the Sellers is acting as agent for its Seller Indemnified Parties (that are not Parties), and the Buyer is acting as agent for its Buyer Indemnified Parties (that are not Parties), with respect to the indemnities intended to be given to those Persons under this Article 6. Each of the Sellers and the Buyer agrees that it will hold any right to indemnification that any relevant Indemnified Party is intended to have under this Article 6 in trust for that Person, and that funds received by the Sellers or the Buyer in respect of any Claims under this Article 6 by the relevant Indemnified Party will be held in trust for that Person.
ARTICLE 7
GENERAL
7.1 The Sellers’ Representative
7.1.1 Each Seller appoints Dr. Jeffrey Coull as its representative (the “Sellers’ Representative”) and its true and lawful attorney in fact, with full power and authority in its name and on its behalf:
7.1.1.1 to direct all payments due to be paid to a Designated Paying Agent, by the Buyer to the Sellers from time to time pursuant to this Agreement, save and except for payments that are to be satisfied by an issuance of shares, on behalf of the Sellers and to enure that the payment is directed to a Designated Paying Agent and distribute such payment among the Sellers in accordance with their respective interests therein or as otherwise directed by the relevant Sellers;
7.1.1.2 to act in the absolute discretion of the Sellers’ Representative with respect to all matters relating to this Agreement, including execution and delivery of any amendment of, or supplement to, this Agreement, any waiver of any condition under, or right arising out of, this Agreement, and any termination of this Agreement; and
7.1.1.3 in general, to do all things and to perform all acts, including negotiating, executing and delivering all agreements, certificates, receipts, instructions, and other instruments, contemplated by, or deemed advisable to complete the transactions contemplated by, this Agreement.
Subject to Section 7.1.5, this appointment and grant of power and authority is coupled with an interest and is irrevocable, and will not be terminated by the death or incapacity of any Seller.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
7.1.2 The Sellers hereby irrevocably direct that all payments due to be paid by the Buyer to the Sellers from time to time pursuant to this Agreement, save and except for payments that are to be satisfied by an issuance of shares, shall be paid to the Designated Paying Agent engaged by the Sellers’ Representative on behalf of the Sellers or as the Sellers’ Representative may otherwise direct and this shall be the Buyer’s good and sufficient authority for so doing.
7.1.3 The Sellers’ Representative is serving in this capacity solely for purposes of administrative convenience, and is not personally liable in his capacity as a Sellers’ Representative for any of the obligations of the Sellers under this Agreement, or any other documents or agreement contemplated hereby, and the Buyer agrees that it will not look to the personal assets of the Sellers’ Representative for the satisfaction of any obligations of the Sellers hereunder (except to the extent that such Sellers’ Representative is also a Seller, and then subject to the terms of this Agreement). The Sellers’ Representative and the Sellers agree as between themselves, but without prejudice to any rights the Buyer may have, that the Sellers’ Representative will have no obligation or liability to any Person for any action taken or not taken by the Sellers’ Representative (in its capacity as Sellers’ Representative) under this Section 7.1 in good faith, and the Sellers will jointly and severally indemnify and hold harmless the Sellers’ Representative from any Loss that the Sellers’ Representative may suffer as a result of any such action or omission by the Sellers’ Representative under this Agreement.
7.1.4 The Buyer will be entitled to rely upon any document or other instrument delivered by the Sellers’ Representative, in that capacity, as being authorized by all of the Sellers, and the Buyer will not be liable to any Seller for any action taken or not taken by the Buyer based on that reliance.
7.1.5 The mandate of the Sellers’ Representative under this Section 7.1 will terminate immediately in the event that this Agreement is terminated prior to Closing.
7.1.6 The Sellers’ Representative shall be entitled to engage counsel, accountants and other advisors, the expense of which shall be wholly for the account of the Sellers and not for the account of the Buyer or the Corporation.
7.1.7 The Sellers’ Representative, or any subsequent Person serving as Sellers’ Representative hereunder, may resign from such role upon at least ten days prior written notice to the Buyer. The Sellers shall appoint a replacement within ten days of such resignation. All rights of such Person to indemnification and exculpation hereunder shall survive such resignation.
7.2 Time of Essence
Time is of the essence in all respects of this Agreement.
7.3 Notices
Any Communication must be in writing and either:
7.3.1 delivered personally or by courier;
7.3.2 sent by prepaid registered mail; or
7.3.3 transmitted by e-mail or functionally equivalent electronic means of transmission, charges (if any) prepaid.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Any Communication must be sent to the intended recipient at its address as follows:
to any of the Sellers at:
C/O The Sellers’ Representative
|
Attention: |
Jeffrey Coull |
|
Tel. No.: |
[***] |
|
E-mail: |
[***] |
to the Buyer or to the Corporation at:
Zealand Pharma A/S
Sydmarken 11
DK-2860 Soborg
Denmark
|
Attention: |
Ravinder Chahil, Vice President, General Counsel |
|
Tel. No.: |
[***] |
|
E-mail: |
[***] |
with a copy to:
[***]
|
Attention: |
[***] |
|
Tel. No.: |
[***] |
|
E-mail: |
[***] |
or at any other address as any Party may at any time advise the others by Communication given or made in accordance with this Section 7.3. Any Communication delivered to the Party to whom it is addressed will be deemed to have been given or made and received on the day it is delivered at that Party’s address, provided that if that day is not a Business Day then the Communication will be deemed to have been given or made and received on the next Business Day. Any Communication sent by prepaid registered mail will be deemed to have been given or made and received on the fifth Business Day after which it is mailed. If a strike or lockout of postal employees is then in effect, or generally known to be impending, every Communication must be delivered personally or by courier or transmitted by facsimile, e-mail or functionally equivalent electronic means of transmission. Any Communication transmitted by facsimile, e-mail or other functionally equivalent electronic means of transmission will be deemed to have been given or made and received on the day on which it is transmitted; but if the Communication is transmitted on a day which is not a Business Day or after 5:00 p.m. (local time of the ecipient), the Communication will be deemed to have been given or made and received on the next Business Day. Sending a copy of a Communication to a Party’s legal counsel is for information purposes only and does not constitute delivery of that Communication to that Party, subject to Section 7.12 (Tender). The failure to send a copy of a Communication to a Party’s legal counsel does not invalidate delivery of that Communication to the Party.
7.4 Severability
Each Section of this Agreement is distinct and severable. If any Section of this Agreement, in whole or in part, is or becomes illegal, invalid, void, voidable or unenforceable in any jurisdiction by any court of competent jurisdiction, the illegality, invalidity or unenforceability of that Section, in whole or in part, will not affect:
7.4.1 the legality, validity or enforceability of the remaining Sections of this Agreement, in whole or in part; or
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
7.4.2 the legality, validity or enforceability of that Section, in whole or in part, in any other jurisdiction.
7.5 Submission to Jurisdiction
Without prejudice to the ability of any Party to enforce this Agreement in any other proper jurisdiction, each of the Parties irrevocably and unconditionally submits and attorns to the exclusive jurisdiction of the courts of the Province of Ontario to determine all issues, whether at law or in equity, arising from this Agreement. To the extent permitted by applicable Law, each of the Parties:
7.5.1 irrevocably waives any objection, including any claim of inconvenient forum, that it may now or in the future have to the venue of any legal proceeding arising out of or relating to this Agreement in the courts of that Province of Ontario, or that the subject matter of this Agreement may not be enforced in those courts;
7.5.2 irrevocably agrees not to seek, and waives any right to, judicial review by any court which may be called upon to enforce the judgment of the courts referred to in this Section 7.5, of the substantive merits of any suit, action or proceeding; and
7.5.3 to the extent that Party has or may acquire any immunity from the jurisdiction of any court or from any legal process, whether through service or notice, attachment before judgment, attachment in aid of execution, execution or otherwise, with respect to itself or its property, irrevocably waives that immunity in respect of its obligations under this Agreement.
7.6 Amendment and Waiver
No amendment, discharge, restatement, supplement, termination or waiver of this Agreement or any Section of this Agreement is binding unless it is in writing and executed by the Party to be bound. No waiver of, failure to exercise or delay in exercising, any Section of this Agreement constitutes a waiver of any other Section (whether or not similar) nor does any waiver constitute a continuing waiver unless otherwise expressly provided.
7.7 Further Assurances
Each Party will, at the requesting Party’s expense, execute and deliver any further agreements and documents and provide any further assurances, undertakings and information as may be reasonably required by the requesting Party to give effect to this Agreement.
7.8 Assignment and Enurement
Neither this Agreement nor any right or obligation under this Agreement may be assigned by any Party without the prior written consent of the other Parties. This Agreement enures to the benefit of and is binding upon the Parties and their respective Successors.
7.9 Counterparts and Electronic Delivery
This Agreement may be executed and delivered by the Parties in one or more counterparts, each of which will be an original, and each of which may be delivered by facsimile, e-mail or other functionally equivalent electronic means of transmission, and those counterparts will together constitute one and the same instrument.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
7.10 Electronic Delivery
Delivery of this Agreement by facsimile, e-mail or other functionally equivalent electronic means of transmission constitutes valid and effective delivery.
7.11 Costs and Expenses
7.11.1 Except as otherwise specified in this Agreement, and without limiting the indemnification provisions in Article 6 (Indemnification and Survival), all costs and expenses (including the fees and disbursements of accountants, financial advisors, legal counsel and other professional advisers) incurred in connection with the negotiation and settlement of this Agreement, and the completion of the transactions contemplated by this Agreement, are to be paid by the Party incurring those costs and expenses. All costs and expenses incurred in connection with the retention of, and any services provided by, the Sellers’ Representative and the Designated Paying Agent are to be paid by the Sellers.
7.11.2 Notwithstanding the foregoing, the Buyer covenants and agrees to reimburse the Corporation for all costs incurred by the Corporation in connection with the Reimbursable Matters (as defined below) from August 17, 2019 up to the date of this Agreement provided that:
7.11.2.1 the Sellers’ Representative delivers to the Buyer at or before Closing a statement setting forth all the amount actually incurred in respect of each line item set forth in Schedule 7.11.2 together with a copy of each invoice in respect of such amounts and any other documentation as is reasonably requested by the Buyer; and
7.11.2.2 the maximum total amount to be reimbursed by the Buyer in connection with all Reimbursable Matters is US$[***].
7.11.3 For purposes of Section 7.11.2, “Reimbursable Matters” means the ordinary course operations of the Corporation, management of the Corporation’s patent portfolio and the legal review, negotiation and preparation of this Agreement and any other agreements and documents to be delivered under this Agreement.
7.11.4 The Sellers hereby irrevocably direct the Buyer to pay the maximum amounts set out in this Section 7.11 due to those Persons as set out in the statement contemplated in Section 7.11.2.2. The Sellers, and not the Corporation nor the Buyer, shall pay to the Persons set out in the statement contemplated in Section 7.11.2.2 any amounts due to them in excess of the maximum amount payable by the Buyer pursuant to this Section 7.11.
7.12 Tender
Any tender of documents or money to the Parties under this Agreement may be made upon the Parties or their respective counsel.
7.13 Payment and Currency
Any money to be advanced, paid or tendered by any Party under this Agreement must be advanced, paid or tendered by bank draft, certified cheque or wire transfer of immediately available funds payable to the Person to whom the amount is due. Unless otherwise specified, the word “dollar” and the “$” sign refer to Canadian currency, and all amounts to be advanced, paid, tendered or calculated under this Agreement are to be advanced, paid, tendered or calculated in Canadian currency.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
7.14 Public Announcements
All public announcements, press releases and publicity concerning the execution of this Agreement, or the Closing of the transactions contemplated by this Agreement, must be jointly planned and co- ordinated by the Sellers, the Corporation and the Buyer, and no Party will act unilaterally in this regard without the prior consent of the other Parties unless, and only to the extent that, disclosure is required to meet the timely disclosure obligations of any Party under applicable securities Laws or stock exchange rules in circumstances where prior consultation with the other Parties is not practicable. Nothing in this Section 7.14 will limit the ability of a Party to make any announcements to employees, clients or other Persons having business relations with a Party to the extent the Party, after consultation with the other Parties, reasonably determines in good faith that the announcement is necessary or advisable.
7.15 Equitable Remedies
The Parties acknowledge that the failure to comply with a covenant or obligation contained in this Agreement may give rise to irreparable injury to a Party, inadequately compensable in damages. Accordingly, a Party may seek to enforce the performance of this Agreement by injunction or specific performance upon application to a court of competent jurisdiction without proof of actual damage (and without the requirement of posting bond or other security).
7.16 No Contra Proferentem
This Agreement has been reviewed by each Party’s professional advisors, and revised during the course of negotiations between the Parties. Each Party acknowledges that this Agreement is the product of their joint efforts, that it expresses their agreement, and that, if there is any ambiguity in any of its provisions, no rule of interpretation favouring one Party over another based on authorship will apply.
7.17 Independent Legal Advice
Each of the Sellers acknowledges that it has read and understands the terms and conditions of this Agreement and acknowledges and agrees that it has had the opportunity to seek, and was not prevented or discouraged by any other Party from seeking, any independent legal advice which it considered necessary before the execution and delivery of this Agreement and that, if it did not avail itself of that opportunity before signing this Agreement, it did so voluntarily without any undue pressure, and agrees that its failure to obtain independent legal advice will not be used by it as a defence to the enforcement of its obligations under this Agreement.
7.18 Language
The Parties have expressly required that this Agreement, any Communication and all other contracts, documents and notices relating to this Agreement be drafted in the English language. Les parties ont expressément exigé que la présente convention, toute communication et tous les autres contrats, documents et avis qui y sont afférents soient rédigés en anglais seulement.
7.19 Third Party Beneficiaries
This Agreement is not intended to, and does not, confer any rights or remedies on any Person other than the Parties (and their respective Successors) and the Indemnified Parties. The Parties reserve their right to vary or rescind, at any time and in any way, the rights, if any, granted by or under this Agreement to any Person who is not a Party, without notice to or consent of that Person.
THE REMAINDER OF THIS PAGE IS INTENTIONALLY LEFT BLANK
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Each of the Parties has executed and delivered this Agreement as of the date noted at the beginning of this Agreement.
[SIGNATURE PAGE FOLLOWS]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
|
|
|
|
|
[SELLER] |
Signature page to Share Purchase Agreement
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
|
|
ZEALAND PHARMA A/S | ||
|
|
| ||
|
|
| ||
|
|
Per: |
/s/ Emmanuel Dulac | |
|
|
|
Name: |
Emmanuel Dulac |
|
|
|
Title: |
Chief Executive Officer |
|
|
|
| |
|
|
Per: |
/s/ Martin Nicklasson | |
|
|
|
Name: |
Martin Nicklasson |
|
|
|
Title: |
Chairman of the Board |
Signature page to Share Purchase Agreement
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
|
|
ENCYCLE THERAPEUTICS INC. | ||
|
|
| ||
|
|
| ||
|
|
Per: |
/s/ Jeffrey Coull | |
|
|
|
Name: |
Jeffrey Coull |
|
|
|
Title: |
Director |
|
|
|
|
|
|
|
Per: |
/s/ Raphael Hofstein | |
|
|
|
Name: |
Raphael Hofstein |
|
|
|
Title: |
Chairman of the Board |
Signature page to Share Purchase Agreement
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
SCHEDULE A
TO SHARE PURCHASE AGREEMENT
SELLERS
[***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
SCHEDULE B
TO SHARE PURCHASE AGREEMENT
DISCLOSURE SCHEDULE
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
SCHEDULE B
TO SHARE PURCHASE AGREEMENT
DISCLOSURE SCHEDULE
Schedule 1.1.74
PERMITTED ENCUMBRANCES
1. Nil.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Schedule 3.2.5
RESIDENCE
2. The following Sellers are non-residents of Canada: [***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Schedule 3.3.5
CONSENTS
1. [***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Schedule 3.3.15
FINANCIAL STATEMENTS
[***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Schedule 3.3.16.3
TAX MATTERS: TAX DEFICIENCIES
[***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Schedule 3.3.16.14
CERTAIN INDEBTEDNESS
[***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Schedule 3.3.17
ABSENCE OF CHANGES: EQUIPMENT AND SUPPLIES
[***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Schedule 3.3.18
UNDISCLOSED LIABILITIES
1. [***].
2. Please refer to Disclosure Schedule 3.3.16.14.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Schedule 3.3.19
ABSENCE OF UNUSUAL TRANSACTIONS
[***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Schedule 3.3.20
TITLE TO AND CONDITION OF ASSETS
[***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Schedule 3.3.22
INTELLECTUAL PROPERTY
1. Patents and Patent applications, Trademark registrations and applications, Copyright registrations and applications, and material Software (3.3.22.1):
(a) Issued Patents and Patent applications
Aziridine aldehydes, aziridine-conjugated amino derivatives, aziridine-conjugated biomolecules and processes for their preparation (filed Oct. 22, 2007)
|
Application/ |
|
Jurisdiction |
|
Owner |
|
License |
|
PCT/CA2007/001882 |
|
International |
|
University of Toronto |
|
Yes |
|
US Patent No. 8,143,375 |
|
US |
|
University of Toronto |
|
Yes |
|
CA Patent No. 2,702,605 |
|
Canada |
|
University of Toronto |
|
Yes |
Cyclic amino acid molecules and methods of preparing the same (filed March 16, 2010)
|
Application/ |
|
Jurisdiction |
|
Owner |
|
License |
|
PCT/CA/2010/000408 |
|
International |
|
University of Toronto |
|
Yes |
|
US Patent No. 9,260,479 |
|
US |
|
University of Toronto |
|
Yes |
|
US Patent No. 10,385,099 |
|
US |
|
University of Toronto |
|
Yes |
|
*EP Patent No. 2408802 |
|
Europe |
|
University of Toronto |
|
Yes |
|
CA Patent Appl. No. 2,756,804 |
|
Canada |
|
University of Toronto |
|
Yes |
|
JP Patent No. 5866277 |
|
Japan |
|
University of Toronto |
|
Yes |
|
CN Patent No. 102498124 |
|
China |
|
University of Toronto |
|
Yes |
|
HK Patent No. 12112752.9 |
|
Hong Kong |
|
University of Toronto |
|
Yes |
|
AU Patent No. 2010225426 |
|
Australia |
|
University of Toronto |
|
Yes |
|
IN Patent Appl. No. 7853/DELNP/2011 |
|
India |
|
University of Toronto |
|
Yes |
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
*validated in Great Britain, France, Germany, Ireland, Switzerland, Spain, Portugal, Italy, Norway, Sweden, Denmark, Netherlands, Belgium
Fragment synthesis of cyclic peptides (filed November 11, 2016)
|
Application/ |
|
Jurisdiction |
|
Owner |
|
License |
|
PCT Patent Appl. No. PCT/CA2016/000275 |
|
International |
|
Encycle/ Université de Montreal |
|
Yes |
|
US Pat. Appl. No. 15/775,319 |
|
US |
|
Encycle/ Université de Montreal |
|
Yes |
|
EP Pat. Appl. No. 16863254.5 |
|
Europe |
|
Encycle/ Université de Montreal |
|
Yes |
|
CA Pat. Appl. No. 3,004,714 |
|
Canada |
|
Encycle/ Université de Montreal |
|
Yes |
|
CN Pat. Appl. No. 2016800785451 |
|
China |
|
Encycle/ Université de Montreal |
|
Yes |
Cyclic peptides targeting integrin alpha-4-beta-7 (filed November 11, 2016)
|
Application/ |
|
Jurisdiction |
|
Owner |
|
License |
|
PCT Patent Appl. No. PCT/CA2016/000274 |
|
International |
|
Encycle/ Université de Montreal |
|
Yes |
|
US Pat. Appl. No. 15/775,309 |
|
US |
|
Encycle/ Université de Montreal |
|
Yes |
|
EP Pat. Appl. No. 16863253.7 |
|
Europe |
|
Encycle/ Université de
|
|
Yes |
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
|
|
|
|
|
Montreal |
|
|
|
CA Pat. Appl. No. 3,004,641 |
|
Canada |
|
Encycle/ Université de Montreal |
|
Yes |
|
CN Pat. Appl. No. 2016800791486 |
|
China |
|
Encycle/ Université de Montreal |
|
Yes |
|
JP Pat. Appl. No. 2019-503340 |
|
Japan |
|
Encycle/ Université de Montreal |
|
Yes |
|
KR Pat. Appl. No. 10-2018-7016390 |
|
South Korea |
|
Encycle/ Université de Montreal |
|
Yes |
|
AU Pat. Appl. No. 2016353547 |
|
Australia |
|
Encycle/ Université de Montreal |
|
Yes |
|
IN Pat. Appl. No. 201817021544 |
|
India |
|
Encycle/ Université de Montreal |
|
Yes |
Cyclic peptide multimers targeting integrin alpha-4-beta-7 (filed November 10, 2017)
|
Application/ |
|
Jurisdiction |
|
Owner |
|
License |
|
Intl. Patent Appl. No. PCT/CA2017/000244 |
|
International |
|
Encycle |
|
N/A |
|
US Pat. Appl. No. 16/348,103 |
|
US |
|
Encycle |
|
N/A |
|
EP Pat. Appl. No. 17870529.9 |
|
Europe |
|
Encycle |
|
N/A |
|
CA Pat. Appl. No. 3,042,576 |
|
Canada |
|
Encycle |
|
N/A |
|
CN Pat. Appl. No. 2017800761057 |
|
China |
|
Encycle |
|
N/A |
|
JP Pat. Appl. No. 2019-524203 |
|
Japan |
|
Encycle |
|
N/A |
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
|
IN Pat. Appl. No. 201927019614 |
|
India |
|
Encycle |
|
N/A |
Homodetic cyclic peptide multimers targeting alpha-4-beta-7 integrin (filed May 10, 2018)
|
Application/ |
|
Jurisdiction |
|
Owner |
|
License |
|
Intl. Patent Appl. No. PCT/CA2018/000087 |
|
International |
|
Encycle |
|
N/A |
(b) Trademark registrations and applications
|
Mark |
|
Application/ |
|
Jurisdiction |
|
Owner |
|
License |
|
Encycle word mark |
|
Reg. No. 976,228 |
|
Canada |
|
Encycle |
|
N/A |
|
Encycle word mark |
|
Reg. No. 5,830,828 |
|
US |
|
Encycle |
|
N/A |
(c) Material Software:
NIL.
(d) Domain Names:
http://www.encycletherapeutics.com/
*created August 8, 2011 (owned)
2. Standard Forms of Agreement (3.3.22.2)
[***]
3. Contracts (3.3.22.3)
[***]
4. Confidentiality agreements (3.3.22.4.6)
[***]
5. License rights (3.3.22.4.7)
[***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Schedule 3.3.22.5
[***] AGREEMENT
[***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Schedule 3.3.25
MATERIAL CONTRACTS
[***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Schedule 3.3.26
ACCOUNTS AND POWER OF ATTORNEY
1. Banks or Other Depository (3.3.26.1): [***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Schedule 3.3.29
ENVIRONMENTAL CONDITIONS
1. NIL.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Schedule 3.3.30
SUPPLIERS
1. Please refer to the following list: [***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Schedule 3.3.31
RIGHTS TO USE PERSONAL INFORMATION
1. Claims (3.3.31.3):
NIL.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Schedule 3.3.36
PENSION AND BENEFIT PLANS
1. Encycle Therapeutics Inc. Amended and Restated Stock Option Plan effective as of December 31, 2017.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Schedule 3.3.37
INSURANCE POLICIES
[***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Schedule 7.11.2
COSTS AND EXPENSES
[***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Exhibit 1.1.62
LEAD PRODUCT
[***]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Exhibit “5.2.1.3”
FORM OF CONSULTING AND TRANSITIONAL SERVICES AGREEMENT
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.

CONSULTING AGREEMENT
This Consulting Agreement (the “Agreement”) is made effective as of [dd. Month yyyy] (the “Effective Date”), by and between
(1) ENCYCLE THERAPEUTICS INC., a corporation incorporated under the laws of Ontario (hereinafter “Encycle”) and
(2) with principal place of business at [insert address] , (“ADVISOR”).
Recitals
(A) WHEREAS, ENCYCLE desires to obtain the Consulting Services (as defined below) of ADVISOR in order to obtain the benefit and unique experience and ability of ADVISOR; and
(B) WHEREAS, ADVISOR is willing to render such Consulting Services upon the terms and conditions set forth in this Agreement.
Agreement
NOW, THEREFORE, in consideration of the mutual covenants contained in this Agreement, and other good and valuable consideration, the receipt and sufficiency of which are hereby acknowledged, the Parties, intending to be legally bound, agree as follows:
1. Definitions
Unless otherwise specifically provided in this Agreement, the following terms shall have the following meanings:
1.1. “Affiliates” means, with respect to a Person, any Person that controls, is controlled by or is under common control with such first Person. For purposes of this definition only, “control” means (a) to possess, directly or indirectly, the power to direct the management or policies of a Person, whether through ownership of voting securities or by contract relating to voting rights or corporate governance, or (b) to own, directly or indirectly, more than fifty percent (50%) of the outstanding voting securities or other ownership interest of such Person.
1.2. “Confidential Information” means any and all data, information or material that is provided or communicated by or on behalf of ENCYCLE or its Affiliates, or otherwise becomes known, to ADVISOR, or is collected or generated by or on behalf of ADVISOR, in connection with this Agreement or the activities contemplated hereunder, including, without limitation, information relating to ENCYCLE’s and/or its Affiliates’ business, together with all Results (as defined below).
1.3. “Consulting Services” means the services set forth in Schedule 1, with such modifications to Schedule 1 as ENCYCLE may reasonably request from time to time.
1.4. “Loss” means any and all liabilities, claims, demands, causes of action, damages, loss and expenses, including interest, penalties, and reasonable lawyers’ fees and disbursements.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
1.5. “Parties” means ENCYCLE and ADVISOR and “Party” shall mean either of ENCYCLE or ADVISOR.
1.6. “Person” means an individual, sole proprietorship, partnership, limited partnership, limited liability partnership, corporation, limited liability company, business trust, joint stock company, trust, incorporated association, joint venture or similar entity or organization, including a government or political subdivision, department or agency of a government.
1.7. “Results” means any ideas, inventions, discoveries, know-how, data, documentation, reports, materials, writings, designs, computer software, processes, principles, methods, techniques and other information, recorded in any form, that are discovered, conceived, reduced to practice or otherwise generated as a result of or in connection with the Services by or on behalf of ADVISOR (whether solely or jointly with others) and any patent, trade secret, copyright or other intellectual property rights pertaining to any of the foregoing.
1.8. “Term” means the period beginning on the Effective Date and continuing six (6) months after the Effective Date, with possibility of extension by written consent of both Parties.
2. Scope of Consulting Services
2.1. ENCYCLE hereby retains ADVISOR, and ADVISOR hereby agrees, to provide the Consulting Services. ADVISOR will furnish, at ADVISOR’s own expense (subject to Article 3), any and all materials, equipment, services or supplies necessary or useful to successfully complete the Consulting Services. ADVISOR shall use his or her best reasonable efforts to successfully complete the Consulting Services and shall perform such Consulting Services with the highest professional standards and in compliance with all applicable laws and regulations. The Parties agree that completion of the Consulting Services within the agreed-upon time period is an essential term of this Agreement. The contact person at ENCYCLE for the Consulting Services will be Dr. Rie Schultz Hansen (Interim Chief Scientific Officer) of Zealand Pharma A/S or such other person as is appointed from time to time.
3. Compensation and Out-of- Pocket Expenses
3.1. Compensation. ENCYCLE shall pay ADVISOR a fee of CA $ in consideration of the ADVISOR’s satisfactory performance of the Consulting Services; provided, however, that the total hours worked by ADVISOR, shall not exceed 30 hours without the prior written consent of ENCYCLE. ENCYCLE may request, and ADVISOR may agree in writing, that ADVISOR will perform additional hours as needed from time to time in a rate and basis to be agreed in addition to the flat fee referred to in the clause. In addition, ENCYCLE will reimburse ADVISOR for reasonable, pre-agreed, necessary and verifiable out-of-pocket expenses incurred by ADVISOR in connection with the Consulting Services. Compensation paid by ENCYCLE under this Agreement are stated exclusive of any sales tax, which ADVISOR may be obliged to charge. For the sake of clarity, air travel in excess of 6 hrs. the expenses will have to be pre-approved in writing, and a full working day will be defined as a total of seven (7) hours and will be compensated as such as outlined in Schedule 2
3.2. Invoices. ADVISOR will provide invoices to ENCYCLE for compensation and out-of-pocket expenses as provided in Section Compensation. Each invoice shall be sent by email to finance@zealandpharma.com att: “Dr. Rie Schultz Hansen” and will include: (a) a written progress report, which will describe the work ADVISOR has performed and evaluate the work performed in relation to the goals of the Consulting Services, (b) a reasonable
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
accounting, and supporting documentation, of reimbursable out-of-pocket expenses incurred, and (c) other information as reasonably requested by ZEALAND. Within thirty (30) days after receipt by ENCYCLE of a satisfactory invoice and the other information set forth in this Section 3.2, ENCYCLE will make payment to ADVISOR at the following address:
[Address]
[For payment by wire transfer please also state: (i) Bank Account Number; (ii) Sort Code; (iii) Swift Code; (iv) Bank Name and Address; (v) ADVISOR’s Payment Reference]
3.3. Timing of Invoices. ENCYCLE will have no obligation to reimburse ADVISOR for compensation or out-of-pocket expenses not invoiced to ENCYCLE within sixty (60) days after such compensation or out-of-pocket expenses was incurred or within thirty (30) days after the effective date of termination whichever is earlier.
3.4. Entire Compensation. Except as modified by Section 10.2 due to early termination, ADVISOR agrees that the compensation and out-of-pocket expenses set forth in Section 3.1 represents ENCYCLE’s full and complete obligation for any and all Consulting Services to be rendered pursuant to this Agreement.
4. Relationship of the Parties
4.1. The status of a Party under this Agreement shall be that of an independent contractor. Nothing contained in this Agreement shall be construed as creating a partnership, joint venture or agency relationship between the Parties or, except as otherwise expressly provided in this Agreement, as granting either Party the authority to bind or contract any obligation in the name of or on the account of the other Party or to make any statements, representations, warranties or commitments on behalf of the other Party. ADVISOR agrees that neither this Agreement nor the Consulting Services performed hereunder will entitle ADVISOR to participate in or accrue or receive benefits under any employee benefits plans, group insurance plans or programs, or any other arrangement, program, or policy maintained by ENCYCLE that provides employee benefits. ADVISOR will be solely and unconditionally responsible for paying any and all taxes and assessments relating to any income or other consideration that ADVISOR derives from this Agreement. All persons employed by a Party shall be employees of such Party and not of the other Party and all costs and obligations incurred by reason of any such employment shall be for the account and expense of such Party.
5. Ownership of Results
5.1. ADVISOR will fully disclose all Results to ENCYCLE. ADVISOR agrees that all Results that relate to ENCYCLE’s technology programs are the exclusive property of ENCYCLE (or such Person as ENCYCLE may designate). ADVISOR hereby assigns and transfers, without additional consideration, to ENCYCLE (or such designee) all right, title and interest in and to such Results throughout the world. For the avoidance of doubt, copyrightable Results will be considered “work made for hire” and ENCYCLE shall own all right, title, and interest in and to such Results. At ENCYCLE’s request and expense, ADVISOR will execute documents and take all actions necessary to perfect ENCYCLE’s ownership of Results.
6. Confidential Information
6.1. ADVISOR agrees that he or she will have access to, or become acquainted with, Confidential Information of ENCYCLE and/or its Affiliate(s). Confidential Information may be disclosed
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
either orally, visually, in writing, by delivery of materials containing Confidential Information or in any other form or medium. Confidential Information does not include information that: (a) ADVISOR can prove by competent evidence was known to ADVISOR prior to the Effective Date and that was not subject to confidentiality restrictions, (b) was lawfully obtained from a third party without any confidentiality obligations, or (c) is or becomes part of the public domain through no violation of any obligation of ADVISOR.
6.2. ADVISOR will keep Confidential Information in strict confidence, will use Confidential Information only in connection with the performance of the Consulting Services, and will not, during or after the Term, without ENCYCLE’s prior written consent, disclose, disseminate or otherwise make available Confidential Information. If ADVISOR becomes aware of disclosure or misuse, he or she will immediately notify ENCYCLE and take reasonable steps to prevent further disclosure or misuse.
6.3. Notwithstanding Section 6.2, if ADVISOR is requested to disclose Confidential Information by a lawful judicial or government order, ADVISOR will immediately notify ENCYCLE to permit ENCYCLE to defend against any such order of disclosure, and ADVISOR will assist in such defense to the extent permitted by law. If ADVISOR is thereafter required to disclose Confidential Information, ADVISOR and ENCYCLE will agree to a mutually satisfactory means to disclose such information.
6.4. The obligations of nondisclosure set forth in this Article 6 will commence upon the Effective Date and shall survive and continue for five (5) years after the expiration or termination of this Agreement.
6.5. In addition, ADVISOR expressly acknowledges that ENCYCLE is listed on the regulated market of Nasdaq in New York and Copenhagen and is thus subject to applicable laws and stock exchange regulations, in particular with regard to transparency rules. ADVISOR accordingly understands and agrees that since it may have access to inside information (i.e., information on ENCYCLE that is specific, (ii) non-public and (iii) likely to have a material effect on the market price of ENCYCLE’s securities if and when made public, i.e., that a reasonable investor would consider material in deciding whether to buy, hold or sell ENCYCLE securities), it must comply with applicable securities laws and regulations, which provide, in particular, that certain uses of inside information, communication of inside information or manipulation of the market price of any securities constitute a criminal offence. ADVISOR also acknowledges that, pursuant to European securities law, ENCYCLE may be required to place its name, or the names of its Affiliates and of their respective Representatives, on an “insider list” that ENCYCLE may be required to communicate at any time to the Danish Financial Authorities upon request.
7. Subcontractors
7.1. ADVISOR shall not engage or make use of subcontractors for the purpose of providing the Consulting Services or any other obligations under this Agreement except as expressly authorized by ENCYCLE in writing. Any such permitted subcontract shall be subject to the applicable terms and conditions of this Agreement, and, upon ENCYCLE’s request, ADVISOR shall require any such subcontractor to enter into an agreement, pursuant to which the terms and conditions of this Agreement shall apply directly between such subcontractor and ENCYCLE prior to disclosing to such subcontractor any of Confidential Information, provided, however, that no such subcontract shall release ADVISOR from any of its obligations under this Agreement except to the extent such obligations are satisfactorily performed by such
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
subcontractor in accordance with this Agreement.
8. Use of Name
8.1. Each Party shall not mention or otherwise use the name, insignia, symbol, trademark, trade name or logotype of the other Party or its Affiliates in any publication, press release, promotional material or other form of publicity without the prior written consent of the other Party in each instance (which shall not be unreasonably withheld). The restrictions imposed by this Section shall not prohibit either Party from making any disclosure identifying the other Party that is required by applicable law, rule or regulation or the requirements of a national securities exchange or another similar regulatory body, provided that any such disclosure shall be governed by Article 6.
9. Representations, Warranties and Covenants of ADVISOR
9.1. ADVISOR represents and warrants to ENCYCLE that (a) it has full power and authority, and has taken all necessary actions and has obtained all necessary authorizations, licenses, consents and approvals required, to execute and perform this Agreement, (b) its retention as an ADVISOR by ENCYCLE and its performance of the Consulting Services do not, and will not, breach any agreement that obligates ADVISOR to keep in confidence any trade secrets or confidential information of ADVISOR or any other third party or to refrain from competing, directly or indirectly, with the business of any other party, (c) neither it nor any of its personnel engaged in the Consulting Services has been debarred or is subject to debarment or has otherwise been disqualified or suspended from performing scientific or clinical investigations or otherwise subjected to any restrictions or sanctions by the FDA or any other governmental or regulatory authority or professional body with respect to the performance of scientific or clinical investigations (a “Debarred Person”), and ADVISOR shall not use in any capacity, in connection with the Consulting Services, any Debarred Person; and (d) the Consulting Services are not intended to be part of any US government-funded grants or activities undertaken by ADVISOR and have not been funded in whole or in part by the US government and, consequently, any Results conceived pursuant to this Agreement shall not be subject to the conditions of 37 CFR Parts 401 and 404.
10. Termination and Effect of Termination
10.1. Termination. This Agreement shall terminate without further notice on the expiry of the Term. However, ENCYCLE may terminate this Agreement in its sole discretion at any time upon thirty (30) days prior written notice to ADVISOR.
10.2. Effect of Termination. Upon termination of this Agreement, ADVISOR shall, promptly cease performance of its obligations under this Agreement. The expiration or termination of this Agreement shall be without prejudice to any rights or obligations of the Parties that may have accrued prior to the termination and, except as otherwise expressly provided herein, shall not limit any rights or remedies which may be available by law or otherwise.
a) Upon the termination or expiration of this Agreement, ADVISOR shall at ENCYCLE’s option, either destroy or return to ENCYCLE all Confidential Information received from ENCYCLE, provided, however, that ADVISOR shall be permitted to retain one copy of such Confidential Information for archival purposes.
b) In the event of such termination, ADVISOR will be entitled to receive (a) any compensation due and payable under Article 3 but not yet paid as of the effective date of termination, and
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
(b) payment of out-of- pocket expenses reimbursable under Article 3 that are paid or incurred by ADVISOR prior to the effective date of termination.
c) Any invoice, together with the other information required pursuant to Article 3, must be received no later than thirty (30) days after the effective date of termination. Such payments will constitute full and complete settlement of all claims of ADVISOR against ENCYCLE in connection with this Agreement.
10.3. Extension. This Agreement may be extended, on the same terms, at any time with mutual written consent by both Parties.
11. Indemnification and Insurance
11.1. In addition to any other remedy available to the Parties, each Party shall defend, indemnify and hold harmless the other Party, its Affiliates and its and their respective officers, directors, partners, shareholders, employees and agents from and against any and all Losses incurred by them to the extent resulting from or arising out of or in connection with (a) any breach of any obligation in this Agreement by the other Party, (b) the inaccuracy or breach of any representation or warranty made by the other Party in this Agreement or (c) the enforcement of a Party’s rights under this Article 11, except to the extent such Losses arise as a result of the negligence, fraud, willful misconduct or wrongful act of the Indemnified Party, its Affiliates or its or their respective officers, directors, partners, shareholders, employees or agents.
11.2. ADVISOR shall maintain during the Term insurance coverage of the types and in the amounts typically carried by providers of consultancy services in the pharmaceutical or research area. ADVISOR will provide to ENCYCLE upon written request certificates of insurance evidencing its insurance coverage and limits.
11.3 ENCYCLE agrees that, subject to ADVISOR providing Consulting Services in good faith, ENCYCLE will not hold ADVISOR liable for any financial losses it might incur at any stage through actions taken pursuant to ADVISOR’S Consulting Services.
12. Assignment
12.1. This Agreement may not be assigned by either Party in whole or in part without the prior written consent of the other Party, except that ENCYCLE without such consent may assign this Agreement and its rights and obligations hereunder to any of its Affiliates or any successor in interest (whether by merger, acquisition, asset purchase or otherwise) to all or substantially all of the business to which this Agreement relates. ENCYCLE shall always have the right to perform any or all of its obligations and exercise any or all of its rights under this Agreement through any of its Affiliates.
13. Governing Law and Jurisdiction
13.1. Without prejudice to the ability of any Party to enforce this Agreement in any other proper jurisdiction, each of the Parties irrevocably and unconditionally submits and attorns to the exclusive jurisdiction of the courts of the Province of Ontario, Canada, to determine all issues, whether at law or in equity, arising from this Agreement.
13.2. The Parties hereby irrevocably and unconditionally consent to the exclusive jurisdiction of Ontario, Canada for any action, suit or proceeding arising out of or relating to this
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Agreement, and agree not to commence any action, suit or proceeding related thereto except in such courts. In all events the proceedings will be conducted in the English Language.
14. Notices
14.1. Any notice, request, or other communication permitted or required under this Agreement shall be in writing, shall refer specifically to this Agreement, and shall be deemed given only if hand delivered or sent by an internationally recognised overnight delivery service, costs prepaid, or by facsimile (with transmission confirmed), to the Party to whom notice is to be given at the address set forth in the preamble to this Agreement or at such other address such Party may have provided to the other Party in accordance with this Article 14. Such notice, shall be deemed to have been given as of the date delivered by hand or transmitted by facsimile (with transmission confirmed), or on the second business day (at the place of delivery) after deposit with an internationally recognised overnight delivery service, whichever is the earlier.
15. Survival
15.1. The respective rights and obligations of the parties in Sections 1, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 16 and this Article 15 will survive the termination or expiration of this Agreement to the extent necessary to the intended preservation of such rights and obligations.
16. Entire Agreement and Amendment
16.1. This Agreement constitutes the entire agreement between the Parties with respect to the subject matter of the Agreement. This Agreement supersedes all prior agreements, whether written or oral, with respect to the subject matter of the Agreement. Each Party confirms that it is not relying on any representations, warranties or covenants of the other Party except as specifically set out in this Agreement. Nothing in this Agreement is intended to limit or exclude any liability for fraud. All Schedules referred to in this Agreement are intended to be and are hereby specifically incorporated into and made a part of this Agreement. No modification will be effective unless in writing and signed by authorized representatives of both Parties.
17. Signature
17.1 Signatures of any Party transmitted by facsimile or electronic mail (including, without limitation, electronic mailing of a so-called portable document format or “pdf’ of a scanned counterpart) shall be treated as and deemed to be original signatures for all purposes, and will have the same binding effect as if they were original, signed instruments delivered in person.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
|
Execution |
|
|
|
|
|
Accepted by ADVISOR: |
|
|
|
|
|
Date |
|
|
|
|
|
|
|
|
|
|
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
|
Signed on behalf of Encycle Therapeutics Inc. |
|
|
|
|
|
Date |
|
|
|
|
|
|
|
|
|
|
|
Encycle Therapeutics Inc. |
|
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Schedule 1
CONSULTING AGREEMENT
Consulting Services to be provided:
The scope of the services will be to:
· [***]
· [***]
· Further services to be agreed between the parties in writing.
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Schedule 2
PAYMENT SCHEDULE
ENCYCLE will compensate the ADVISOR for performance of the Consulting Services in accordance with the following schedule:
[***]
Furthermore, ENCYCLE will reimburse ADVISOR for any reasonable and documented out-of-pocket expenses related to the work conducted for ENCYCLE if submitted within sixty (60) days. (Examples are travel expenses to and from meetings, parking expenses etc.). The expenses should not exceed 10% of the value of the total agreed consultancy agreement, unless previously agreed in writing.]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
Exhibit “5.2.1.5”
FORM OF RELEASE
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
RELEASE
|
TO: |
|
Encycle Therapeutics Inc. (the “Corporation”) |
|
|
|
|
|
AND TO: |
|
Zealand Pharma A/S (the “Buyer”) |
|
|
|
|
|
RE: |
|
Share purchase agreement dated October , 2019 between the Buyer, the Corporation, and the Sellers identified in Schedule A to the share purchase agreement (the “Purchase Agreement”) |
For value received, each of the undersigned irrevocably releases the Corporation and Buyer, the directors, officers, employees, and representatives of the Corporation and Buyer and each of their respective legal representatives, successors and assigns (all of those persons and entities being called the “Releasees”) from all claims of any kind which the undersigned or its heirs, executors, administrators, legal representatives, successors and assigns ever had, now have, or may in the future have against any of the Releasees by reason of any cause, whether known or unknown, arising from, connected with, or in any way related to the undersigned having been a shareholder, creditor, employee, consultant and/or representative of the Corporation up to and including the date of this release; but expressly excluding (i) any claims, rights or entitlements which any of the undersigned may have for indemnification under applicable law and (ii) any obligations that the Buyer has to the undersigned arising from, connected with or in any way related to the Purchase Agreement.
In addition, each of the undersigned:
1. represents and warrants that it, he, or she has not assigned and will not assign to any other person or entity any of the claims which the undersigned is releasing in this Release; and
2. agrees not to make any claim or to initiate any proceedings against any person who or entity which, in respect of the claims released by this Release, might claim contribution from, or to be indemnified by, any of the Releasees.
This Release may be executed and delivered in one or more counterparts, each of which will be an original, and each of which may be delivered by facsimile, e-mail or other functionally equivalent electronic means of transmission, and those counterparts will together constitute one and the same instrument.
The provisions of this Release shall enure to the benefit of each of the Releasees and will be binding upon the undersigned and his or her heirs, executors, administrators, legal representatives, successors and assigns.
This Release is governed by, and is to be interpreted in accordance with, the laws of the Province of Ontario and the laws of Canada applicable in that Province.
The undersigned have executed this Release as of , 2019.
[Signature pages follow]
CERTAIN IDENTIFIED INFORMATION HAS BEEN EXCLUDED FROM THIS EXHIBIT BECAUSE IT IS
BOTH (I) NOT MATERIAL AND (II) WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY
DISCLOSED. SUCH EXCLUDED INFORMATION HAS BEEN MARKED WITH “[***]”.
|
|
| |
|
|
|
[Seller] |
Signature page to Release
Exhibit 8.1
SUBSIDIARIES OF THE REGISTRANT
|
Company Summary |
|
Domicile |
|
Ownership |
|
Voting |
|
|
|
|
|
|
|
|
|
|
|
Zealand Pharma A/S subsidiaries |
|
|
|
|
|
|
|
|
ZP Holding SPV K/S |
|
Denmark |
|
100 |
% |
100 |
% |
|
ZP General Partner 1 ApS |
|
Denmark |
|
100 |
% |
100 |
% |
|
Zealand Pharma US Inc. |
|
United States |
|
100 |
% |
100 |
% |
|
Encycle Therapeutics Inc. |
|
Canada |
|
100 |
% |
100 |
% |
|
ZP SPV 3 K/S |
|
Denmark |
|
100 |
% |
100 |
% |
|
ZP General Partner 3 ApS |
|
Denmark |
|
100 |
% |
100 |
% |
|
|
|
|
|
|
|
|
|
|
ZP Holding SPV K/S subsidiaries |
|
|
|
|
|
|
|
|
ZP SPV 1 K/S |
|
Denmark |
|
100 |
% |
100 |
% |
|
ZP General Partner 2 ApS |
|
Denmark |
|
100 |
% |
100 |
% |
EXHIBIT 12.1
CERTIFICATION PURSUANT TO
RULE 13a-14(a)/RULE 15d-14(a) UNDER
THE EXCHANGE ACT AND SECTION 302
OF THE SARBANES-OXLEY ACT OF 2002
I, Emmanuel Dulac, certify that:
1. I have reviewed this annual report on Form 20-F of Zealand Pharma A/S;
2. Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report;
3. Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the company as of, and for, the periods presented in this report;
4. The company’s other certifying officer and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the company and have:
a. designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the company, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared;
b. designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles;
c. evaluated the effectiveness of the company’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and
d. disclosed in this report any change in the company’s internal control over financial reporting that occurred during the period covered by the annual report that has materially affected, or is reasonably likely to materially affect, the company’s internal control over financial reporting; and
5. The company’s other certifying officer and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the company’s auditors and the audit committee of the company’s board of directors (or persons performing the equivalent functions):
a. all significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the company’s ability to record, process, summarize and report financial information; and
b. any fraud, whether or not material, that involves management or other employees who have a significant role in the company’s internal control over financial reporting.
|
Date: March 13, 2020 |
|
|
|
|
|
/s/ Emmanuel Dulac |
|
|
|
|
|
Title: President and Chief Executive Officer |
|
EXHIBIT 12.2
CERTIFICATION PURSUANT TO
RULE 13a-14(a)/RULE 15d-14(a) UNDER THE EXCHANGE ACT
AND SECTION 302
OF THE SARBANES-OXLEY ACT OF 2002
I, Matthew Dallas, certify that:
1. I have reviewed this annual report on Form 20-F of Zealand Pharma A/S;
2. Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report;
3. Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the company as of, and for, the periods presented in this report;
4. The company’s other certifying officer and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the company and have:
a. designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the company, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared;
b. designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles;
c. evaluated the effectiveness of the company’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and
d. disclosed in this report any change in the company’s internal control over financial reporting that occurred during the period covered by the annual report that has materially affected, or is reasonably likely to materially affect, the company’s internal control over financial reporting; and
5. The company’s other certifying officer and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the company’s auditors and the audit committee of the company’s board of directors (or persons performing the equivalent functions):
a. all significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the company’s ability to record, process, summarize and report financial information; and
b. any fraud, whether or not material, that involves management or other employees who have a significant role in the company’s internal control over financial reporting.
|
Date: March 13, 2020 |
|
|
|
|
|
/s/ Matthew Dallas |
|
|
|
|
|
Title: Senior Vice President and Chief Financial Officer |
|
EXHIBIT 13.1
PRINCIPAL EXECUTIVE OFFICER CERTIFICATION
PURSUANT TO 18 U.S.C. SECTION 1350, AS ADOPTED PURSUANT TO SECTION 906 OF THE
SARBANES-OXLEY ACT OF 2002
I, Emmanuel Dulac, Chief Executive Officer of the Company, certify, pursuant to 18 U.S.C. §1350, as adopted pursuant to §906 of the Sarbanes- Oxley Act of 2002, that, to the best of my knowledge:
(1) The Annual Report on Form 20-F of the Company for the period ended December 31, 2019 (the “Report”) fully complies with the requirements of Section 13(a) or 15(d) of the Securities Exchange Act of 1934, as amended; and
(2) The information contained in the Report fairly presents, in all material respects, the financial condition and results of operations of the Company.
|
Date: March 13, 2020 |
|
|
|
|
|
|
|
|
/s/ Emmanuel Dulac |
|
|
|
|
|
Title: President and Chief Executive Officer |
|
EXHIBIT 13.2
PRINCIPAL FINANCIAL OFFICER CERTIFICATION
PURSUANT TO 18 U.S.C. SECTION 1350, AS ADOPTED PURSUANT TO SECTION 906 OF THE
SARBANES-OXLEY ACT OF 2002
I, Matthew Dallas, Chief Financial Officer of the Company, hereby certify, pursuant to 18 U.S.C. § 1350, as adopted pursuant to § 906 of the Sarbanes- Oxley Act of 2002, that, to the best of my knowledge:
(1) The Annual Report on Form 20-F of the Company for the period ended December 31, 2019 (the “Report”) fully complies with the requirements of Section 13(a) or 15(d) of the Securities Exchange Act of 1934, as amended; and
(2) The information contained in the Report fairly presents, in all material respects, the financial condition and results of operations of the Company.
|
Date: March 13, 2020 |
|
|
|
|
|
|
|
|
/s/ Matthew Dallas |
|
|
|
|
|
Title: Senior Vice President and Chief Financial Officer |
|
EXHIBIT 15.2
CONSENT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
We consent to the incorporation by reference in Registration Statement No. 333-230654 on Form F-3 of our report dated March 13, 2020, relating to the financial statements of Zealand Pharma A/S, appearing in this Annual Report on Form 20-F for the year ended December 31, 2019.
Copenhagen, March 13, 2020
Deloitte
Statsautoriseret Revisionspartnerselskab
CVR no. 33963556
|
/s/ Sumit Sudan |
|
/s/ Kåre Valtersdorf |
|
State Authorised |
|
State Authorised |
|
Public Accountant |
|
Public Accountant |